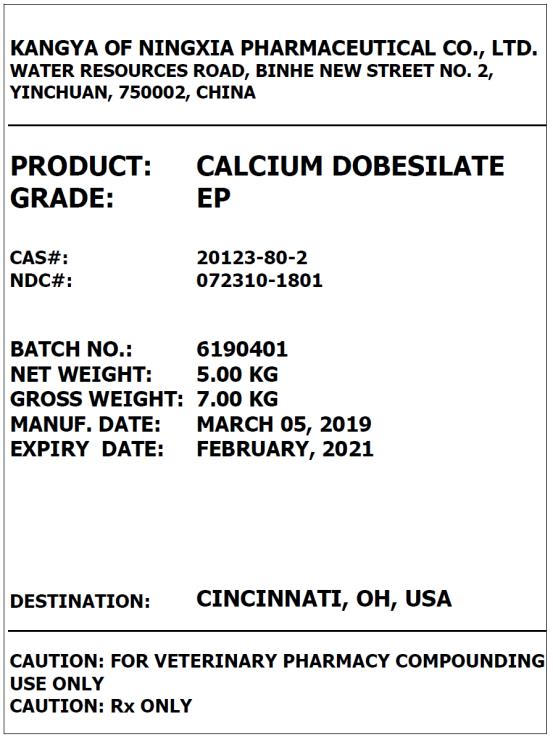
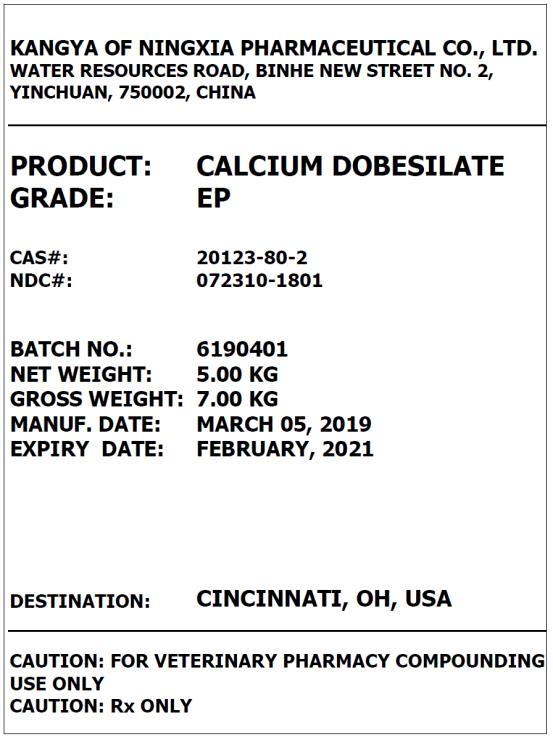
Label: CALCIUM DOBESILATE powder
- NDC Code(s): 72310-1801-1
- Packager: Kangya of Ningxia Pharmaceutical Co., Ltd.
- Category: BULK INGREDIENT
- DEA Schedule: None
- Marketing Status: Bulk Ingredient For Animal Drug Compounding
Drug Label Information
Updated June 25, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL – BULK PRODUCT
-
INGREDIENTS AND APPEARANCE
CALCIUM DOBESILATE
calcium dobesilate powderProduct Information Product Type BULK INGREDIENT Item Code (Source) NDC:72310-1801 Route of Administration NOT APPLICABLE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM DOBESILATE (UNII: 5921X1560Q) (CALCIUM DOBESILATE - UNII:5921X1560Q) CALCIUM DOBESILATE 1 kg in 1 kg Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72310-1801-1 25 kg in 1 DRUM 05/11/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BULK INGREDIENT FOR ANIMAL DRUG COMPOUNDING 05/11/2018 Labeler - Kangya of Ningxia Pharmaceutical Co., Ltd. (547761650)