Label: TERBINAFINE HYDROCHLORIDE cream
- NDC Code(s): 50090-1278-0
- Packager: A-S Medication Solutions
- This is a repackaged label.
- Source NDC Code(s): 51672-2080
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated December 9, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
- Warnings
-
Directions
- adults and children 12 years and over:
- use the tip of the cap to break the seal and open the tube
- wash the affected skin with soap and water and dry completely before applying
-
for athlete's foot wear well-fitting, ventilated shoes.
Change shoes and socks at least once daily.-
between the toes only:
apply twice a day (morning and night) for 1 week or as directed by a doctor -
on the bottom or sides of the foot:
apply twice a day (morning and night) for 2 weeks or as directed by a doctor
-
between the toes only:
-
for jock itch and ringworm:
apply once a day (morning or night) for 1 week or as directed by a doctor - wash hands after each use
- children under 12 years: ask a doctor
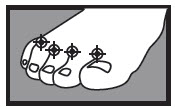 1 week between the toes
1 week between the toes 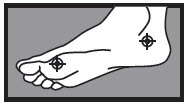 2 weeks on the bottom or sides of the foot
2 weeks on the bottom or sides of the foot - adults and children 12 years and over:
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
- HOW SUPPLIED
- Terbinafine Hydrochloride
-
INGREDIENTS AND APPEARANCE
TERBINAFINE HYDROCHLORIDE
terbinafine hydrochloride creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50090-1278(NDC:51672-2080) Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TERBINAFINE HYDROCHLORIDE (UNII: 012C11ZU6G) (TERBINAFINE - UNII:G7RIW8S0XP) TERBINAFINE HYDROCHLORIDE 1 g in 100 g Inactive Ingredients Ingredient Name Strength benzyl alcohol (UNII: LKG8494WBH) cetyl alcohol (UNII: 936JST6JCN) cetyl palmitate (UNII: 5ZA2S6B08X) isopropyl myristate (UNII: 0RE8K4LNJS) polysorbate 60 (UNII: CAL22UVI4M) water (UNII: 059QF0KO0R) sodium hydroxide (UNII: 55X04QC32I) sorbitan monostearate (UNII: NVZ4I0H58X) stearyl alcohol (UNII: 2KR89I4H1Y) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50090-1278-0 1 in 1 CARTON 11/28/2014 1 15 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077511 07/02/2007 Labeler - A-S Medication Solutions (830016429) Establishment Name Address ID/FEI Business Operations A-S Medication Solutions 830016429 RELABEL(50090-1278)


