Label: WESCAPS- ascorbic acid, thiamine mononitrate, riboflavin, niacinamide, pyridoxine hydrochloride, folic acid, cyanocobalamin, biotin, and calcium pantothenate capsule, gelatin coated
- NHRIC Code(s): 69367-314-01
- Packager: Westminster Pharmaceuticals, LLC
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated December 16, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- HEALTH CLAIM
-
DESCRIPTION
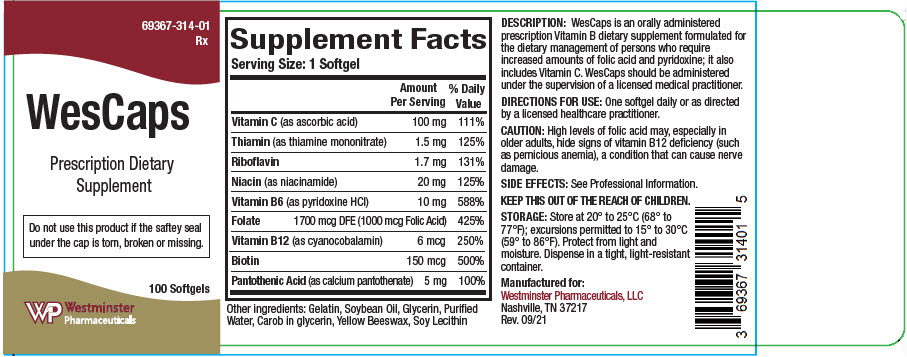
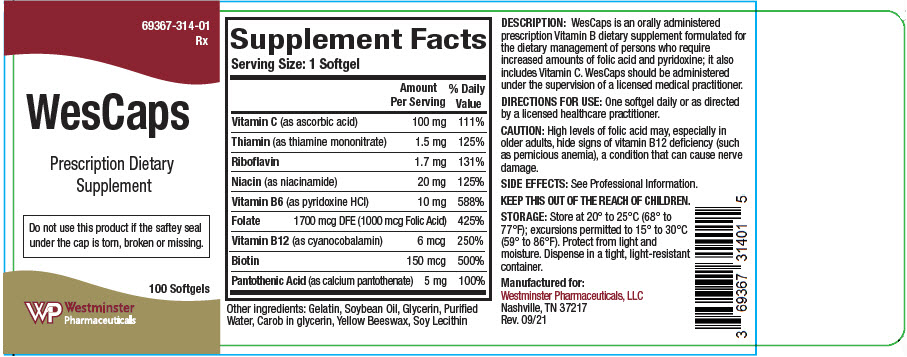
WesCaps is an orally administered prescription Vitamin B dietary supplement formulated for the dietary management of persons who require increases amounts of folic acid and pyridoxine; it also includes vitamin C. WesCaps should be administered under the supervision of a licensed medical practitioner.
-
STATEMENT OF IDENTITY
Supplement Facts Serving Size: 1 Softgel Amount Per Serving % Daily Value Vitamin C (as ascorbic acid) 100 mg 111% Thiamin (as thiamine mononitrate) 1.5 mg 125% Riboflavin 1.7 mg 131% Niacin (as niacinamide) 20 mg 125% Vitamin B6 (as pyridoxine HCl) 10 mg 588% Folate 1700 mcg DFE (1000 mcg Folic Acid) 425% Vitamin B12 (as cyanocobalamin) 6 mcg 250% Biotin 150 mcg 500% Pantothenic Acid (as calcium pantothenate) 5 mg 100% Other ingredients: Gelatin, Soybean Oil, Glycerin, Purified Water, Carob in glycerin, Yellow Beeswax, Soy Lecithin
Side Effects
Allergic reactions have been reported following the use of oral folate.
Mild transient diarrhea, polycythemia vera, itching, transitory exanthema and the feeling of swelling of the entire body has been associated with cobalamin.
Paresthesia, somnolence, nausea, and headaches have been reported with pyridoxine.
KEEP THIS PRODUCT OUT OF THE REACH OF CHILDREN.
-
INTERACTIONS
Drugs which may interact with folate include but are not limited to:
- First generation anticonvulsants (folate may reduce their effectiveness)
- Capecitabine (folate may enhance its toxicity)
- Fluorouracil (folate may enhance its toxicity)
Drugs that may interfere with folate metabolism include, but are not limited to:
- Dihydrofolate Reductase Inhibitors
- Fluoxetine
- Nonsteroidal Anti-inflammatory Drugs
- Sulfasalazine
- Warfarin
Drugs which may interfere with the absorption of folate from the gastrointestinal tract or decrease plasma levels include, but are not limited to:
- Cholestyramine
- Colestipol
- Colchicine
- Levodopa
- Cycloserine
- Isotretinoin
- Oral Contraceptives
- Methylprednisolone
- Pancreatic Enzymes
- Pentamidine
- Smoking and Alcohol
- Sulfasalazine
- Metformin
Drugs which may interact with vitamin B6 include, but are not limited to:
- Levodopa (the action of levodopa may be antagonized by vitamin B6.)
Drugs which may interact with vitamin B12 include, but are not limited to:
- Antibiotics, cholestyramine, colchicine, colestipol, metformin, para-aminosalicylic acid, and potassium chloride (may decrease the absorption of vitamin B12)
- Nitrous oxide (can produce a functional vitamin B12 deficiency)
- CONTRAINDICTIONS
- CAUTION
- PREGNANCY and NURSING MOTHERS
- DIRECTIONS FOR USE
- STORAGE
- HOW SUPPLIED
- HEALTH CLAIM
- HEALTH CLAIM
- PRINCIPAL DISPLAY PANEL - 100 Softgel Bottle Label
-
INGREDIENTS AND APPEARANCE
WESCAPS
ascorbic acid, thiamine mononitrate, riboflavin, niacinamide, pyridoxine hydrochloride, folic acid, cyanocobalamin, biotin, and calcium pantothenate capsule, gelatin coatedProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:69367-314 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 100 mg THIAMINE MONONITRATE (UNII: 8K0I04919X) (THIAMINE ION - UNII:4ABT0J945J) THIAMINE 1.5 mg RIBOFLAVIN (UNII: TLM2976OFR) (RIBOFLAVIN - UNII:TLM2976OFR) RIBOFLAVIN 1.7 mg NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 20 mg PYRIDOXINE HYDROCHLORIDE (UNII: 68Y4CF58BV) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE 10 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 1 mg CYANOCOBALAMIN (UNII: P6YC3EG204) (CYANOCOBALAMIN - UNII:P6YC3EG204) CYANOCOBALAMIN 6 ug BIOTIN (UNII: 6SO6U10H04) (BIOTIN - UNII:6SO6U10H04) BIOTIN 150 ug CALCIUM PANTOTHENATE (UNII: 568ET80C3D) (PANTOTHENIC ACID - UNII:19F5HK2737, CALCIUM CATION - UNII:2M83C4R6ZB) PANTOTHENIC ACID 5 mg Inactive Ingredients Ingredient Name Strength SOYBEAN OIL (UNII: 241ATL177A) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:69367-314-01 100 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 12/13/2021 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color scoring 1 shape size (solid drugs) 8 mm imprint Labeler - Westminster Pharmaceuticals, LLC (079516651)