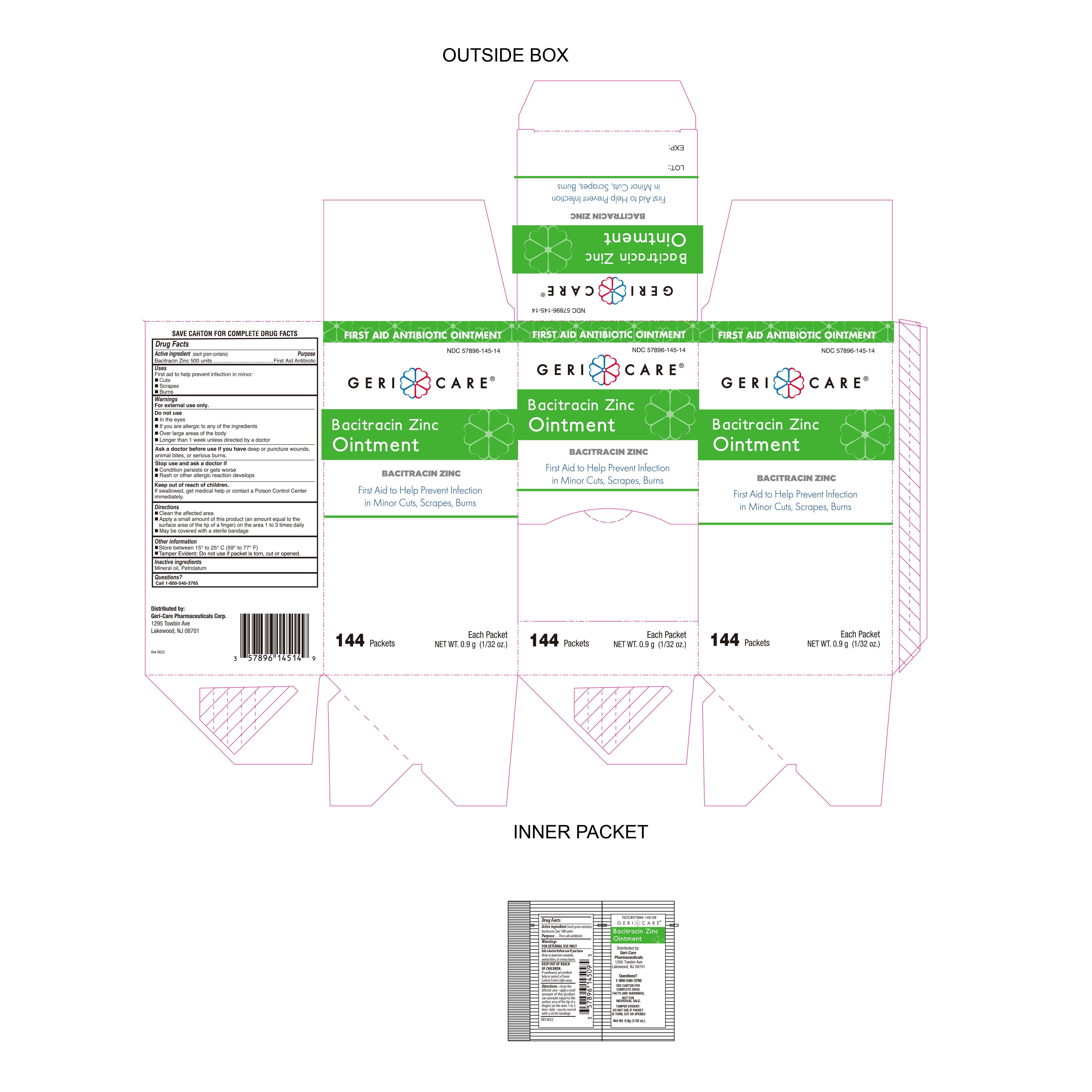
Label: BACITRACIN ZINC ointment
- NDC Code(s): 57896-145-14
- Packager: Geri-Care Pharmaceuticals, Corp.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 2, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
- Warnings
- Do Not Use
- Stop Use and Ask Doctor if
- Directions
- Keep out of reach of children
- Inactive Ingredients
- Questions
- Storage Information
- Other Information
- Packaging
-
INGREDIENTS AND APPEARANCE
BACITRACIN ZINC
bacitracin zinc ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57896-145 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 500 [USP'U] in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) MINERAL OIL (UNII: T5L8T28FGP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57896-145-14 144 in 1 CARTON 04/01/2022 1 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M004 04/01/2022 Labeler - Geri-Care Pharmaceuticals, Corp. (611196254) Registrant - Trifecta Pharmaceuticals USA LLC. (079424163)