Label: BRIMONIDINE TARTRATE solution/ drops
- NDC Code(s): 46708-749-05, 46708-749-10, 46708-749-15
- Packager: Alembic Pharmaceuticals Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated August 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use BRIMONIDINE TARTRATE OPHTHALMIC SOLUTION safely and effectively. See full prescribing information for BRIMONIDINE TARTRATE OPHTHALMIC SOLUTION.
BRIMONIDINE TARTRATE Ophthalmic Solution
Initial U.S. Approval: 1996
INDICATIONS AND USAGE
Brimonidine Tartrate Ophthalmic Solution is an alpha adrenergic receptor agonist indicated for the reduction of elevated intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. (1)
DOSAGE AND ADMINISTRATION
- One drop in the affected eye(s), three times daily, approximately 8 hours apart. (2)
DOSAGE FORMS AND STRENGTHS
- Solution containing 1 mg/mL brimonidine tartrate. (3)
CONTRAINDICATIONS
- Neonates and infants (under the age of 2 years). (4.1)
WARNINGS AND PRECAUTIONS
- Potentiation of vascular insufficiency. (5.1)
ADVERSE REACTIONS
Most common adverse reactions occurring in approximately 5% to 20% of patients receiving brimonidine ophthalmic solution (0.1%-0.2%) included allergic conjunctivitis, burning sensation, conjunctival folliculosis, conjunctival hyperemia, eye pruritus, hypertension, ocular allergic reaction, oral dryness, and visual disturbance. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Alembic Pharmaceuticals, Inc. at 1-866-210-9797 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Antihypertensives/cardiac glycosides may lower blood pressure. (7.1)
- Use with CNS depressants may result in an additive or potentiating effect. (7.2)
- Tricyclic antidepressants may potentially blunt the hypotensive effect of systemic clonidine. (7.3)
- Monoamine oxidase inhibitors may result in increased hypotension. (7.4)
USE IN SPECIFIC POPULATIONS
- Use with caution in children ≥ 2 years of age. (8.4)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 8/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS & USAGE
2 DOSAGE & ADMINISTRATION
3 DOSAGE FORMS & STRENGTHS
4 CONTRAINDICATIONS
4.1 Neonates and Infants (under the age of 2 years)
4.2 Hypersensitivity Reactions
5 WARNINGS AND PRECAUTIONS
5.1 Potentiation of Vascular Insufficiency
5.2 Severe Cardiovascular Disease
5.3 Contamination of Topical Ophthalmic Products After Use
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Antihypertensives/Cardiac Glycosides
7.2 CNS Depressants
7.3 Tricyclic Antidepressants
7.4 Monoamine Oxidase Inhibitors
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Special Populations
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis & Mutagenesis & Impairment Of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS & USAGE
-
2 DOSAGE & ADMINISTRATION
The recommended dose is one drop of brimonidine tartrate ophthalmic solution in the affected eye(s) three times daily, approximately 8 hours apart. Brimonidine tartrate ophthalmic solution may be used concomitantly with other topical ophthalmic drug products to lower intraocular pressure. If more than one topical ophthalmic product is to be used, the different products should be instilled at least 5 minutes apart.
- 3 DOSAGE FORMS & STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Potentiation of Vascular Insufficiency
Brimonidine tartrate ophthalmic solution may potentiate syndromes associated with vascular insufficiency. Brimonidine tartrate ophthalmic solution should be used with caution in patients with depression, cerebral or coronary insufficiency, Raynaud’s phenomenon, orthostatic hypotension, or thromboangiitis obliterans.
5.2 Severe Cardiovascular Disease
Although brimonidine tartrate ophthalmic solution had minimal effect on the blood pressure of patients in clinical studies, caution should be exercised in treating patients with severe cardiovascular disease.
5.3 Contamination of Topical Ophthalmic Products After Use
There have been reports of bacterial keratitis associated with the use of multiple-dose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or a disruption of the ocular epithelial surface (see PATIENT COUNSELING INFORMATION, 17).
-
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
Adverse reactions occurring in approximately 10-20% of the subjects receiving brimonidine ophthalmic solution (0.1-0.2%) included: allergic conjunctivitis, conjunctival hyperemia, and eye pruritus. Adverse reactions occurring in approximately 5-9% included: burning sensation, conjunctival folliculosis, hypertension, ocular allergic reaction, oral dryness, and visual disturbance.
Adverse reactions occurring in approximately 1-4% of the subjects receiving brimonidine ophthalmic solution (0.1-0.2%) included: abnormal taste, allergic reaction, asthenia, blepharitis, blepharoconjunctivitis, blurred vision, bronchitis, cataract, conjunctival edema, conjunctival hemorrhage, conjunctivitis, cough, dizziness, dyspepsia, dyspnea, epiphora, eye discharge, eye dryness, eye irritation, eye pain, eyelid edema, eyelid erythema, fatigue, flu syndrome, follicular conjunctivitis, foreign body sensation, gastrointestinal disorder, headache, hypercholesterolemia, hypotension, infection (primarily colds and respiratory infections), insomnia, keratitis, lid disorder, pharyngitis, photophobia, rash, rhinitis, sinus infection, sinusitis, somnolence, stinging, superficial punctate keratopathy, tearing, visual field defect, vitreous detachment, vitreous disorder, vitreous floaters, and worsened visual acuity.
The following reactions were reported in less than 1% of subjects: corneal erosion, hordeolum, nasal dryness, and taste perversion.
6.2 Postmarketing Experience
The following reactions have been identified during postmarketing use of brimonidine tartrate ophthalmic solutions in clinical practice. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The reactions, which have been chosen for inclusion due to either their seriousness, frequency of reporting, possible causal connection to brimonidine tartrate ophthalmic solutions, or a combination of these factors, include: bradycardia, depression, hypersensitivity, iritis, keratoconjunctivitis sicca, miosis, nausea, skin reactions (including erythema, eyelid pruritus, rash, and vasodilation), syncope, and tachycardia. Apnea, bradycardia, coma, hypotension, hypothermia, hypotonia, lethargy, pallor, respiratory depression, and somnolence have been reported in infants receiving brimonidine tartrate ophthalmic solutions.
-
7 DRUG INTERACTIONS
7.1 Antihypertensives/Cardiac Glycosides
Because brimonidine tartrate ophthalmic solution may reduce blood pressure, caution in using drugs such as antihypertensives and/or cardiac glycosides with brimonidine tartrate ophthalmic solution is advised.
7.2 CNS Depressants
Although specific drug interaction studies have not been conducted with brimonidine tartrate ophthalmic solution, the possibility of an additive or potentiating effect with CNS depressants (alcohol, barbiturates, opiates, sedatives, or anesthetics) should be considered.
7.3 Tricyclic Antidepressants
Tricyclic antidepressants have been reported to blunt the hypotensive effect of systemic clonidine. It is not known whether the concurrent use of these agents with brimonidine tartrate ophthalmic solution in humans can lead to resulting interference with the IOP lowering effect. Caution is advised in patients taking tricyclic antidepressants which can affect the metabolism and uptake of circulating amines.
7.4 Monoamine Oxidase Inhibitors
Monoamine oxidase (MAO) inhibitors may theoretically interfere with the metabolism of brimonidine and potentially result in an increased systemic side-effect such as hypotension. Caution is advised in patients taking MAO inhibitors which can affect the metabolism and uptake of circulating amines
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category B: Teratogenicity studies have been performed in animals.
Brimonidine tartrate was not teratogenic when given orally during gestation days 6 through 15 in rats and days 6 through 18 in rabbits. The highest doses of brimonidine tartrate in rats (2.5 mg/kg/day) and rabbits (5.0 mg/kg/day) achieved AUC exposure values 360- and 20-fold higher, or 260- and 15-fold higher, respectively, than similar values estimated in humans treated with brimonidine tartrate ophthalmic solution 0.1% or 0.15%, 1 drop in both eyes three times daily.
There are no adequate and well-controlled studies in pregnant women; however, in animal studies, brimonidine crossed the placenta and entered into the fetal circulation to a limited extent. Because animal reproduction studies are not always predictive of human response, brimonidine tartrate ophthalmic solution should be used during pregnancy only if the potential benefit to the mother justifies the potential risk to the fetus.
8.3 Nursing Mothers
It is not known whether brimonidine tartrate is excreted in human milk, although in animal studies, brimonidine tartrate has been shown to be excreted in breast milk. Because of the potential for serious adverse reactions from brimonidine tartrate ophthalmic solution in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
Brimonidine tartrate ophthalmic solution is contraindicated in children under the age of 2 years (see CONTRAINDICATIONS, 4.1). During postmarketing surveillance, apnea, bradycardia, coma, hypotension, hypothermia, hypotonia, lethargy, pallor, respiratory depression, and somnolence have been reported in infants receiving brimonidine. The safety and effectiveness of brimonidine tartrate have not been studied in children below the age of 2 years.
In a well-controlled clinical study conducted in pediatric glaucoma patients (ages 2 to 7 years) the most commonly observed adverse reactions with brimonidine tartrate ophthalmic solution 0.2% dosed three times daily were somnolence (50-83% in patients ages 2 to 6 years) and decreased alertness. In pediatric patients 7 years of age (>20 kg), somnolence appears to occur less frequently (25%). Approximately 16% of patients on brimonidine tartrate ophthalmic solution discontinued from the study due to somnolence.
8.5 Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and other adult patients.
8.6 Special Populations
Brimonidine tartrate ophthalmic solution has not been studied in patients with hepatic impairment.
Brimonidine tartrate ophthalmic solution has not been studied in patients with renal impairment. The effect of dialysis on brimonidine pharmacokinetics in patients with renal failure is not known.
-
10 OVERDOSAGE
Very limited information exists on accidental ingestion of brimonidine in adults; the only adverse reaction reported to date has been hypotension. Symptoms of brimonidine overdose have been reported in neonates, infants, and children receiving brimonidine tartrate ophthalmic solution as part of medical treatment of congenital glaucoma or by accidental oral ingestion (see USE IN SPECIFIC POPULATIONS, 8.4). Treatment of an oral overdose includes supportive and symptomatic therapy; a patent airway should be maintained.
-
11 DESCRIPTION
Brimonidine tartrate ophthalmic solution 0.1%, sterile, is a relatively selective alpha-2 adrenergic receptor agonist (topical intraocular pressure lowering agent).
The structural formula of brimonidine tartrate is:
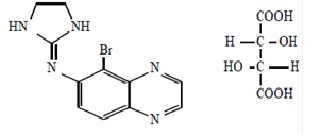
5-bromo-6-(2-imidazolidinylideneamino) quinoxaline L-tartrate; MW= 442.24
In solution, brimonidine tartrate ophthalmic solution has a clear, greenish-yellow color. It has an osmolality of 250-350 mOsmol/kg and a pH of
7.4-8.0.
Brimonidine tartrate appears as wheatish to pale-yellow powder and is soluble in both water (0.6 mg/mL) and in the product vehicle (1.4 mg/mL) at pH 7.7.
Each mL of brimonidine tartrate ophthalmic solution contains the active ingredient brimonidine tartrate 0.1% (1.0 mg/mL) with the inactive ingredients sodium carboxymethylcellulose; sodium borate; boric acid; sodium chloride; potassium chloride; calcium chloride; magnesium chloride; stabilized oxychloro complex 0.005% (0.05 mg/mL) as a preservative; water for injection; and hydrochloric acid and/or sodium hydroxide to adjust pH.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Brimonidine tartrate ophthalmic solution is a relatively selective alpha-2 adrenergic receptor agonist with a peak ocular hypotensive effect occurring at two hours post-dosing.
Fluorophotometric studies in animals and humans suggest that brimonidine tartrate has a dual mechanism of action by reducing aqueous humor production and increasing uveoscleral outflow.
12.3 Pharmacokinetics
Absorption
After ocular administration of either a 0.1% or 0.2% solution, plasma concentrations peaked within 0.5 to 2.5 hours and declined with a systemic half-life of approximately 2 hours.
Distribution
The protein binding of brimonidine has not been studied.
Metabolism
In humans, brimonidine is extensively metabolized by the liver.
Excretion
Urinary excretion is the major route of elimination of brimonidine and its metabolites. Approximately 87% of an orally-administered radioactive dose of brimonidine was eliminated within 120 hours, with 74% found in the urine. -
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis & Mutagenesis & Impairment Of Fertility
No compound-related carcinogenic effects were observed in either mice or rats following a 21month and 24-month study, respectively. In these studies, dietary administration of brimonidine tartrate at doses up to 2.5 mg/kg/day in mice and 1 mg/kg/day in rats achieved 150 and 120 times or 90 and 80 times, respectively, the plasma Cmax drug concentration in humans treated with one drop of brimonidine tartrate ophthalmic solution 0.1% or 0.15% into both eyes 3 times per day, the recommended daily human dose.
Brimonidine tartrate was not mutagenic or clastogenic in a series of in vitro and in vivo studies including the Ames bacterial reversion test, chromosomal aberration assay in Chinese Hamster Ovary (CHO) cells, and three in vivo studies in CD-1 mice: a host-mediated assay, cytogenetic study, and dominant lethal assay.
Reproduction and fertility studies in rats with brimonidine tartrate demonstrated no adverse effect on male or female fertility at doses which achieve up to approximately 125 and 90 times the systemic exposure following the maximum recommended human ophthalmic dose of brimonidine tartrate ophthalmic solution 0.1% or 0.15% respectively.
-
14 CLINICAL STUDIES
Elevated IOP presents a major risk factor in glaucomatous field loss. The higher the level of IOP, the greater the likelihood of optic nerve damage and visual field loss. Brimonidine tartrate has the action of lowering intraocular pressure with minimal effect on cardiovascular and pulmonary parameters.
Clinical study were conducted to evaluate the safety, efficacy, and acceptability of brimonidine tartrate ophthalmic solution, 0.15% compared with ALPHAGAN® administered three-times-daily in patients with open-angle glaucoma or ocular hypertension. Those results indicated that brimonidine tartrate ophthalmic solution, 0.15% is comparable in IOP lowering effect to ALPHAGAN® (brimonidine tartrate ophthalmic solution) 0.2% and effectively lowers IOP in patients with open-angle glaucoma or ocular hypertension by approximately 2-6 mmHg.
A clinical study was conducted to evaluate the safety, efficacy, and acceptability of brimonidine tartrate ophthalmic solution 0.1% compared with ALPHAGAN® administered three-times-daily in patients with open-angle glaucoma or ocular hypertension. Those results indicated that brimonidine tartrate ophthalmic solution 0.1% is equivalent in IOP lowering effect to ALPHAGAN® (brimonidine tartrate ophthalmic solution) 0.2% and effectively lowers IOP in patients with open-angle glaucoma or ocular hypertension by approximately 2-6 mmHg.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Brimonidine tartrate ophthalmic solution, 0.1% is supplied sterile, in white opaque LDPE bottles and tips with purple high density polyethylene (HDPE) caps as follows:
5 mL in 5 mL bottle NDC 46078-749-05
10 mL in 10 mL bottle NDC 46708-749-10
15 mL in 15 mL bottle NDC 46708-749-15
Storage: Store at 15° to 25°C (59° to 77°F).
-
17 PATIENT COUNSELING INFORMATION
Patients should be instructed that ocular solutions, if handled improperly or if the tip of the dispensing container contacts the eye or surrounding structures, can become contaminated by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions (see WARNINGS AND PRECAUTIONS, 5.3). Always replace the cap after using. If solution changes color or becomes cloudy, do not use. Do not use the product after the expiration date marked on the bottle.
Patients also should be advised that if they have ocular surgery or develop an intercurrent ocular condition (e.g., trauma or infection), they should immediately seek their physician's advice concerning the continued use of the present multidose container.
If more than one topical ophthalmic drug is being used, the drugs should be administered at least five minutes apart.
As with other similar medications, brimonidine tartrate ophthalmic solution may cause fatigue and/or drowsiness in some patients. Patients who engage in hazardous activities should be cautioned of the potential for a decrease in mental alertness.
The trademarks referenced herein are owned by their respective companies.
Manufactured by:
Alembic Pharmaceuticals Limited
Karakhadi -391450,
Gujarat, India
Revised: August 2023
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Brimonidine tartrate ophthalmic solution, 0.1% - 5 mL Bottle Label

Brimonidine tartrate ophthalmic solution, 0.1% - 5 mL Carton Label

Brimonidine tartrate ophthalmic solution, 0.1% - 10 mL Bottle Label

Brimonidine tartrate ophthalmic solution, 0.1% - 10 mL Carton Label

Brimonidine tartrate ophthalmic solution, 0.1% - 15 mL Bottle Label

Brimonidine tartrate ophthalmic solution, 0.1% - 15 mL Carton Label

-
INGREDIENTS AND APPEARANCE
BRIMONIDINE TARTRATE
brimonidine tartrate solution/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:46708-749 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BRIMONIDINE TARTRATE (UNII: 4S9CL2DY2H) (BRIMONIDINE - UNII:E6GNX3HHTE) BRIMONIDINE TARTRATE 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength BORIC ACID (UNII: R57ZHV85D4) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) POTASSIUM CHLORIDE (UNII: 660YQ98I10) WATER (UNII: 059QF0KO0R) SODIUM BORATE (UNII: 91MBZ8H3QO) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) SODIUM CHLORIDE (UNII: 451W47IQ8X) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:46708-749-05 1 in 1 CARTON 08/01/2023 1 5 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:46708-749-10 1 in 1 CARTON 08/01/2023 2 10 mL in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:46708-749-15 1 in 1 CARTON 08/01/2023 3 15 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA216909 08/01/2023 Labeler - Alembic Pharmaceuticals Limited (650574663) Establishment Name Address ID/FEI Business Operations Alembic Pharmaceuticals Limited (F3) 675480734 MANUFACTURE(46708-749)






