Label: ALVAIZ- eltrombopag tablet, film coated
- NDC Code(s): 0480-3273-56, 0480-3274-56, 0480-3275-56, 0480-3276-56
- Packager: Teva Pharmaceuticals, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated May 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ALVAIZ safely and effectively. See full prescribing information for ALVAIZ.
ALVAIZ™ (eltrombopag tablets), for oral use
Initial U.S. Approval: 2008WARNING: RISK FOR HEPATIC DECOMPENSATION IN PATIENTS WITH CHRONIC HEPATITIS C and RISK OF HEPATOTOXICITY
See full prescribing information for complete boxed warning.
In patients with chronic hepatitis C, ALVAIZ in combination with interferon and ribavirin may increase the risk of hepatic decompensation. (5.1)
ALVAIZ may increase the risk of severe and potentially life-threatening hepatotoxicity. Monitor hepatic function and discontinue dosing as recommended. (5.2)
INDICATIONS AND USAGE
ALVAIZ is a thrombopoietin receptor agonist indicated:
- for the treatment of thrombocytopenia in adult and pediatric patients 6 years and older with persistent or chronic immune thrombocytopenia (ITP) who have had an insufficient response to corticosteroids, immunoglobulins, or splenectomy. ALVAIZ should be used only in patients with ITP whose degree of thrombocytopenia and clinical condition increase the risk for bleeding. (1.1)
- for the treatment of thrombocytopenia in adult patients with chronic hepatitis C to allow the initiation and maintenance of interferon-based therapy. ALVAIZ should be used only in patients with chronic hepatitis C whose degree of thrombocytopenia prevents the initiation of interferon-based therapy or limits the ability to maintain interferon-based therapy. (1.2)
- for the treatment of adult patients with severe aplastic anemia who have had an insufficient response to immunosuppressive therapy. (1.3)
Limitations of Use:
- ALVAIZ is not indicated for the treatment of patients with myelodysplastic syndrome (MDS). (1.4)
- Safety and efficacy have not been established in combination with direct-acting antiviral agents used without interferon for treatment of chronic hepatitis C infection. (1.4)
DOSAGE AND ADMINISTRATION
- ALVAIZ is not substitutable with other eltrombopag products on a milligram per milligram basis. (2.1)
- Take ALVAIZ without a meal or with a meal low in calcium (≤ 50 mg). Take ALVAIZ at least 2 hours before or 4 hours after any medications or products containing polyvalent cations, such as antacids, calcium-rich foods, and mineral supplements. (2.4, 7.1, 12.3)
- Persistent or Chronic ITP: Initiate ALVAIZ at 36 mg orally once daily for most adult and pediatric patients 6 years and older. Dose reductions are needed for patients with hepatic impairment and some patients of East-/Southeast-Asian ancestry. Adjust to maintain platelet count greater than or equal to 50 x 109/L. Do not exceed 54 mg per day. (2.1, 8.6, 8.7)
- Chronic Hepatitis C-associated Thrombocytopenia: Initiate ALVAIZ at 18 mg orally once daily for all patients. Adjust to achieve target platelet count required to initiate antiviral therapy. Do not exceed a daily dose of 72 mg. (2.2)
- Refractory Severe Aplastic Anemia: Initiate ALVAIZ at 36 mg orally once daily. Reduce initial dose in patients with hepatic impairment or patients of East-/Southeast-Asian ancestry. Adjust to maintain platelet count greater than 50 x 109/L. Do not exceed 108 mg per day. (2.3, 8.6, 8.7)
DOSAGE FORMS AND STRENGTHS
Tablets: 9 mg, 18 mg, 36 mg, and 54 mg (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- Hepatotoxicity: Monitor liver function before and during therapy. (5.2)
- Increased Risk of Death and Progression of Myelodysplastic Syndromes to Acute Myeloid Leukemia. (5.3)
- Thrombotic/Thromboembolic Complications: Portal vein thrombosis has been reported in patients with chronic liver disease receiving eltrombopag. Monitor platelet counts regularly. (5.4)
ADVERSE REACTIONS
Across all indications, the most common adverse reactions (≥ 20% in any indication) were: anemia, nausea, pyrexia, alanine aminotransferase increased, cough, fatigue, headache, and diarrhea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Teva at 1-888-838-2872 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
Lactation: Advise women not to breastfeed during treatment. (8.2)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 5/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: RISK FOR HEPATIC DECOMPENSATION IN PATIENTS WITH CHRONIC HEPATITIS C and RISK OF HEPATOTOXICITY
1 INDICATIONS AND USAGE
1.1 Treatment of Thrombocytopenia in Patients with Persistent or Chronic Immune Thrombocytopenia
1.2 Treatment of Thrombocytopenia in Patients with Hepatitis C Infection
1.3 Treatment of Severe Aplastic Anemia
1.4 Limitations of Use
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage Information
2.2 Recommended Dosage for Persistent or Chronic Immune Thrombocytopenia
2.3 Recommended Dosage for Chronic Hepatitis C-associated Thrombocytopenia
2.4 Recommended Dosage for Refractory Severe Aplastic Anemia
2.5 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hepatic Decompensation in Patients with Chronic Hepatitis C
5.2 Hepatotoxicity
5.3 Increased Risk of Death and Progression of Myelodysplastic Syndromes to Acute Myeloid Leukemia
5.4 Thrombotic/Thromboembolic Complications
5.5 Cataracts
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Polyvalent Cations (Chelation)
7.2 Transporters
7.3 Protease Inhibitors
7.4 Peginterferon Alfa-2a/b Therapy
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
8.7 Ethnicity
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Persistent or Chronic ITP
14.2 Chronic Hepatitis C-associated Thrombocytopenia
14.3 Severe Aplastic Anemia
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: RISK FOR HEPATIC DECOMPENSATION IN PATIENTS WITH CHRONIC HEPATITIS C and RISK OF HEPATOTOXICITY
In patients with chronic hepatitis C, ALVAIZ in combination with interferon and ribavirin may increase the risk of hepatic decompensation [see Warnings and Precautions (5.1)].
ALVAIZ may increase the risk of severe and potentially life-threatening hepatotoxicity. Monitor hepatic function and discontinue dosing as recommended [see Warnings and Precautions (5.2)].
-
1 INDICATIONS AND USAGE
1.1 Treatment of Thrombocytopenia in Patients with Persistent or Chronic Immune Thrombocytopenia
ALVAIZ™ (eltrombopag tablets) are indicated for the treatment of thrombocytopenia in adult and pediatric patients 6 years and older with persistent or chronic immune thrombocytopenia (ITP) who have had an insufficient response to corticosteroids, immunoglobulins, or splenectomy. ALVAIZ should be used only in patients with ITP whose degree of thrombocytopenia and clinical condition increase the risk for bleeding.
1.2 Treatment of Thrombocytopenia in Patients with Hepatitis C Infection
ALVAIZ is indicated for the treatment of thrombocytopenia in adult patients with chronic hepatitis C to allow the initiation and maintenance of interferon-based therapy. ALVAIZ should be used only in patients with chronic hepatitis C whose degree of thrombocytopenia prevents the initiation of interferon-based therapy or limits the ability to maintain interferon-based therapy.
1.3 Treatment of Severe Aplastic Anemia
ALVAIZ is indicated for the treatment of adult patients with severe aplastic anemia who have had an insufficient response to immunosuppressive therapy.
1.4 Limitations of Use
- ALVAIZ is not indicated for the treatment of patients with myelodysplastic syndromes (MDS) [see Warnings and Precautions (5.3)].
- Safety and efficacy have not been established in combination with direct-acting antiviral agents used without interferon for treatment of chronic hepatitis C infection.
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage Information
- Eltrombopag is available for different indications and in different dosage forms and tablet strengths.
- ALVAIZ is not substitutable with other eltrombopag products on a milligram per milligram basis due to the observed bioavailability in studies conducted on ALVAIZ.
- Patients must be able to swallow ALVAIZ tablets whole [see Dosage and Administration (2.5)].
2.2 Recommended Dosage for Persistent or Chronic Immune Thrombocytopenia
Use the lowest dose of ALVAIZ to achieve and maintain a platelet count greater than or equal to 50 x 109/L as necessary to reduce the risk for bleeding. Dose adjustments are based upon the platelet count response. Do not use ALVAIZ to normalize platelet counts [see Warnings and Precautions (5.4)]. In clinical trials, platelet counts generally increased within 1 to 2 weeks after starting ALVAIZ and decreased within 1 to 2 weeks after discontinuing ALVAIZ [see Clinical Studies (14.1)].
Initial Dose Regimen: Adult and Pediatric Patients 6 Years and Older with ITP: Initiate ALVAIZ at a dose of 36 mg orally once daily, except in patients who are of East-/Southeast-Asian ancestry or who have mild to severe hepatic impairment (Child-Pugh Class A, B, C).
For patients of East-/Southeast-Asian ancestry with ITP, initiate ALVAIZ at a reduced dose of 18 mg once daily [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
For patients with ITP and mild, moderate, or severe hepatic impairment (Child-Pugh Class A, B, C), initiate ALVAIZ at a reduced dose of 18 mg once daily [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
For patients of East-/Southeast-Asian ancestry with ITP and hepatic impairment (Child-Pugh Class A, B, C), consider initiating ALVAIZ at a reduced dose of 9 mg once daily [see Clinical Pharmacology (12.3)].
Monitoring and Dose Adjustment: After initiating ALVAIZ, adjust the dose to achieve and maintain a platelet count greater than or equal to 50 x 109/L as necessary to reduce the risk for bleeding. Do not exceed a dose of 54 mg daily. Monitor clinical hematology and liver tests regularly throughout therapy with ALVAIZ and modify the dosage regimen of ALVAIZ based on platelet counts as outlined in Table 1. During therapy with ALVAIZ, assess complete blood counts (CBCs) with differentials, including platelet counts, weekly until a stable platelet count has been achieved. Obtain CBCs with differentials, including platelet counts, monthly thereafter.
Table 1. Dose Adjustments of ALVAIZ in Patients with Persistent or Chronic Immune Thrombocytopenia Platelet Count Result
Dose Adjustment or Response
< 50 x 109/L following at least 2 weeks of ALVAIZ
Increase daily dose by 18 mg to a maximum of 54 mg/day.
For patients taking 9 mg once daily, increase the dose to 18 mg daily before increasing the dose amount by 18 mg.
≥ 200 x 109/L to ≤ 400 x
109/L at any time
Decrease the daily dose by 18 mg. Wait 2 weeks to assess the effects of this and any subsequent dose adjustments.
For patients taking 18 mg once daily, decrease the dose to 9 mg once daily.
> 400 x 109/L
Stop ALVAIZ; increase the frequency of platelet monitoring to twice weekly.
Once the platelet count is < 150 x 109/L, reinitiate therapy at a daily dose reduced by 18 mg.
For patients taking 18 mg once daily, reinitiate therapy at a daily dose of 9 mg.
> 400 x 109/L after 2 weeks of therapy at lowest dose of ALVAIZ
Discontinue ALVAIZ.
In patients with ITP and hepatic impairment (Child-Pugh Class A, B, C), after initiating ALVAIZ or after any subsequent dosing increase, wait 3 weeks before increasing the dose.
Modify the dosage regimen of concomitant ITP medications, as medically appropriate, to avoid excessive increases in platelet counts during therapy with ALVAIZ. Do not administer more than one dose of ALVAIZ within any 24-hour period.
Discontinuation: Discontinue ALVAIZ if the platelet count does not increase to a level sufficient to avoid clinically important bleeding after 4 weeks of therapy with ALVAIZ at the maximum daily dose of 54 mg. Excessive platelet count responses, as outlined in Table 1, or important liver test abnormalities also necessitate discontinuation of ALVAIZ [see Warnings and Precautions (5.2)]. Obtain CBCs with differentials, including platelet counts, weekly for at least 4 weeks following discontinuation of ALVAIZ.
2.3 Recommended Dosage for Chronic Hepatitis C-associated Thrombocytopenia
Use the lowest dose of ALVAIZ to achieve and maintain a platelet count necessary to initiate and maintain antiviral therapy with pegylated interferon and ribavirin. Dose adjustments are based upon the platelet count response. Do not use ALVAIZ to normalize platelet counts [see Warnings and Precautions (5.4)]. In clinical trials, platelet counts generally began to rise within the first week of treatment with ALVAIZ [see Clinical Studies (14.2)].
Initial Dose Regimen: Initiate ALVAIZ at a dose of 18 mg orally once daily.
Monitoring and Dose Adjustment: Adjust the dose of ALVAIZ in 18-mg increments every 2 weeks as necessary to achieve the target platelet count required to initiate antiviral therapy. Monitor platelet counts every week prior to starting antiviral therapy.
During antiviral therapy, adjust the dose of ALVAIZ to avoid dose reductions of peginterferon. Monitor CBCs with differentials, including platelet counts, weekly during antiviral therapy until a stable platelet count is achieved. Monitor platelet counts monthly thereafter. Do not exceed a dose of 72 mg daily. Monitor clinical hematology and liver tests regularly throughout therapy with ALVAIZ.
For specific dosage instructions for peginterferon or ribavirin, refer to their respective prescribing information.
Table 2. Dose Adjustments of ALVAIZ in Adults with Thrombocytopenia Due to Chronic Hepatitis C Platelet Count Result
Dose Adjustment or Response
< 50 x 109/L following at least
2 weeks of ALVAIZIncrease daily dose by 18 mg to a maximum of 72 mg/day.
≥ 200 x 109/L to ≤ 400 x
109/L at any time
Decrease the daily dose by 18 mg.
Wait 2 weeks to assess the effects of this and any subsequent dose adjustments.
> 400 x 109/L
Stop ALVAIZ; increase the frequency of platelet monitoring to twice weekly.
Once the platelet count is < 150 x 109/L, reinitiate therapy at a daily dose reduced by 18 mg.
For patients taking 18 mg once daily, reinitiate therapy at a daily dose of 9 mg.
> 400 x 109/L after 2 weeks of therapy at lowest dose of ALVAIZ
Discontinue ALVAIZ.
Discontinuation: The prescribing information for pegylated interferon and ribavirin include recommendations for antiviral treatment discontinuation for treatment futility. Refer to pegylated interferon and ribavirin prescribing information for discontinuation recommendations for antiviral treatment futility.
ALVAIZ should be discontinued when antiviral therapy is discontinued. Excessive platelet count responses, as outlined in Table 2, or important liver test abnormalities also necessitate discontinuation of ALVAIZ [see Warnings and Precautions (5.2)].
2.4 Recommended Dosage for Refractory Severe Aplastic Anemia
Use the lowest dose of ALVAIZ to achieve and maintain a hematologic response. Dose adjustments are based upon the platelet count. Hematologic response requires dose titration, generally up to 108 mg, and may take up to 16 weeks after starting ALVAIZ [see Clinical Studies (14.3)].
Initial Dose Regimen: Initiate ALVAIZ at a dose of 36 mg orally once daily.
For patients with severe aplastic anemia of East-/Southeast-Asian ancestry or those with mild, moderate, or severe hepatic impairment (Child-Pugh Class A, B, C), initiate ALVAIZ at a reduced dose of 18 mg once daily [see Use in Specific Populations (8.6, 8.7), Clinical Pharmacology (12.3)].
Monitoring and Dose Adjustment: Adjust the dose of ALVAIZ in 36-mg increments every 2 weeks as necessary to achieve the target platelet count greater than or equal to 50 x 109/L as necessary. Do not exceed a dose of 108 mg daily. Monitor clinical hematology and liver tests regularly throughout therapy with ALVAIZ and modify the dosage regimen of ALVAIZ based on platelet counts as outlined in Table 3.
Table 3. Dose Adjustments of ALVAIZ in Patients with Refractory Severe Aplastic Anemia Platelet Count Result
Dose Adjustment or Response
< 50 x 109/L following at least
2 weeks of ALVAIZIncrease daily dose by 36 mg to a maximum of 108 mg/day.
For patients taking 18 mg once daily, increase the dose to 36 mg daily before increasing the dose amount by 36 mg.
≥ 200 x 109/L to ≤ 400 x 109/L
at any timeDecrease the daily dose by 36 mg. Wait 2 weeks to assess the effects of this and any subsequent dose adjustments.
> 400 x 109/L
Stop ALVAIZ for 1 week.
Once the platelet count is < 150 x 109/L, reinitiate therapy at a dose reduced by 36 mg.
> 400 x 109/L after 2 weeks of therapy at lowest dose of ALVAIZ
Discontinue ALVAIZ.
For patients who achieve tri-lineage response, including transfusion independence, lasting at least 8 weeks: the dose of ALVAIZ may be reduced by 50% [see Clinical Studies (14.3)]. If counts remain stable after 8 weeks at the reduced dose, then discontinue ALVAIZ and monitor blood counts. If platelet counts drop to less than 30 x 109/L, hemoglobin to less than 9 g/dL, or absolute neutrophil count (ANC) to less than 0.5 x 109/L, ALVAIZ may be reinitiated at the previous effective dose.
Discontinuation: If no hematologic response has occurred after 16 weeks of therapy with ALVAIZ, discontinue therapy. If new cytogenetic abnormalities are observed, consider discontinuation of ALVAIZ [see Adverse Reactions (6.1)]. Excessive platelet count responses (as outlined in Table 3) or important liver test abnormalities also necessitate discontinuation of ALVAIZ [see Warnings and Precautions (5.2)].
2.5 Administration
Administration of Tablets: Take ALVAIZ without a meal or with a meal low in calcium (≤ 50 mg). Take ALVAIZ at least 2 hours before or 4 hours after other medications (e.g., antacids), calcium-rich foods (containing > 50 mg calcium e.g., dairy products, calcium-fortified juices, and certain fruits and vegetables), or supplements containing polyvalent cations, such as iron, calcium, aluminum, magnesium, selenium, and zinc [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
Swallow tablets whole. Do not split, chew, or crush tablets and mix with food or liquids.
-
3 DOSAGE FORMS AND STRENGTHS
- 9 mg, round, biconvex, blue film-coated tablets, debossed with “TV” on one side and “Z9” on the other side.
- 18 mg, round, biconvex, off-white film-coated tablets debossed with “TV” on one side and “Z18” on the other side.
- 36 mg, round, biconvex, red film-coated tablets, debossed with “TV” on one side and “Z36” on the other side.
- 54 mg, round, biconvex, orange film-coated tablets, debossed with “TV” on one side and “Z54” on the other side.
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Hepatic Decompensation in Patients with Chronic Hepatitis C
In patients with chronic hepatitis C, ALVAIZ in combination with interferon and ribavirin may increase the risk of hepatic decompensation. In two controlled clinical trials in patients with chronic hepatitis C and thrombocytopenia, ascites and encephalopathy occurred more frequently on the arm receiving treatment with eltrombopag plus antivirals (7%) than the placebo plus antivirals arm (4%). Patients with low albumin levels (less than 3.5 g/dL) or Model for End-Stage Liver Disease (MELD) score greater than or equal to 10 at baseline had a greater risk for hepatic decompensation on the arm receiving treatment with eltrombopag plus antivirals. Discontinue ALVAIZ if antiviral therapy is discontinued.
5.2 Hepatotoxicity
ALVAIZ may increase the risk of severe and potentially life-threatening hepatotoxicity [see Adverse Reactions (6.1)]. One patient (< 1%) with ITP treated with eltrombopag in clinical trials experienced drug-induced liver injury. Eleven patients (1%) with chronic hepatitis C treated with eltrombopag in clinical trials experienced drug-induced liver injury.
Treatment of ITP, Chronic Hepatitis C-associated Thrombocytopenia, and Refractory Severe Aplastic Anemia
Measure serum ALT, AST, and bilirubin prior to initiation of ALVAIZ, every 2 weeks during the dose adjustment phase, and monthly following establishment of a stable dose. ALVAIZ inhibits UDP-glucuronosyltransferase (UGT)1A1 and organic anion-transporting polypeptide (OATP)1B1, which may lead to indirect hyperbilirubinemia. If bilirubin is elevated, perform fractionation. Evaluate abnormal serum liver tests with repeat testing within 3 to 5 days. If the abnormalities are confirmed, monitor serum liver tests weekly until resolved or stabilized. Discontinue ALVAIZ if ALT levels increase to greater than or equal to 3 x ULN in patients with normal liver function or greater than or equal to 3 x baseline (or greater than 5 x ULN, whichever is the lower) in patients with pre-treatment elevations in transaminases and are:
- progressively increasing, or
- persistent for greater than or equal to 4 weeks, or
- accompanied by increased direct bilirubin, or
- accompanied by clinical symptoms of liver injury or evidence for hepatic decompensation.
If the potential benefit for reinitiating treatment with ALVAIZ is considered to outweigh the risk for hepatotoxicity, then consider cautiously reintroducing ALVAIZ and measure serum liver tests weekly during the dose adjustment phase. Hepatotoxicity may reoccur if ALVAIZ is reinitiated. If liver test abnormalities persist, worsen, or recur, then permanently discontinue ALVAIZ.
5.3 Increased Risk of Death and Progression of Myelodysplastic Syndromes to Acute Myeloid Leukemia
A randomized, double-blind, placebo-controlled, multicenter trial in patients with International Prognostic Scoring System (IPSS) intermediate-1, intermediate-2 or high risk MDS with thrombocytopenia, receiving azacitidine in combination with either eltrombopag (n = 179) or placebo (n = 177) was terminated due to lack of efficacy and safety reasons, including increased progression to acute myeloid leukemia (AML). Patients received eltrombopag or placebo at a starting dose of 200 mg once daily, up to a maximum of 300 mg once daily, in combination with azacitidine for at least six cycles. The incidence of death (overall survival) was 32% (57/179) in the eltrombopag arm versus 29% (51/177) in the placebo arm (HR [95% CI] = 1.42 [0.97, 2.08], showing an increased relative risk of death in this trial by 42% in the eltrombopag arm). The incidence of progression to AML was 12% (21/179) in the eltrombopag arm versus 6% (10/177) in the placebo arm (HR [95% CI] = 2.66 [1.31, 5.41], showing an increased relative risk of progression to AML in this trial by 166% in the eltrombopag arm).
5.4 Thrombotic/Thromboembolic Complications
Thrombotic/thromboembolic complications may result from increases in platelet counts with ALVAIZ. Reported thrombotic/thromboembolic complications included both venous and arterial events and were observed at low and at normal platelet counts.
Consider the potential for an increased risk of thromboembolism when administering ALVAIZ to patients with known risk factors for thromboembolism (e.g., Factor V Leiden, ATIII deficiency, antiphospholipid syndrome, chronic liver disease). To minimize the risk for thrombotic/thromboembolic complications, do not use ALVAIZ in an attempt to normalize platelet counts. Follow the dose adjustment guidelines to achieve and maintain target platelet counts [see Dosage and Administration (2.1, 2.2, 2.3)].
In two controlled clinical trials in patients with chronic hepatitis C and thrombocytopenia, 3% (31/955) treated with eltrombopag experienced a thrombotic event compared with 1% (5/484) on placebo. The majority of events were of the portal venous system (1% in patients treated with eltrombopag versus less than 1% for placebo).
In a controlled trial in patients with chronic liver disease and thrombocytopenia not related to ITP undergoing elective invasive procedures (N = 292), the risk of thrombotic events was increased in patients treated with 75 mg of eltrombopag once daily. Seven thrombotic complications (six patients) were reported in the group that received eltrombopag and three thrombotic complications were reported in the placebo group (two patients). All of the thrombotic complications reported in the group that received eltrombopag were portal vein thrombosis (PVT). Symptoms of PVT included abdominal pain, nausea, vomiting, and diarrhea. Five of the six patients in the group that received eltrombopag experienced a thrombotic complication within 30 days of completing treatment with eltrombopag and at a platelet count above 200 x 109/L. The risk of portal venous thrombosis was increased in thrombocytopenic patients with chronic liver disease treated with 75 mg of eltrombopag once daily for 2 weeks in preparation for invasive procedures.
5.5 Cataracts
In the three controlled clinical trials in adults with persistent or chronic ITP, cataracts developed or worsened in 15 (7%) patients who received 50 mg of eltrombopag daily and 8 (7%) placebo-group patients. In the extension trial, cataracts developed or worsened in 11% of patients who underwent ocular examination prior to therapy with eltrombopag. In the two controlled clinical trials in patients with chronic hepatitis C and thrombocytopenia, cataracts developed or worsened in 8% of patients treated with eltrombopag and 5% of patients treated with placebo.
Cataracts were observed in toxicology studies of eltrombopag in rodents [see Nonclinical Toxicology (13.2)]. Perform a baseline ocular examination prior to administration of eltrombopag and, during therapy with eltrombopag, regularly monitor patients for signs and symptoms of cataracts.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions associated with ALVAIZ are described in other sections.
- Hepatic Decompensation in Patients with Chronic Hepatitis C [see Warnings and Precautions (5.1)]
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- Increased Risk of Death and Progression of Myelodysplastic Syndromes to Acute Myeloid Leukemia [see Warnings and Precautions (5.3)]
- Thrombotic/Thromboembolic Complications [see Warnings and Precautions (5.4)]
- Cataracts [see Warnings and Precautions (5.5)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ALVAIZ has been established based on adequate and well-controlled studies of eltrombopag olamine in adult and pediatric patients 6 years and older with persistent or chronic ITP, adult patients with chronic hepatitis C-associated thrombocytopenia, and adult patients with refractory severe aplastic anemia. Below is a display of the adverse reactions of eltrombopag olamine in these adequate and well-controlled studies.
Persistent or Chronic Immune Thrombocytopenia: Adults: In clinical trials, hemorrhage was the most common serious adverse reaction and most hemorrhagic reactions followed discontinuation of eltrombopag. Other serious adverse reactions included thrombotic/thromboembolic complications [see Warnings and Precautions (5.4)]. The data described below reflect exposure of eltrombopag to patients with persistent or chronic ITP aged 18 to 85 years, of whom 66% were female, in three placebo-controlled trials and one open-label extension trial [see Clinical Studies (14.1)]. Eltrombopag was administered to 330 patients for at least 6 months and 218 patients for at least 1 year.
Table 4 presents the most common adverse drug reactions (experienced by greater than or equal to 3% of patients receiving eltrombopag) from the three placebo-controlled trials, with a higher incidence in eltrombopag versus placebo.
Table 4. Adverse Reactions (≥ 3%) From Three Placebo-controlled Trials in Adults with Persistent or Chronic Immune Thrombocytopenia Adverse Reaction
Eltrombopag 50 mg
n = 241
(%)Placebo
n = 128
(%)
Nausea
9
3
Diarrhea
9
7
Upper respiratory tract infection
7
6
Vomiting
6
< 1
Urinary tract infectiona
5
4
Increased ALT
5
3
Myalgia
5
2
Oropharyngeal pain
4
3
Increased AST
4
2
Pharyngitis
4
2
Back pain
3
2
Influenza
3
2
Paresthesia
3
2
Rash
3
2
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
a Includes PTs of urinary tract infection, cystitis, urinary tract infection bacterial, and bacteriuria.
In the three controlled clinical persistent or chronic ITP trials, alopecia, musculoskeletal pain, blood alkaline phosphatase increased, and dry mouth were the adverse reactions reported in 2% of patients treated with eltrombopag and in no patients who received placebo.
Among 302 patients with persistent or chronic ITP who received eltrombopag in the single-arm extension trial, the adverse reactions occurred in a pattern similar to that seen in the placebo-controlled trials. Table 5 presents the most common treatment-related adverse reactions (experienced by greater than or equal to 3% of patients receiving eltrombopag) from the extension trial.
Table 5. Treatment-related Adverse Reactions (≥3%) From Extension Trial in Adults with Persistent or Chronic Immune Thrombocytopenia Adverse Reaction
Eltrombopag 50 mg
n = 302
(%)
Headache
10
ALT increased
5
AST increased
5
Cataract
5
Fatigue
5
Blood bilirubin increased
4
Nausea
4
Hyperbilirubinemia
3
Diarrhea
3
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
In the three controlled persistent or chronic ITP trials, serum liver test abnormalities (predominantly Grade 2 or less in severity) were reported in 11% and 7% of patients for eltrombopag and placebo, respectively. Four patients (1%) treated with eltrombopag and three patients in the placebo group (2%) discontinued treatment due to hepatobiliary laboratory abnormalities. Seventeen of the patients treated with eltrombopag in the controlled trials with hepatobiliary laboratory abnormalities were re-exposed to eltrombopag in the extension trial. Eight of these patients again experienced liver test abnormalities (less than or equal to Grade 3) resulting in discontinuation of eltrombopag in one patient. In the extension persistent or chronic ITP trial, six additional patients had eltrombopag discontinued due to liver test abnormalities (less than or equal to Grade 3).
In the three controlled persistent or chronic ITP trials, cataracts developed or worsened in 7% of patients treated with eltrombopag and 7% of patients in the placebo group. All patients had documented, preexisting risk factors for cataractogenesis, including corticosteroid use. In the extension trial, cataracts developed or worsened in 11% of patients who underwent ocular examination prior to therapy with eltrombopag. Seventy-two percent of patients had preexisting risk factors, including corticosteroid use.
The safety of eltrombopag was also assessed in all patients treated in 7 adult persistent or chronic ITP clinical trials (N = 763 eltrombopag-treated patients and 179 placebo-treated patients). Thromboembolic events were reported in 6% of eltrombopag-treated patients versus 0% of placebo-treated patients and thrombotic microangiopathy with acute renal failure was reported in < 1% of eltrombopag-treated patients versus 0% of placebo-treated patients.
In a placebo-controlled trial of eltrombopag in patients with chronic liver disease and thrombocytopenia not related to ITP, six patients treated with eltrombopag and one patient in the placebo group developed portal vein thromboses [see Warnings and Precautions (5.4)].
Pediatric Patients: The data described below reflect median exposure to eltrombopag of 91 days for 107 pediatric patients (aged 1 to 17 years) with persistent or chronic ITP, of whom 53% were female, across the randomized phase of two placebo-controlled trials. ALVAIZ is not indicated for pediatric patients <6 years of age with persistent or chronic ITP.
Table 6 presents the most common adverse drug reactions (experienced by greater than or equal to 3% of pediatric patients 1 year and older receiving eltrombopag) across the two placebo-controlled trials, with a higher incidence for eltrombopag versus placebo.
Table 6. Adverse Reactions (≥ 3%) with a Higher Incidence for Eltrombopag Versus Placebo from Two Placebo-controlled Trials in Pediatric Patients 1 Year and Older with Persistent or Chronic Immune Thrombocytopenia Adverse Reaction
Eltrombopag
n = 107
(%)
Placebo
n = 50
(%)
Upper respiratory tract infection
17
6
Nasopharyngitis
12
4
Cough
9
0
Diarrhea
9
2
Pyrexia
9
8
Abdominal pain
8
4
Oropharyngeal pain
8
2
Toothache
6
0
ALT increaseda
6
0
Rash
5
2
AST increased
4
0
Rhinorrhea
4
0
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
a Includes adverse reactions or laboratory abnormalities > 3 x ULN.
In the two controlled clinical persistent or chronic ITP trials, cataracts developed or worsened in 2 (1%) patients treated with eltrombopag. Both patients had received chronic oral corticosteroids, a risk factor for cataractogenesis.
Chronic Hepatitis C-associated Thrombocytopenia: In the two placebo-controlled trials, 955 patients with chronic hepatitis C-associated thrombocytopenia received eltrombopag. Table 7 presents the most common adverse drug reactions (experienced by greater than or equal to 10% of patients receiving eltrombopag compared with placebo).
Table 7. Adverse Reactions (≥ 10% and Greater Than Placebo) from Two Placebo-controlled Trials in Adults with Chronic Hepatitis C Adverse Reaction
Eltrombopag
+ Peginterferon/Ribavirin
n = 955
(%)
Placebo
+ Peginterferon/Ribavirin
n = 484
(%)
Anemia
40
35
Pyrexia
30
24
Fatigue
28
23
Headache
21
20
Nausea
19
14
Diarrhea
19
11
Decreased appetite
18
14
Influenza-like illness
18
16
Insomniaa
16
15
Asthenia
16
13
Cough
15
12
Pruritus
15
13
Chills
14
9
Myalgia
12
10
Alopecia
10
6
Peripheral edema
10
5
a Includes PTs of insomnia, initial insomnia, and poor quality sleep.
Rash was reported in 9% and 7% of patients receiving eltrombopag and placebo, respectively.
In the two controlled clinical trials in patients with chronic hepatitis C, hyperbilirubinemia was reported in 8% of patients receiving eltrombopag compared with 3% for placebo. Total bilirubin greater than or equal to 1.5 x ULN was reported in 76% and 50% of patients receiving eltrombopag and placebo, respectively. ALT or AST greater than or equal to 3 x ULN was reported in 34% and 38% of patients for eltrombopag and placebo, respectively.
In the two controlled clinical trials in patients with chronic hepatitis C, cataracts developed or worsened in 8% of patients treated with eltrombopag and 5% of patients treated with placebo.
The safety of eltrombopag was also assessed in all patients treated with eltrombopag in the two controlled trials, including patients who initially received eltrombopag in the pre-antiviral treatment phase of the trial and were later randomized to the placebo arm (N = 1520 eltrombopag-treated patients). Hepatic failure was reported in 0.8% of eltrombopag-treated patients and 0.4% of placebo-treated patients.
Severe Aplastic Anemia:
Refractory Severe Aplastic Anemia
In the single-arm, open-label trial, 43 patients with refractory severe aplastic anemia received eltrombopag. Eleven patients (26%) were treated for greater than 6 months and 7 patients (16%) were treated for greater than 1 year. The most common adverse reactions (greater than or equal to 20%) were nausea, fatigue, cough, diarrhea, and headache.
Table 8. Adverse Reactions (≥ 10%) From One Open-label Trial in Adults with Refractory Severe Aplastic Anemia Adverse Reaction
Eltrombopag
n = 43
(%)
Nausea
33
Fatigue
28
Cough
23
Diarrhea
21
Headache
21
Pain in extremity
19
Pyrexia
14
Dizziness
14
Oropharyngeal pain
14
Abdominal pain
12
Muscle spasms
12
Transaminases increased
12
Arthralgia
12
Rhinorrhea
12
Rash and hyperbilirubinemia were reported in 7% of patients; cataract was reported in 2% of patients.
In this trial, concurrent ALT or AST greater than 3 x ULN with total bilirubin greater than 1.5 x ULN were reported in 5% of patients. Total bilirubin greater than 1.5 x ULN occurred in 14% of patients.
In this trial, patients had bone marrow aspirates evaluated for cytogenetic abnormalities. Eight patients had a new cytogenetic abnormality reported on therapy, including 5 patients who had complex changes in chromosome 7.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of eltrombopag. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
Skin and Subcutaneous Tissue Disorders: Skin discoloration, including hyperpigmentation and skin yellowing.
-
7 DRUG INTERACTIONS
7.1 Polyvalent Cations (Chelation)
Eltrombopag chelates polyvalent cations (such as iron, calcium, aluminum, magnesium, selenium, and zinc) in foods, mineral supplements, and antacids.
Take ALVAIZ at least 2 hours before or 4 hours after any medications or products containing polyvalent cations, such as antacids, dairy products, and mineral supplements to avoid significant reduction in absorption of ALVAIZ due to chelation [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
7.2 Transporters
Use caution when concomitantly administering ALVAIZ and drugs that are substrates of OATP1B1 (e.g., atorvastatin, bosentan, ezetimibe, fluvastatin, glyburide, olmesartan, pitavastatin, pravastatin, rosuvastatin, repaglinide, rifampin, simvastatin acid, SN-38 [active metabolite of irinotecan], valsartan) or breast cancer resistance protein (BCRP) (e.g., imatinib, irinotecan, lapatinib, methotrexate, mitoxantrone, rosuvastatin, sulfasalazine, topotecan). Monitor patients closely for signs and symptoms of excessive exposure to the drugs that are substrates of OATP1B1 or BCRP and consider reduction of the dose of these drugs, if appropriate. In clinical trials with eltrombopag, a dose reduction of rosuvastatin by 50% was recommended.
7.3 Protease Inhibitors
HIV Protease Inhibitors: No dose adjustment is recommended when ALVAIZ is coadministered with lopinavir/ritonavir (LPV/RTV). Drug interactions with other HIV protease inhibitors have not been evaluated.
Hepatitis C Virus Protease Inhibitors: No dose adjustments are recommended when ALVAIZ is coadministered with boceprevir or telaprevir. Drug interactions with other hepatitis C virus (HCV) protease inhibitors have not been evaluated.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from a small number of published case reports and postmarketing experience with eltrombopag use in pregnant women are insufficient to assess any drug-associated risks for major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction and developmental toxicity studies, oral administration of eltrombopag to pregnant rats during organogenesis resulted in embryolethality and reduced fetal weights at maternally toxic doses. These effects were observed at doses resulting in exposures that were six times the human clinical exposure based on area under the curve (AUC) in patients with persistent or chronic ITP at 75 mg/day, and three times the AUC in patients with chronic hepatitis C at 100 mg/day (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and of miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In an early embryonic development study, female rats received oral eltrombopag at doses of 10, 20, or 60 mg/kg/day (0.8, 2, and 6 times, respectively, the human clinical exposure based on AUC in patients with ITP at 75 mg/day and 0.3, 1, and 3 times, respectively, the human clinical exposure based on AUC in patients with chronic hepatitis C at 100 mg/day). Increased pre- and post-implantation loss and reduced fetal weight were observed at the highest dose which also caused maternal toxicity.
In an embryo-fetal development study eltrombopag was administered orally to pregnant rats during the period of organogenesis at doses of 10, 20, or 60 mg/kg/day (0.8, 2, and 6 times, respectively, the human clinical exposure based on AUC in patients with ITP at 75 mg/day and 0.3, 1, and 3 times, respectively, the human clinical exposure based on AUC in patients with chronic hepatitis C at 100 mg/day). Decreased fetal weights (6% to 7%) and a slight increase in the presence of cervical ribs were observed at the highest dose which also caused maternal toxicity. However, no evidence of major structural malformations was observed.
In an embryo-fetal development study eltrombopag was administered orally to pregnant rabbits during the period of organogenesis at doses of 30, 80, or 150 mg/kg/day (0.04, 0.3, and 0.5 times, respectively, the human clinical exposure based on AUC in patients with ITP at 75 mg/day and 0.02, 0.1, and 0.3 times, respectively, the human clinical exposure based on AUC in patients with chronic hepatitis C at 100 mg/day). No evidence of fetotoxicity, embryolethality, or teratogenicity was observed.
In a pre- and post-natal developmental toxicity study in pregnant rats (F0), oral eltrombopag was administered from gestation Day 6 through lactation Day 20. No adverse effects on maternal reproductive function or on the development of the offspring (F1) were observed at doses up to 20 mg/kg/day (2 times the human clinical exposure based on AUC in patients with ITP at 75 mg/day and similar to the human clinical exposure based on AUC in patients with chronic hepatitis C at 100 mg/day). Eltrombopag was detected in the plasma of offspring (F1). The plasma concentrations in pups increased with dose following administration of drug to the F0 dams.
8.2 Lactation
Risk Summary
There are no data regarding the presence of eltrombopag or its metabolites in human milk, the effects on the breastfed child, or the effects on milk production. However, eltrombopag was detected in the pups of lactating rats 10 days postpartum suggesting the potential for transfer during lactation. Due to the potential for serious adverse reactions in a breastfed child from eltrombopag, breastfeeding is not recommended during treatment.
8.3 Females and Males of Reproductive Potential
Contraception
Based on animal reproduction studies, eltrombopag can cause fetal harm when administered to a pregnant woman. Sexually-active females of reproductive potential should use effective contraception (methods that result in less than 1% pregnancy rates) when using ALVAIZ during treatment and for at least 7 days after stopping treatment with ALVAIZ.
8.4 Pediatric Use
The safety and effectiveness of ALVAIZ have been established in pediatric patients 6 years and older with persistent or chronic ITP.
The safety and effectiveness of eltrombopag in pediatric patients 6 years and older with persistent or chronic ITP were evaluated in two double-blind, placebo-controlled trials [see Adverse Reactions (6.1), Clinical Studies (14.1)]. The pharmacokinetics of eltrombopag have been established in 130 pediatric patients 6 years and older with ITP dosed once daily [see Clinical Pharmacology (12.3)]. See Dosage and Administration (2.1) for dosing recommendations for pediatric patients 6 years and older.
The safety and effectiveness of ALVAIZ have not been established in pediatric patients less than 6 years of age with persistent or chronic ITP. Pediatric patients must be able to swallow ALVAIZ tablets whole [see Dosage and Administration (2.5)].
The safety and effectiveness in pediatric patients with thrombocytopenia associated with chronic hepatitis C and refractory severe aplastic anemia have not been established.
8.5 Geriatric Use
Of the 106 patients in two randomized clinical trials of eltrombopag 50 mg in persistent or chronic ITP, 22% were 65 years of age and over, while 9% were 75 years of age and over. Of the 1439 patients in two randomized clinical trials of eltrombopag in patients with chronic hepatitis C and thrombocytopenia, 7% were 65 years of age and over, while < 1% were 75 years of age and over. Of the 196 patients who received eltrombopag for the treatment of severe aplastic anemia, 18% were 65 years of age and over, while 3% were 75 years of age and over. No overall differences in safety or effectiveness were observed between these patients and younger patients.
8.6 Hepatic Impairment
Patients with Persistent or Chronic ITP and Severe Aplastic Anemia
Reduce the initial dose of eltrombopag in patients with persistent or chronic ITP (adult and pediatric patients 6 years and older only) or refractory severe aplastic anemia who also have hepatic impairment (Child-Pugh Class A, B, C) [see Dosage and Administration (2.1, 2.3), Warnings and Precautions (5.2), Clinical Pharmacology (12.3)].
In a clinical trial in patients with severe aplastic anemia who had not received prior definitive immunosuppressive therapy, patients with baseline ALT or AST > 5 x ULN were ineligible to participate [see Dosage and Administration (2.3), Warnings and Precautions (5.2), Clinical Pharmacology (12.3)].
Patients with Chronic Hepatitis C
No dosage adjustment is recommended in patients with chronic hepatitis C and hepatic impairment [see Clinical Pharmacology (12.3)].
8.7 Ethnicity
Reduce the initial dose of ALVAIZ for patients of East- /Southeast-Asian ancestry with ITP (adult and pediatric patients 6 years and older only) or adults with severe aplastic anemia [see Dosage and Administration (2.1, 2.3), Clinical Pharmacology (12.3)]. No reduction in the initial dose of ALVAIZ is recommended in patients of East-/Southeast-Asian ancestry with chronic hepatitis C [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
In the event of overdose, platelet counts may increase excessively and result in thrombotic/thromboembolic complications.
In one report, a subject who ingested 5000 mg of eltrombopag had a platelet count increase to a maximum of 929 x 109/L at 13 days following the ingestion. The patient also experienced rash, bradycardia, ALT/AST elevations, and fatigue. The patient was treated with gastric lavage, oral lactulose, intravenous fluids, omeprazole, atropine, furosemide, calcium, dexamethasone, and plasmapheresis; however, the abnormal platelet count and liver test abnormalities persisted for 3 weeks. After 2 months’ follow-up, all events had resolved without sequelae.
In case of an overdose, consider oral administration of a metal cation-containing preparation, such as calcium, aluminum, or magnesium preparations to chelate eltrombopag and thus limit absorption. Closely monitor platelet counts. Reinitiate treatment with eltrombopag in accordance with dosing and administration recommendations [see Dosage and Administration (2.1, 2.2)].
-
11 DESCRIPTION
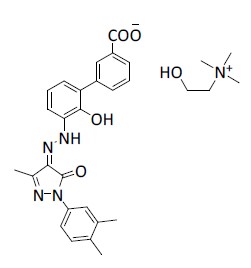
ALVAIZ (eltrombopag) tablets contain eltrombopag choline, a small molecule thrombopoietin (TPO) receptor agonist for oral administration.
Eltrombopag choline is a biphenyl hydrazone. The chemical name for eltrombopag choline is (Z)-3’-(2-(1-(3,4-dimethylphenyl)-3-methyl-5-oxo-1h-Pyrazol-4(5h)-ylidene)hydrazinyl)-2’-hydroxybiphenyl-3-carboxylic acid -choline (1:1). It has the molecular formula C30H35N5O5. The molecular weight is 545.63 g/mol for eltrombopag choline and 442.47 g/mol for eltrombopag free acid.
Eltrombopag choline has the following structural formula:
Eltrombopag choline is practically insoluble in aqueous buffer across a pH range of 1.2 to 6.8.
ALVAIZ (eltrombopag) tablets contain 9 mg, 18 mg, 36 mg, or 54 mg of eltrombopag, supplied as 11.1 mg, 22.2 mg, 44.4 mg, and 66.6 mg of eltrombopag choline, respectively. The inactive ingredients of ALVAIZ tablets are:
Tablet Core: anhydrous lactose, copovidone, croscarmellose sodium, edetate disodium dihydrate, magnesium stearate, poloxamer 188, polyethylene glycol 4000, povidone K12, silicified microcrystalline cellulose, and silicon dioxide.
Coating:
Strength
9 mg:
FD&C Blue No. 1 aluminum lake, polyethylene glycol 3350, polyvinyl alcohol, talc, and titanium dioxide.
18 mg:
Hypromellose 2910, polyethylene glycol 4000, talc, and titanium dioxide.
36 mg:
Iron oxide red, iron oxide yellow, polyethylene glycol 3350, polyvinyl alcohol, talc, and titanium dioxide.
54 mg:
FD&C Red #40 aluminum lake, hypromellose 2910, iron oxide yellow, polyethylene glycol 400, and titanium dioxide.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Eltrombopag is a TPO-receptor agonist that interacts with the transmembrane domain of the human TPO-receptor (also known as cMpl) and initiates signaling cascades that induce proliferation and differentiation of megakaryocytes leading to increased platelet production.
12.2 Pharmacodynamics
In clinical trials, treatment with eltrombopag resulted in dose-dependent increases in platelet counts following repeated (daily) dosing. The increase in platelet counts reached a maximum approximately two weeks after the initiation of dosing, and returned to baseline within approximately two weeks after the last dose of eltrombopag.
Cardiac Electrophysiology
At doses up to 150 mg (the maximum recommended dose) daily for 5 days, eltrombopag did not prolong the QT/QTc interval to any relevant extent.
12.3 Pharmacokinetics
Eltrombopag demonstrated a dose-proportional increase in exposure between doses of 50 to 150 mg/day in healthy adult subjects. Eltrombopag AUC was approximately 1.7-fold higher in patients with persistent or chronic ITP and approximately 2.8-fold higher in patients with HCV compared to healthy subjects. Steady-state was achieved after approximately 1 week of once daily treatment, with geometric mean accumulation ratio of 1.56 (90% confidence interval 1.20, 1.63) at 75 mg/day. Eltrombopag AUC was approximately 3.2-fold higher in patients with definitive immunosuppressive therapy-naïve severe aplastic anemia compared to healthy subjects suggesting higher relative exposure compared to healthy subjects or patients with ITP and similar exposure compared to patients with chronic hepatitis C.
Absorption
Eltrombopag is absorbed with a peak concentration occurring 1.5 to 5.5 hours after oral administration under fasting conditions and 1.0 to 8.0 hours after oral administration under fed conditions (high-fat, high-calorie meal).
Effect of Food
Administration of eltrombopag after a high fat, high calorie meal decreased AUCinf by approximately 36% and Cmax by approximately 39%, and delayed Tmax by 0.5 hours as compared to fasted conditions.
A meal low in calcium (≤ 50 mg calcium) did not significantly impact plasma eltrombopag exposure, regardless of calorie and fat content.
Distribution
The concentration of eltrombopag in blood cells is approximately 50% to 79% of plasma concentrations based on a radiolabel study. In vitro studies suggest that eltrombopag is highly bound to human plasma proteins (greater than 99%). Eltrombopag is a substrate of BCRP, but is not a substrate for P-glycoprotein (P-gp) or OATP1B1.
Elimination
The plasma elimination half-life of eltrombopag is approximately 21 to 32 hours in healthy subjects and 26 to 35 hours in patients with ITP.
Metabolism: Absorbed eltrombopag is extensively metabolized, predominantly through pathways, including cleavage, oxidation, and conjugation with glucuronic acid, glutathione, or cysteine. In vitro studies suggest that CYP1A2 and CYP2C8 are responsible for the oxidative metabolism of eltrombopag. UGT1A1 and UGT1A3 are responsible for the glucuronidation of eltrombopag.
Excretion: The predominant route of eltrombopag excretion is via feces (59%), and 31% of the dose is found in the urine. Unchanged eltrombopag in feces accounts for approximately 20% of the dose; unchanged eltrombopag is not detectable in urine.
Specific Populations
Ethnicity
Eltrombopag concentrations in East-/Southeast-Asian ancestry patients with ITP or chronic hepatitis C, were 50% to 55% higher compared with non-Asian subjects [see Dosage and Administration (2.1, 2.3)].
Eltrombopag exposure in healthy African-American subjects was approximately 40% higher than that observed in Caucasian subjects in one clinical pharmacology trial and similar in three other clinical pharmacology trials. The effect of African-American ethnicity on exposure and related safety and efficacy of eltrombopag has not been established.
Hepatic Impairment
Following a single dose of eltrombopag, plasma eltrombopag AUC0-INF was 41% higher in patients with mild hepatic impairment (Child-Pugh Class A) compared with subjects with normal hepatic function.
Plasma eltrombopag AUC0-INF was approximately 2-fold higher in patients with moderate (Child-Pugh Class B) and severe hepatic impairment (Child-Pugh Class C) compared with subjects with normal hepatic function. The half-life of eltrombopag was prolonged 2-fold in these patients. This clinical trial did not evaluate protein-binding effects.
Chronic Liver Disease
Following repeat doses of eltrombopag in patients with thrombocytopenia and with chronic liver disease, mild hepatic impairment resulted in an 87% to 110% higher plasma eltrombopag AUC(0-τ) and moderate hepatic impairment resulted in approximately 141% to 240% higher plasma eltrombopag AUC(0-τ) values compared with patients with normal hepatic function. The half-life of eltrombopag was prolonged 3-fold in patients with mild hepatic impairment and 4-fold in patients with moderate hepatic impairment. This clinical trial did not evaluate protein-binding effects.
Chronic Hepatitis C
Patients with chronic hepatitis C treated with eltrombopag had higher plasma AUC(0-τ) values as compared with healthy subjects, and AUC(0-τ) increased with increasing Child-Pugh score. Patients with chronic hepatitis C and mild hepatic impairment had approximately 100% to 144% higher plasma AUC(0-τ) compared with healthy subjects. This clinical trial did not evaluate protein-binding effects.
Renal Impairment
Following a single dose of eltrombopag, the average total plasma eltrombopag AUC0-INF was
32% to 36% lower in subjects with mild (estimated creatinine clearance (CLCr) by Cockcroft-Gault equation: 50 to 80 mL/min), to moderate (CLCr of 30 to 49 mL/min) renal impairment and 60% lower in subjects with severe (CLCr less than 30 mL/min) renal impairment compared with healthy subjects. The effect of renal impairment on unbound (active) eltrombopag exposure has not been assessed.Pediatric Patients
The pharmacokinetics of eltrombopag have been evaluated in 168 pediatric patients 1 year and older with ITP dosed once daily in two trials. Plasma eltrombopag apparent clearance following oral administration (CL/F) increased with increasing body weight. East-/Southeast-Asian pediatric patients with ITP had approximately 43% higher plasma eltrombopag AUC(0-τ) values as compared with non-Asian patients.
Plasma eltrombopag AUC(0-τ) and Cmax in pediatric patients aged 12 to 17 years was similar to that observed in adults. The pharmacokinetic parameters of eltrombopag in pediatric patients with ITP are shown in Table 9.
Table 9. Geometric Mean (95% CI) Steady-state Plasma Eltrombopag Pharmacokinetic Parametersa in Patients with ITP (Normalized to a Once-daily 50-mg Dose, Equivalent to ALVAIZ 36 mg Once-daily) Age
Cmaxb
(mcg/mL)
AUC(0-τ)b
(mcg·hr/mL)
Adults (n = 108)
7.03
(6.44, 7.68)
101
(91.4, 113)
12 to 17 years (n = 62)
6.80
(6.17, 7.50)
103
(91.1, 116)
6 to 11 years (n = 68)
10.3
(9.42, 11.2)
153
(137, 170)
aPK parameters presented as geometric mean (95% CI).
bBased on population PK post-hoc estimates.
Drug Interaction Studies
Clinical Studies
Effect of Drugs on Eltrombopag
Effect of Polyvalent Cation-containing Antacids on Eltrombopag:
The coadministration of a single dose of eltrombopag with a polyvalent cation-containing antacid (1,524 mg aluminum hydroxide, 1,425 mg magnesium carbonate, and sodium alginate) decreased plasma eltrombopag AUC0-INF and Cmax by approximately 70%. The contribution of sodium alginate to this interaction is not known.
Effect of HIV Protease Inhibitors on Eltrombopag:
The coadministration of repeat-dose lopinavir 400 mg/ritonavir 100 mg (twice daily) with a single dose of eltrombopag decreased plasma eltrombopag AUC0-INF by 17%.
Effect of HCV Protease Inhibitors on Eltrombopag:
The coadministration of repeat-dose telaprevir (750 mg every 8 hours) or boceprevir (800 mg every 8 hours) with a single dose of eltrombopag to healthy adult subjects in a clinical trial did not alter plasma eltrombopag AUC0-INF or Cmax to a significant extent.
Effect of Cyclosporine on Eltrombopag:
The coadministration of a single dose of eltrombopag with a single dose of an OATP and BCRP inhibitor cyclosporine (200 mg or 600 mg) decreased plasma eltrombopag AUC0-INF by 18% to 24% and Cmax by 25% to 39%.
Effect of Pegylated Interferon alfa-2a + Ribavirin and Pegylated Interferon alfa-2b + Ribavirin on Eltrombopag:
The presence of pegylated interferon alfa + ribavirin therapy did not significantly affect the clearance of eltrombopag.
Effect of Eltrombopag on Other Drugs
Effect of Eltrombopag on Cytochrome P450 Enzymes Substrates:
The coadministration of multiple doses of eltrombopag (once daily for 7 days) did not result in the inhibition or induction of the metabolism of a combination of probe substrates for CYP1A2 (caffeine), CYP2C19 (omeprazole), CYP2C9 (flurbiprofen), or CYP3A4 (midazolam) in humans.
Effect of Eltrombopag on Rosuvastatin:
The coadministration of multiple doses of eltrombopag (once daily for 5 days) with a single dose of rosuvastatin (OATP1B1 and BCRP substrate; 10 mg) increased plasma rosuvastatin AUC0-INF by 55% and Cmax by 103%.
Effect of Eltrombopag on HCV Protease Inhibitors:
The coadministration of repeat-dose telaprevir (750 mg every 8 hours) or boceprevir (800 mg every 8 hours) with a single dose of eltrombopag to healthy adult subjects in a clinical trial did not alter plasma telaprevir or boceprevir AUC0-INF or Cmax to a significant extent.
In vitro Studies
Eltrombopag Effect on Metabolic Enzymes
Eltrombopag has demonstrated the potential to inhibit CYP2C8, CYP2C9, UGT1A1, UGT1A3, UGT1A4, UGT1A6, UGT1A9, UGT2B7, and UGT2B15.
Eltrombopag Effect on Transporters
Eltrombopag has demonstrated the potential to inhibit OATP1B1 and BCRP.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Eltrombopag does not stimulate platelet production in rats, mice, or dogs because of unique TPO receptor specificity. Data from these animals do not fully model effects in humans.
Eltrombopag was not carcinogenic in mice at doses up to 75 mg/kg/day or in rats at doses up to 40 mg/kg/day (exposures up to 4 times the human clinical exposure based on AUC in patients with ITP at 75 mg/day and 2 times the human clinical exposure based on AUC in patients with chronic hepatitis C at 100 mg/day).
Eltrombopag was not mutagenic or clastogenic in a bacterial mutation assay or in two in vivo assays in rats (micronucleus and unscheduled DNA synthesis, 10 times the human clinical exposure based on Cmax in patients with ITP at 75 mg/day and 7 times the human clinical exposure based on Cmax in patients with chronic hepatitis C at 100 mg/day). In the in vitro mouse lymphoma assay, eltrombopag was marginally positive (less than 3-fold increase in mutation frequency).
Eltrombopag did not affect female fertility in rats at doses up to 20 mg/kg/day (2 times the human clinical exposure based on AUC in patients with ITP at 75 mg/day and similar to the human clinical exposure based on AUC in patients with chronic hepatitis C at 100 mg/day). Eltrombopag did not affect male fertility in rats at doses up to 40 mg/kg/day, the highest dose tested (3 times the human clinical exposure based on AUC in patients with ITP at 75 mg/day and 2 times the human clinical exposure based on AUC in patients with chronic hepatitis C at 100 mg/day).
13.2 Animal Toxicology and/or Pharmacology
Treatment-related cataracts were detected in rodents in a dose- and time-dependent manner. At greater than or equal to 6 times the human clinical exposure based on AUC in patients with ITP at 75 mg/day and 3 times the human clinical exposure based on AUC in patients with chronic hepatitis C at 100 mg/day, cataracts were observed in mice after 6 weeks and in rats after 28 weeks of dosing. At greater than or equal to 4 times the human clinical exposure based on AUC in patients with ITP at 75 mg/day and 2 times the human clinical exposure based on AUC in patients with chronic hepatitis C at 100 mg/day, cataracts were observed in mice after 13 weeks and in rats after 39 weeks of dosing [see Warnings and Precautions (5.5)].
Renal tubular toxicity was observed in studies up to 14 days in duration in mice and rats at exposures that were generally associated with morbidity and mortality. Tubular toxicity was also observed in a 2-year oral carcinogenicity study in mice at doses of 25, 75, and 150 mg/kg/day. The exposure at the lowest dose was 1.2 times the human clinical exposure based on AUC in patients with ITP at 75 mg/day and 0.6 times the human clinical exposure based on AUC in patients with chronic hepatitis C at 100 mg/day. No similar effects were observed in mice after 13 weeks at exposures greater than those associated with renal changes in the 2-year study, suggesting that this effect is both dose- and time-dependent.
-
14 CLINICAL STUDIES
The effectiveness of ALVAIZ has been established based on adequate and well-controlled studies of eltrombopag olamine in adult and pediatric patients 6 years and older with persistent or chronic ITP, adult patients with chronic hepatitis C-associated thrombocytopenia, and adult patients with refractory severe aplastic anemia. Below is a display of the efficacy information for eltrombopag olamine in these adequate and well-controlled studies.
14.1 Persistent or Chronic ITP
Adults: The efficacy and safety of eltrombopag in adult patients with persistent or chronic ITP were evaluated in three randomized, double-blind, placebo-controlled trials and in an open-label extension trial.
In Study TRA100773B and Study TRA100773A (referred to as Study 773B and Study 773A, respectively [NCT00102739]), patients who had completed at least one prior ITP therapy and who had a platelet count less than 30 x 109/L were randomized to receive either eltrombopag or placebo daily for up to 6 weeks, followed by 6 weeks off therapy. During the trials, eltrombopag or placebo was discontinued if the platelet count exceeded 200 x 109/L.
The median age of the patients was 50 years and 60% were female. Approximately 70% of the patients had received at least 2 prior ITP therapies (predominantly corticosteroids, immunoglobulins, rituximab, cytotoxic therapies, danazol, and azathioprine) and 40% of the patients had undergone splenectomy. The median baseline platelet counts (approximately 18 x 109/L) were similar among all treatment groups.
Study 773B randomized 114 patients (2:1) to eltrombopag 50 mg or placebo. Of 60 patients with documented time since diagnosis, approximately 17% met the definition of persistent ITP with time since diagnosis of 3 to 12 months. Study 773A randomized 117 patients (1:1:1:1) among placebo or 1 of 3 dose regimens of eltrombopag , 30 mg, 50 mg, or 75 mg each administered daily. Of 51 patients with documented time since diagnosis, approximately 14% met the definition of persistent ITP.
The efficacy of eltrombopag in this trial was evaluated by response rate, defined as a shift from a baseline platelet count of less than 30 x 109/L to greater than or equal to 50 x 109/L at any time during the treatment period (Table 10).
Table 10. Studies 773B and 773A: Platelet Count Response (≥ 50 x 109/L) Rates in Adults with Persistent or Chronic Immune Thrombocytopenia Study
Eltrombopag
50 mg DailyPlacebo
773B
43/73 (59%)a
6/37 (16%)
773A
19/27 (70%)a
3/27 (11%)
ap-value < 0.001 for eltrombopag versus placebo.
The platelet count response to eltrombopag was similar among patients who had or had not undergone splenectomy. In general, increases in platelet counts were detected 1 week following initiation of eltrombopag and the maximum response was observed after 2 weeks of therapy. In the placebo and 50-mg dose groups of eltrombopag, the trial drug was discontinued due to an increase in platelet counts to greater than 200 x 109/L in 3% and 27% of the patients, respectively. The median duration of treatment with the 50-mg dose of eltrombopag was 43 days in Study 773B and 42 days in Study 773A.
Of 7 patients who underwent hemostatic challenges, additional ITP medications were required in 3 of 3 placebo group patients and 0 of 4 patients treated with eltrombopag. Surgical procedures accounted for most of the hemostatic challenges. Hemorrhage requiring transfusion occurred in one placebo group patient and no patients treated with eltrombopag.
In the RAISE study (NCT00370331), 197 patients were randomized (2:1) to receive either eltrombopag 50 mg once daily (n = 135) or placebo (n = 62) for 6 months, during which time the dose of eltrombopag could be adjusted based on individual platelet counts. Of 145 patients with documented time since diagnosis, 19% met the definition of persistent ITP. Patients were allowed to taper or discontinue concomitant ITP medications after being treated with eltrombopag for 6 weeks. Patients were permitted to receive rescue treatments at any time during the trial as clinically indicated.
The median ages of the patients treated with eltrombopag and placebo were 47 years and 52.5 years, respectively. Approximately half of the patients treated with eltrombopag and placebo (47% and 50%, respectively) were receiving concomitant ITP medication (predominantly corticosteroids) at randomization and had baseline platelet counts less than or equal to 15 x 109/L (50% and 48%, respectively). A similar percentage of patients treated with eltrombopag and placebo (37% and 34%, respectively) had a prior splenectomy.
The efficacy of eltrombopag in this trial was evaluated by the odds of achieving a platelet count greater than or equal to 50 x 109/L and less than or equal to 400 x 109/L for patients receiving eltrombopag relative to placebo and was based on patient response profiles throughout the 6-month treatment period. In 134 patients who completed 26 weeks of treatment, a sustained platelet response (platelet count greater than or equal to 50 x 109/L and less than or equal to 400 x 109/L for 6 out of the last 8 weeks of the 26-week treatment period in the absence of rescue medication at any time) was achieved by 60% of patients treated with eltrombopag, compared with 10% of patients treated with placebo (splenectomized patients: eltrombopag 51%, placebo 8%; non-splenectomized patients: eltrombopag 66%, placebo 11%). The proportion of responders in the group of patients treated with eltrombopag was between 37% and 56% compared with 7% and 19% in the placebo treatment group for all on-therapy visits. Patients treated with eltrombopag were significantly more likely to achieve a platelet count between 50 x 109/L and 400 x 109/L during the entire 6-month treatment period compared with those patients treated with placebo.
Outcomes of treatment are presented in Table 11 for all patients enrolled in the trial.
Table 11. RAISE: Outcomes of Treatment in Adults with Persistent or Chronic Immune Thrombocytopenia Outcome
Eltrombopag
n = 135
Placebon = 62
Mean number of weeks with platelet counts ≥ 50 x 109/L
11.3
2.4
Requiring rescue therapy, n (%)
24 (18)
25 (40)
Among 94 patients receiving other ITP therapy at baseline, 37 (59%) of 63 patients treated with eltrombopag and 10 (32%) of 31 patients in the placebo group discontinued concomitant therapy at some time during the trial.
In the EXTEND study (NCT00351468), patients who completed any prior clinical trial with eltrombopag were enrolled in an open-label, single-arm trial in which attempts were made to decrease the dose or eliminate the need for any concomitant ITP medications. Eltrombopag was administered to 302 patients in EXTEND; 218 patients completed 1 year, 180 patients completed 2 years, 107 patients completed 3 years, 75 patients completed 4 years, 34 patients completed 5 years, and 18 patients completed 6 years of therapy. The median baseline platelet count was 19 x 109/L prior to administration of eltrombopag. Median platelet counts at 1, 2, 3, 4, 5, 6, and 7 years on study were 85 x 109/L, 85 x 109/L, 105 x 109/L, 64 x 109/L, 75 x 109/L, 119 x 109/L, and 76 x 109/L, respectively.
Pediatric Patients: The efficacy and safety of eltrombopag in pediatric patients 1 year and older with persistent or chronic ITP were evaluated in two double-blind, placebo-controlled trials. ALVAIZ is not indicated for pediatric patients less than 6 years of age with persistent or chronic ITP. The trials differed in time since ITP diagnosis: at least 6 months versus at least 12 months. During the trials, doses could be increased every 2 weeks to a maximum of 75 mg once daily. The dose of eltrombopag was reduced if the platelet count exceeded 200 x 109/L and interrupted and reduced if it exceeded 400 x 109/L.
In the PETIT2 study (NCT01520909), patients refractory or relapsed to at least one prior ITP therapy with a platelet count less than 30 x 109/L (n = 92) were stratified by age and randomized (2:1) to eltrombopag (n = 63) or placebo (n = 29). The starting dose for patients aged 6 to 17 years was 50 mg once daily for those at least 27 kg and 37.5 mg once daily for those less than 27 kg, administered as oral tablets. A reduced dose of 25 mg once daily was used for East-/Southeast-Asian patients aged 6 to 17 years regardless of weight.
The 13-week, randomized, double-blind period was followed by a 24-week, open-label period where patients from both arms were eligible to receive eltrombopag.
The median age of the patients was 9 years and 48% were female. Approximately 62% of patients had a baseline platelet count less than or equal to 15 x 109/L, a characteristic that was similar between treatment arms. The percentage of patients with at least 2 prior ITP therapies (predominantly corticosteroids and immunoglobulins) was 73% in the group treated with eltrombopag and 90% in the group treated with placebo. Four patients in the group treated with eltrombopag had undergone splenectomy.
The efficacy of eltrombopag in this trial was evaluated by the proportion of subjects on eltrombopag achieving platelet counts ≥ 50 x 109/L (in the absence of rescue therapy) for at least 6 out of 8 weeks between Weeks 5 to 12 of the randomized, double-blind period (Table 12).
Table 12. PETIT2: Platelet Count Response (≥ 50 x 109/L Without Rescue) for 6 out of 8 Weeks (between Weeks 5 to 12) Overall and by Age Cohort in Pediatric Patients 1 Year and Older with Chronic Immune Thrombocytopenia Age Cohort
Eltrombopag
Placebo
Overall
12 to 17 years
6 to 11 years
1 to 5 years
26/63 (41%)a
10/24 (42%)
11/25 (44%)
5/14 (36%)
1/29 (3%)
1/10 (10%)
0/13 (0%)
0/6 (0%)
ap-value = < 0.001 for eltrombopag versus placebo.
More pediatric patients treated with eltrombopag (75%) compared with placebo (21%) had at least one platelet count greater than or equal to 50 x 109/L during the first 12 weeks of randomized treatment in absence of rescue therapy. Fewer pediatric patients treated with eltrombopag required rescue treatment during the randomized, double-blind period compared with placebo-treated patients (19% [12/63] versus 24% [7/29]). In the patients who achieved a platelet response (≥ 50 x 109/L without rescue) for 6 out of 8 weeks (between weeks 5 to 12), 62% (16/26) had an initial response in the first 2 weeks after starting eltrombopag.
Patients were permitted to reduce or discontinue baseline ITP therapy only during the open-label phase of the trial. Among 15 patients receiving other ITP therapy at baseline, 53% (8/15) reduced (n = 1) or discontinued (n = 7) concomitant therapy, mainly corticosteroids, without needing rescue therapy.
In the PETIT study (NCT00908037), patients refractory or relapsed to at least one prior ITP therapy with a platelet count less than 30 x 109/L (n = 67) were stratified by age and randomized (2:1) to eltrombopag (n = 45) or placebo (n = 22). Approximately 15% of patients met the definition of persistent ITP. The starting dose for patients aged 12 to 17 years was 37.5 mg once daily regardless of weight or race. The starting dose for patients aged 6 to 11 years was 50 mg once daily for those greater than or equal to 27 kg and 25 mg once daily for those less than 27 kg, administered as oral tablets. Reduced doses of 25 mg (for those greater than or equal to 27 kg) and 12.5 mg (for those less than 27 kg), each once daily, were used for East-/Southeast-Asian patients in this age range.
The 7-week, randomized, double-blind period was followed by an open-label period of up to 24 weeks where patients from both arms were eligible to receive eltrombopag.
The median age of the patients was 10 years and 60% were female. Approximately 51% of patients had a baseline platelet count less than or equal to 15 x 109/L. The percentage of patients with at least 2 prior ITP therapies (predominantly corticosteroids and immunoglobulins) was 84% in the group treated with eltrombopag and 86% in the group treated with placebo. Five patients in the group treated with eltrombopag had undergone splenectomy.
The efficacy of eltrombopag in this trial was evaluated by the proportion of patients achieving platelet counts greater than or equal to 50 x 109/L (in absence of rescue therapy) at least once between Weeks 1 and 6 of the randomized, double-blind period (Table 13). Platelet response to eltrombopag was consistent across the age cohorts.
Table 13. PETIT: Platelet Count Response (≥ 50 x 109/L Without Rescue) Rates in Pediatric Patients 1 Year and Older with Persistent or Chronic Immune Thrombocytopenia Age Cohort
Eltrombopag
Placebo
Overall
28/45 (62%)a
7/22 (32%)
12 to 17 years
10/16 (62%)
0/8 (0%)
6 to 11 years
12/19 (63%)
3/9 (33%)
a p-value = 0.011 for eltrombopag versus placebo.
Fewer pediatric patients treated with eltrombopag required rescue treatment during the randomized, double-blind period compared with placebo-treated patients (13% [6/45] versus 50% [11/22]).
Patients were permitted to reduce or discontinue baseline ITP therapy only during the open-label phase of the trial. Among 13 patients receiving other ITP therapy at baseline, 46% (6/13) reduced (n = 3) or discontinued (n = 3) concomitant therapy, mainly corticosteroids, without needing rescue therapy.
14.2 Chronic Hepatitis C-associated Thrombocytopenia
The efficacy and safety of eltrombopag for the treatment of thrombocytopenia in adult patients with chronic hepatitis C were evaluated in two randomized, double-blind, placebo-controlled trials. The ENABLE1 study (NCT00516321) utilized peginterferon alfa-2a (PEGASYS®) plus ribavirin for antiviral treatment and the ENABLE2 study (NCT00529568) utilized peginterferon alfa-2b (PEGINTRON®) plus ribavirin. In both trials, patients with a platelet count of less than 75 x 109/L were enrolled and stratified by platelet count, screening HCV RNA, and HCV genotype. Patients were excluded if they had evidence of decompensated liver disease with Child-Pugh score greater than 6 (Class B and C), history of ascites, or hepatic encephalopathy. The median age of the patients in both trials was 52 years, 63% were male, and 74% were Caucasian. Sixty-nine percent of patients had HCV genotypes 1, 4, 6, with the remainder genotypes 2 and 3. Approximately 30% of patients had been previously treated with interferon and ribavirin. The majority of patients (90%) had bridging fibrosis and cirrhosis, as indicated by noninvasive testing. A similar proportion (95%) of patients in both treatment groups had Child-Pugh Class A (score 5 to 6) at baseline. A similar proportion of patients (2%) in both treatment groups had baseline international normalized ratio (INR) greater than 1.7. Median baseline platelet counts (approximately 60 x 109/L) were similar in both treatment groups. The trials consisted of 2 phases – a pre-antiviral treatment phase and an antiviral treatment phase. In the pre-antiviral treatment phase, patients received open-label eltrombopag to increase the platelet count to a threshold of greater than or equal to 90 x 109/L for ENABLE1 and greater than or equal to 100 x 109/L for ENABLE2. Eltrombopag was administered at an initial dose of 25 mg once daily for 2 weeks and increased in 25-mg increments over 2- to 3-week periods to achieve the optimal platelet count to initiate antiviral therapy. The maximal time patients could receive open-label eltrombopag was 9 weeks. If threshold platelet counts were achieved, patients were randomized (2:1) to the same dose of eltrombopag at the end of the pre-treatment phase or to placebo. Eltrombopag was administered in combination with pegylated interferon and ribavirin per their respective prescribing information for up to 48 weeks.
The efficacy of eltrombopag for both trials was evaluated by sustained virologic response (SVR) defined as the percentage of patients with undetectable HCV-RNA at 24 weeks after completion of antiviral treatment. The median time to achieve the target platelet count greater than or equal to 90 x 109/L was approximately 2 weeks. Ninety-five percent of patients were able to initiate antiviral therapy.
In both trials, a significantly greater proportion of patients treated with eltrombopag achieved SVR (see Table 14). The improvement in the proportion of patients who achieved SVR was consistent across subgroups based on baseline platelet count (less than 50 x 109/L versus greater than or equal to 50 x 109/L). In patients with high baseline viral loads (greater than or equal to 800,000), the SVR rate was 18% (82/452) for eltrombopag versus 8% (20/239) for placebo.
Table 14. ENABLE1 and ENABLE2: Sustained Virologic Response (SVR) in Adults With Chronic Hepatitis C Pre-antiviral Treatment Phase
ENABLE1a
ENABLE2b
n = 715
n = 805
% Patients who achieved target platelet counts and initiated antiviral therapyc
95%
94%
Antiviral Treatment Phase
Eltrombopag
n = 450
%
Placebo
n = 232%
Eltrombopag
n = 506%
Placebo
n = 253%
Overall SVRd
HCV Genotype 2,3
HCV Genotype 1,4,6
23
35
18
14
24
10
19
34
13
13
25
7
aEltrombopag given in combination with peginterferon alfa-2a (180 mcg once weekly for 48 weeks for genotypes 1/4/6; 24 weeks for genotype 2 or 3) plus ribavirin (800 to 1,200 mg daily in 2 divided doses orally).
bEltrombopag given in combination with peginterferon alfa-2b (1.5 mcg/kg once weekly for 48 weeks for genotypes 1/4/6; 24 weeks for genotype 2 or 3) plus ribavirin (800 to 1,400 mg daily in 2 divided doses orally).
cTarget platelet count was ≥ 90 x 109/L for ENABLE1 and ≥ 100 x 109/L for ENABLE2.
dp-value < 0.05 for eltrombopag versus placebo.
The majority of patients treated with eltrombopag (76%) maintained a platelet count greater than or equal to 50 x 109/L compared with 19% for placebo. A greater proportion of patients on eltrombopag did not require any antiviral dose reduction as compared with placebo (45% versus 27%).
14.3 Severe Aplastic Anemia
Refractory Severe Aplastic Anemia
Eltrombopag was studied in a single-arm, single-center, open-label trial (Study ETB115AUS28T, referred to as Study US28T [NCT00922883]) in 43 patients with severe aplastic anemia who had an insufficient response to at least one prior immunosuppressive therapy and who had a platelet count less than or equal to 30 x 109/L. Eltrombopag was administered at an initial dose of 50 mg once daily for 2 weeks and increased over 2-week periods up to a maximum dose of 150 mg once daily. The efficacy of eltrombopag in the study was evaluated by the hematologic response assessed after 12 weeks of treatment. Hematologic response was defined as meeting 1 or more of the following criteria: 1) platelet count increases to 20 x 109/L above baseline, or stable platelet counts with transfusion independence for a minimum of 8 weeks; 2) hemoglobin increase by greater than 1.5 g/dL, or a reduction in greater than or equal to 4 units of red blood cell (RBC) transfusions for 8 consecutive weeks; 3) ANC increase of 100% or an ANC increase greater than 0.5 x 109/L. Eltrombopag was discontinued after 16 weeks if no hematologic response was observed. Patients who responded continued therapy in an extension phase of the trial.
The treated population had median age of 45 years (range, 17 to 77 years) and 56% were male. At baseline, the median platelet count was 20 x 109/L, hemoglobin was 8.4 g/dL, ANC was 0.58 x 109/L, and absolute reticulocyte count was 24.3 x 109/L. Eighty-six percent of patients were red blood cell (RBC) transfusion dependent and 91% were platelet transfusion dependent. The majority of patients (84%) received at least 2 prior immunosuppressive therapies. Three patients had cytogenetic abnormalities at baseline.
Table 15 presents the efficacy results.
Table 15. Study US28T: Hematologic Response in Patients with Refractory Severe Aplastic Anemia Outcome
Eltrombopag
n = 43
Response ratea, n (%)
95% CI (%)
17 (40)
(25, 56)
Median of duration of response in months (95% CI)
NRb (3.0, NRb)
aIncludes single- and multi-lineage.
bNR = not reached due to few events (relapsed).
In the 17 responders, the platelet transfusion-free period ranged from 8 to 1096 days with a median of 200 days, and the RBC transfusion-free period ranged from 15 to 1082 days with a median of 208 days.
In the extension phase, 8 patients achieved a multi-lineage response; 4 of these patients subsequently tapered off treatment with eltrombopag and maintained the response (median follow up: 8.1 months, range, 7.2 to 10.6 months).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
ALVAIZ (eltrombopag tablets) is for oral administration and are available as follows.
9 mg - Blue, round, biconvex, film-coated tablets debossed with “TV” on one side and “Z9” on the other side. The tablets are available in bottles of 30 (NDC 0480-3273-56).
18 mg - Off-white, round, biconvex, film-coated tablets debossed with “TV” on one side and “Z18” on the other side. The tablets are available in bottles of 30 (NDC 0480-3274-56).
36 mg - Red, round, biconvex, film-coated tablets debossed with “TV” on one side and “Z36” on the other side. The tablets are available in bottles of 30 (NDC 0480-3275-56).
54 mg - Orange, round, biconvex, film-coated tablets debossed with “TV” on one side and “Z54” on the other side. The tablets are available in bottles of 30 (NDC 0480-3276-56).
-
17 PATIENT COUNSELING INFORMATION
Advise the patient or caregiver to read the FDA-approved patient labeling (Medication Guide).
Prior to treatment, patients should fully understand and be informed of the following risks and considerations for ALVAIZ:
Risks
Hepatotoxicity
- Therapy with ALVAIZ may be associated with hepatobiliary laboratory abnormalities [see Warnings and Precautions (5.2)].
- Advise patients with chronic hepatitis C and cirrhosis that they may be at risk for hepatic decompensation when receiving ALVAIZ with alfa interferon therapy [see Warnings and Precautions (5.1)].
- Advise patients that they should report any of the following signs and symptoms of liver problems to their healthcare provider right away [see Warnings and Precautions (5.2)].
- yellowing of the skin or the whites of the eyes (jaundice)
- unusual darkening of the urine
- unusual tiredness
- right upper stomach area pain
- confusion
- swelling of the stomach area (abdomen)
Risk of Bleeding Upon ALVAIZ Discontinuation
- Advise patients that thrombocytopenia and risk of bleeding may reoccur upon discontinuing ALVAIZ, particularly if ALVAIZ is discontinued while the patient is on anticoagulants or antiplatelet agents. Advise patients that during therapy with ALVAIZ, they should continue to avoid situations or medications that may increase the risk for bleeding.
Thrombotic/Thromboembolic Complications
- Advise patients that too much ALVAIZ may result in excessive platelet counts and a risk for thrombotic/thromboembolic complications [see Warnings and Precautions (5.4)].
Cataracts
- Advise patients to have a baseline ocular examination prior to administration of ALVAIZ and be monitored for signs and symptoms of cataracts during therapy [see Warnings and Precautions (5.5)].
Drug Interactions
- Advise patients to take ALVAIZ at least 2 hours before or 4 hours after calcium-rich foods, mineral supplements, and antacids which contain polyvalent cations, such as iron, calcium, aluminum, magnesium, selenium, and zinc [see Dosage and Administration (2.4), Drug Interactions (7.1)].
Lactation
- Advise women not to breastfeed during treatment with ALVAIZ [see Use in Specific Populations (8.2)].
Administration of ALVAIZ
- For patients with persistent or chronic ITP, therapy with ALVAIZ is administered to achieve and maintain a platelet count greater than or equal to 50 x 109/L as necessary to reduce the risk for bleeding [see Indications and Usage (1.1)].
- For patients with chronic hepatitis C, therapy with ALVAIZ is administered to achieve and maintain a platelet count necessary to initiate and maintain antiviral therapy with pegylated interferon and ribavirin [see Indications and Usage (1.2)].
- Advise patients to take ALVAIZ without a meal or with a meal low in calcium (≤ 50 mg) and at least 2 hours before or 4 hours after other medications (e.g., antacids) and calcium-rich foods [see Dosage and Administration (2.4)].
Brands listed are the trademarks of their respective owners.
Dispense with Medication Guide available at: www.tevausa.com/medguides
Manufactured For:
Teva Pharmaceuticals
Parsippany, NJ 07054Rev. A 5/2024
-
MEDICATION GUIDE
Dispense with Medication Guide available at: www.tevausa.com/medguides MEDICATION GUIDE
ALVAIZ™ (al-VAYZ)
(eltrombopag) tablets
What is the most important information I should know about ALVAIZ?
ALVAIZ may cause serious side effects, including:
Liver problems:
- If you have chronic hepatitis C virus and take ALVAIZ with interferon and ribavirin treatment, ALVAIZ may increase your risk of liver problems. If your healthcare provider tells you to stop your treatment with interferon and ribavirin, you will also need to stop taking ALVAIZ.
- ALVAIZ may increase your risk of liver problems that may be severe and possibly life threatening. Your healthcare provider will do blood tests to check your liver function before you start taking ALVAIZ and during your treatment. Your healthcare provider may stop your treatment with ALVAIZ if you have changes in your liver function blood tests.
Tell your healthcare provider right away if you have any of these signs and symptoms of liver problems:
- yellowing of the skin or the whites of the eyes (jaundice)
- unusual darkening of the urine
- unusual tiredness
- right upper stomach area (abdomen) pain
- confusion
- swelling of the stomach area (abdomen)
See “What are the possible side effects of ALVAIZ?” for other side effects of ALVAIZ.
What is ALVAIZ?
ALVAIZ is a prescription medicine used to treat:
- adults and children 6 years of age and older with low blood platelet counts due to persistent or chronic immune thrombocytopenia (ITP), when other medicines to treat ITP or surgery to remove the spleen have not worked well enough.
- adults with
- low blood platelet counts due to chronic hepatitis C virus (HCV) infection before and during treatment with interferon.
- severe aplastic anemia (SAA) when other medicines to treat SAA have not worked well enough.
ALVAIZ is used to try to raise platelet counts in order to lower your risk for bleeding.
ALVAIZ is not used to make platelet counts normal.
ALVAIZ is not for use in people with a pre-cancerous condition called myelodysplastic syndrome (MDS), or in people with low platelet counts caused by certain other medical conditions or diseases.
It is not known if ALVAIZ is safe and effective when used with other antiviral medicines to treat chronic hepatitis C.
It is not known if ALVAIZ is safe and effective in children:
- younger than 6 years with persistent or chronic ITP
- with low blood platelet counts due to chronic hepatitis C
- whose severe aplastic anemia (SAA) has not improved after previous treatments
Before you take ALVAIZ, tell your healthcare provider about all of your medical conditions, including if you:
- have liver problems
- have a precancerous condition called MDS or a blood cancer
- have or had a blood clot
- have a history of cataracts
- have had surgery to remove your spleen (splenectomy)
- have bleeding problems
- are of East-Asian or Southeast-Asian ancestry. You may need a lower dose of ALVAIZ.
- are pregnant or plan to become pregnant. It is not known if ALVAIZ will harm an unborn baby. Tell your healthcare provider if you become pregnant or think you may be pregnant during treatment with ALVAIZ.
- Females who are able to become pregnant, should use effective birth control (contraception) during treatment with ALVAIZ and for at least 7 days after stopping treatment with ALVAIZ. Talk to your healthcare provider about birth control methods that may be right for you during this time.
- are breastfeeding or plan to breastfeed. You should not breastfeed during your treatment with ALVAIZ. Talk to your healthcare provider about the best way to feed your baby during this time.
- Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. ALVAIZ may affect the way certain medicines work. Certain other medicines may affect the way ALVAIZ works.
Especially tell your healthcare provider if you take:
- certain medicines used to treat high cholesterol, called “statins”
- a blood thinner medicine
Certain medicines may keep ALVAIZ from working correctly. Take ALVAIZ at least 2 hours before or 4 hours after taking these products:
- antacid medicine used to treat stomach ulcers or heartburn
- multivitamins or products that contain iron, calcium, aluminum, magnesium, selenium, and zinc which may be found in mineral supplements.
Ask your healthcare provider if you are not sure if your medicine is one that is listed above.
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
How should I take ALVAIZ?
- Take ALVAIZ exactly as your healthcare provider tells you to take it. Your healthcare provider will prescribe the dose of ALVAIZ tablets.
- Swallow ALVAIZ tablets whole. Do not split, chew, or crush ALVAIZ tablets and do not mix with food or liquids. Tell your healthcare provider if your or your child are not able to swallow ALVAIZ tablets whole. You will need to take a different medicine.
- Do not stop taking ALVAIZ without talking with your healthcare provider first. Do not change your dose or schedule for taking ALVAIZ unless your healthcare provider tells you to change it.
- The dose of ALVAIZ is not the same as other eltrombopag products. Do not change between ALVAIZ and other eltrombopag products unless your healthcare provider tells you.
- Take ALVAIZ without a meal or with a meal low in calcium (50 mg or less) and at least 2 hours before or 4 hours after eating calcium-rich foods, such as dairy products, calcium-fortified juices, and certain fruits and vegetables.
- Do not take more than 1 dose of ALVAIZ in 1 day.
- If you take too much ALVAIZ, you may have a higher risk of serious side effects. Call your healthcare provider right away.
- Your healthcare provider will check your platelet count during your treatment with ALVAIZ and change your dose of ALVAIZ as needed.
- Tell your healthcare provider about any bruising or bleeding that happens during treatment and after you stop taking ALVAIZ.
- If you have SAA, your healthcare provider may do tests to monitor your bone marrow during treatment with ALVAIZ.
What should I avoid while taking ALVAIZ?
Avoid situations and medicines that may increase your risk of bleeding during treatment with ALVAIZ.
What are the possible side effects of ALVAIZ?
ALVAIZ may cause serious side effects, including:
- See “What is the most important information I should know about ALVAIZ?”
- Increased risk of worsening of a precancerous blood condition called myelodysplastic syndrome (MDS) to acute myelogenous leukemia (AML). ALVAIZ is not for use in people with a precancerous condition called myelodysplastic syndromes (MDS). See “What is ALVAIZ?” If you have MDS and receive ALVAIZ, you have an increased risk that your MDS condition may worsen and become a blood cancer called AML. If your MDS worsens to become AML, you may have an increased risk of death from AML.
-
High platelet counts and higher risk for blood clots. Your risk of getting a blood clot is increased if your platelet count is too high during treatment with ALVAIZ. Your risk of getting a blood clot may also be increased during treatment with ALVAIZ if you have normal or low platelet counts. You may have severe problems or die from some forms of blood clots, such as clots that travel to the lungs or that cause heart attacks or strokes. Your healthcare provider will check your blood platelet counts, and change your dose or stop ALVAIZ if your platelet counts get too high. Tell your healthcare provider right away if you have signs and symptoms of a blood clot in the leg, such as swelling, pain, or tenderness in your leg.
People with chronic liver disease may be at risk for a type of blood clot in the stomach area (abdomen). Tell your healthcare provider right away if you have stomach-area (abdomen) pain, nausea, vomiting, or diarrhea as these may be symptoms of this type of blood clot.
- New or worsened cataracts (a clouding of the lens in the eye). New or worsened cataracts can happen in people taking ALVAIZ. Your healthcare provider will check your eyes before and during your treatment with ALVAIZ. Tell your healthcare provider about any changes in your eyesight during treatment with ALVAIZ.
The most common side effects of ALVAIZ in adults and children include:
- low red blood cell count (anemia)
- nausea
- fever
- abnormal liver function tests
- cough
- tiredness
- headache
- diarrhea
Laboratory tests may show abnormal changes to the cells in your bone marrow.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of ALVAIZ. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store ALVAIZ tablets?
- Store ALVAIZ tablets at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep ALVAIZ in the bottle given to you.
Keep ALVAIZ and all medicines out of the reach of children.
General information about the safe and effective use of ALVAIZ.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ALVAIZ for a condition for which it was not prescribed. Do not give ALVAIZ to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your pharmacist or healthcare provider for information about ALVAIZ that is written for health professionals.
What are the ingredients in ALVAIZ?
Active ingredient: eltrombopag choline
Inactive ingredients:
Tablet Core: anhydrous lactose, copovidone, croscarmellose sodium, edetate disodium dihydrate, magnesium stearate, poloxamer 188, polyethylene glycol 4000, povidone K12, silicified microcrystalline cellulose, and silicon dioxide
Coating:
- 9 mg tablets: FD&C Blue No. 1 aluminum lake, polyethylene glycol 3350, polyvinyl alcohol, talc, and titanium dioxide.
- 18 mg tablets: Hypromellose 2910, polyethylene glycol 4000, talc, and titanium dioxide.
- 36 mg tablets: Iron oxide red, iron oxide yellow, polyethylene glycol 3350, polyvinyl alcohol, talc, and titanium dioxide.
- 54 mg tablets: FD&C Red #40 aluminum lake, hypromellose 2910, iron oxide yellow, polyethylene glycol 400, and titanium dioxide.
Manufactured For: Teva Pharmaceuticals, Parsippany, NJ 07054
For more information about call Teva at 1-888-838-2872.
This Medication Guide has been approved by the U.S. Food and Drug Administration. Rev. A 5/2024
- PACKAGE.LABEL.PRINCIPAL DISPLAY PANEL
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALVAIZ
eltrombopag tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0480-3273 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ELTROMBOPAG CHOLINE (UNII: F9G8XE24IB) (ELTROMBOPAG - UNII:S56D65XJ9G) ELTROMBOPAG 9 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) COPOVIDONE K25-31 (UNII: D9C330MD8B) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) EDETATE DISODIUM (UNII: 7FLD91C86K) MAGNESIUM STEARATE (UNII: 70097M6I30) POLOXAMER 188 (UNII: LQA7B6G8JG) POLYETHYLENE GLYCOL 4000 (UNII: 4R4HFI6D95) POVIDONE K12 (UNII: 333AG72FWJ) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) FD&C BLUE NO. 1 ALUMINUM LAKE (UNII: J9EQA3S2JM) POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color blue Score no score Shape ROUND Size 6mm Flavor Imprint Code TV;Z9 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0480-3273-56 30 in 1 BOTTLE; Type 0: Not a Combination Product 02/13/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA216774 02/13/2024 ALVAIZ
eltrombopag tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0480-3274 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ELTROMBOPAG CHOLINE (UNII: F9G8XE24IB) (ELTROMBOPAG - UNII:S56D65XJ9G) ELTROMBOPAG 18 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) COPOVIDONE K25-31 (UNII: D9C330MD8B) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) EDETATE DISODIUM (UNII: 7FLD91C86K) MAGNESIUM STEARATE (UNII: 70097M6I30) POLOXAMER 188 (UNII: LQA7B6G8JG) POLYETHYLENE GLYCOL 4000 (UNII: 4R4HFI6D95) POVIDONE K12 (UNII: 333AG72FWJ) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) HYPROMELLOSE 2910 (3 MPA.S) (UNII: 0VUT3PMY82) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white (off-white) Score no score Shape ROUND Size 8mm Flavor Imprint Code TV;Z18 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0480-3274-56 30 in 1 BOTTLE; Type 0: Not a Combination Product 02/13/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA216774 02/13/2024 ALVAIZ
eltrombopag tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0480-3275 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ELTROMBOPAG CHOLINE (UNII: F9G8XE24IB) (ELTROMBOPAG - UNII:S56D65XJ9G) ELTROMBOPAG 36 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) COPOVIDONE K25-31 (UNII: D9C330MD8B) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) EDETATE DISODIUM (UNII: 7FLD91C86K) MAGNESIUM STEARATE (UNII: 70097M6I30) POLOXAMER 188 (UNII: LQA7B6G8JG) POLYETHYLENE GLYCOL 4000 (UNII: 4R4HFI6D95) POVIDONE K12 (UNII: 333AG72FWJ) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color red Score no score Shape ROUND Size 10mm Flavor Imprint Code TV;Z36 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0480-3275-56 30 in 1 BOTTLE; Type 0: Not a Combination Product 02/13/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA216774 02/13/2024 ALVAIZ
eltrombopag tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0480-3276 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ELTROMBOPAG CHOLINE (UNII: F9G8XE24IB) (ELTROMBOPAG - UNII:S56D65XJ9G) ELTROMBOPAG 54 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) COPOVIDONE K25-31 (UNII: D9C330MD8B) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) EDETATE DISODIUM (UNII: 7FLD91C86K) MAGNESIUM STEARATE (UNII: 70097M6I30) POLOXAMER 188 (UNII: LQA7B6G8JG) POLYETHYLENE GLYCOL 4000 (UNII: 4R4HFI6D95) POVIDONE K12 (UNII: 333AG72FWJ) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) FD&C RED NO. 40 (UNII: WZB9127XOA) HYPROMELLOSE 2910 (5 MPA.S) (UNII: R75537T0T4) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color orange Score no score Shape ROUND Size 12mm Flavor Imprint Code TV;Z54 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0480-3276-56 30 in 1 BOTTLE; Type 0: Not a Combination Product 02/13/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA216774 02/13/2024 Labeler - Teva Pharmaceuticals, Inc. (022629579)








