Label: EMINENCE BRIGHT SKIN MOISTURIZER BROAD SPECTRUM SPF 30 SUNSCREEN- octinoxate, zinc oxide cream
- NDC Code(s): 15751-2272-2, 15751-2272-5, 15751-2272-8
- Packager: Eminence Organic Skin Care
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 29, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
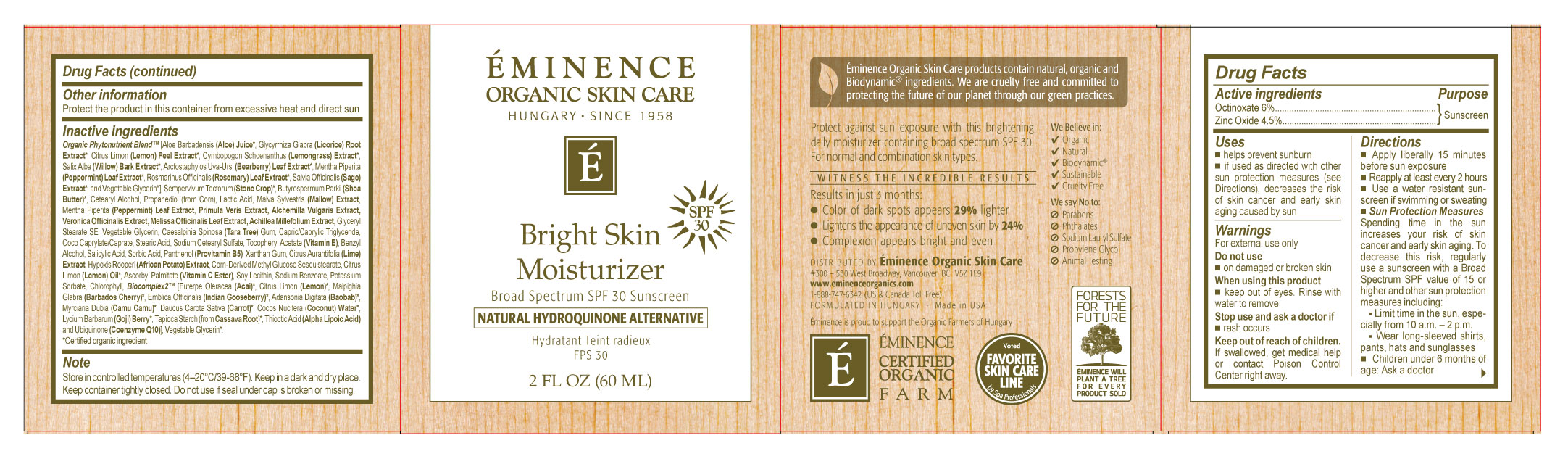
- EMINENCE Bright Skin Moisturizer BROAD SPECTRUM SPF 30 SUNSCREEN
- Active Ingredients
- Purpose
- Uses
- Warnings
-
Directions
- Apply liberally 15 minutes before sun exposure
- Reapply at least every 2 hours
- Use a water resistant sunscreen if swimming or sweating
- Limit time in the sun, especially from 10 a.m. - 2 p.m/
- Wear long sleeved shirts, pants, hats and sunglasses
- Children under 6 months of age: Ask a doctor
Sun Protection Measures
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- Other Information
-
Inactive Ingredients
Stone Crop, Stone Crop Juice, Water, Glycerin, Mallow Extract, Pep per mint Leaf Extract, Primrose Extract, Lady’s Mantle Extract, Common Speedwell Ex tract, Balm Mint Extract, Yarrow Ex tract, Tara Tree Gum, African Po tato Extract, Licorice Root, Bearberry Ex tract, Bioflavonoids Cetearyl Alcohol, Stearic Acid, Sodium Cetearyl Sulfate, Methyl Glucose Sesquistearate, Coco Cap rylate, Glyceryl Palmitate, Corn Germ Oil, Hydrolyzed Wheat Protein, Sor bi tol, Diglycerin, Lactic Acid, Lysine, Vitamin A, Vitamin C Ester, Vitamin E, Coenzyme Q10, Alpha Lipoic Acid, Xanthan Gum, Lemon Oil, Lavender Oil, Lemongrass, Soybean Oil.
- Distributed By
- Note:
- INACTIVE INGREDIENT
- Bright Skin Moisturizer 8.4 oz
- Bright Skin Moisturizer 2 oz
- Bright Skin Moisturizer
-
INGREDIENTS AND APPEARANCE
EMINENCE BRIGHT SKIN MOISTURIZER BROAD SPECTRUM SPF 30 SUNSCREEN
octinoxate, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:15751-2272 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 6 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 4.5 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) MALVA SYLVESTRIS LEAF (UNII: 17H39B00T5) MENTHA PIPERITA LEAF (UNII: A389O33LX6) ALCHEMILLA XANTHOCHLORA FLOWERING TOP (UNII: 9M289NKI69) VERONICA OFFICINALIS FLOWERING TOP (UNII: 9IH82J936J) MELISSA OFFICINALIS LEAF (UNII: 50D2ZE9219) ACHILLEA MILLEFOLIUM (UNII: 2FXJ6SW4PK) CAESALPINIA SPINOSA RESIN (UNII: WL3883U2PO) HYPOXIS HEMEROCALLIDEA ROOT (UNII: V9TSU13YI3) LICORICE (UNII: 61ZBX54883) ARCTOSTAPHYLOS UVA-URSI LEAF (UNII: 3M5V3D1X36) CITRUS BIOFLAVONOIDS (UNII: BD70459I50) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) STEARIC ACID (UNII: 4ELV7Z65AP) SODIUM CETOSTEARYL SULFATE (UNII: 7ZBS06BH4B) GLYCERYL PALMITATE (UNII: 6Y2XJ05B35) CORN (UNII: 0N8672707O) HYDROLYZED WHEAT PROTEIN (ENZYMATIC, 3000 MW) (UNII: J2S07SB0YL) DIGLYCERIN (UNII: 3YC120743U) LACTIC ACID (UNII: 33X04XA5AT) LYSINE (UNII: K3Z4F929H6) VITAMIN A (UNII: 81G40H8B0T) ASCORBIC ACID (UNII: PQ6CK8PD0R) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) UBIDECARENONE (UNII: EJ27X76M46) .ALPHA.-LIPOIC ACID (UNII: 73Y7P0K73Y) XANTHAN GUM (UNII: TTV12P4NEE) LEMON OIL (UNII: I9GRO824LL) LAVENDER OIL (UNII: ZBP1YXW0H8) WEST INDIAN LEMONGRASS OIL (UNII: 5BIA40E9ED) SOYBEAN OIL (UNII: 241ATL177A) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:15751-2272-8 1 in 1 BOX 06/27/2017 1 250 g in 1 JAR; Type 0: Not a Combination Product 2 NDC:15751-2272-2 1 in 1 BOX 06/27/2017 2 60 g in 1 JAR; Type 0: Not a Combination Product 3 NDC:15751-2272-5 1 in 1 BOX 06/27/2017 3 15 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 07/26/2012 Labeler - Eminence Organic Skin Care (205753317)