Label: MAETRIA MIRACLE RECIPE PREMIUMSHAMPOO- salicylic acid shampoo
-
Contains inactivated NDC Code(s)
NDC Code(s): 82934-401-01 - Packager: J 1010 Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 13, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
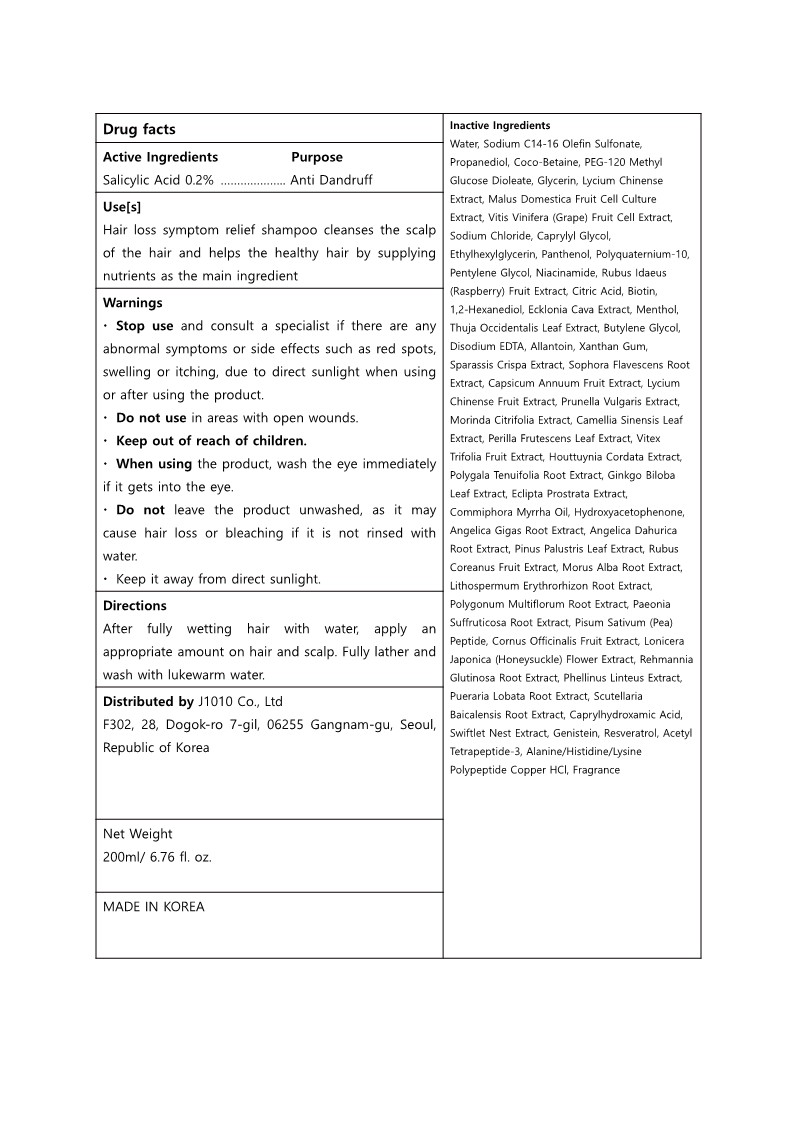
- Active ingredients
- Purpose
- Uses
-
Warnings
· Stop use and consult a specialist if there are any abnormal symptoms or side effects such as red spots, swelling or itching, due to direct sunlight when using or after using the product.
· Do not use in areas with open wounds. · Keep out of reach of children.
· When using the product, wash the eye immediately if it gets into the eye.
· Do not leave the product unwashed, as it may cause hair loss or bleaching if it is not rinsed with water.
· Keep it away from direct sunlight. - Warnings
- Warnings
- Warnings
- Warnings
- Directions
-
Inactive ingredients
Water, Sodium C14-16 Olefin Sulfonate, Propanediol, Coco-Betaine, PEG-120 Methyl Glucose Dioleate, Glycerin, Lycium Chinense Extract, Malus Domestica Fruit Cell Culture Extract, Vitis Vinifera (Grape) Fruit Cell Extract, Sodium Chloride, Caprylyl Glycol, Ethylhexylglycerin, Panthenol, Polyquaternium-10, Pentylene Glycol, Niacinamide, Rubus Idaeus (Raspberry) Fruit Extract, Citric Acid, Biotin,
1,2-Hexanediol, Ecklonia Cava Extract, Menthol, Thuja Occidentalis Leaf Extract, Butylene Glycol, Disodium EDTA, Allantoin, Xanthan Gum, Sparassis Crispa Extract, Sophora Flavescens Root Extract, Capsicum Annuum Fruit Extract, Lycium Chinense Fruit Extract, Prunella Vulgaris Extract, Morinda Citrifolia Extract, Camellia Sinensis Leaf Extract, Perilla Frutescens Leaf Extract, Vitex Trifolia Fruit Extract, Houttuynia Cordata Extract, Polygala Tenuifolia Root Extract, Ginkgo Biloba Leaf Extract, Eclipta Prostrata Extract, Commiphora Myrrha Oil, Hydroxyacetophenone, Angelica Gigas Root Extract, Angelica Dahurica Root Extract, Pinus Palustris Leaf Extract, Rubus Coreanus Fruit Extract, Morus Alba Root Extract, Lithospermum Erythrorhizon Root Extract, Polygonum Multiflorum Root Extract, Paeonia Suffruticosa Root Extract, Pisum Sativum (Pea) Peptide, Cornus Officinalis Fruit Extract, Lonicera Japonica (Honeysuckle) Flower Extract, Rehmannia Glutinosa Root Extract, Phellinus Linteus Extract, Pueraria Lobata Root Extract, Scutellaria Baicalensis Root Extract, Caprylhydroxamic Acid, Swiftlet Nest Extract, Genistein, Resveratrol, Acetyl Tetrapeptide-3, Alanine/Histidine/Lysine Polypeptide Copper HCl, Fragrance - Display panel
-
INGREDIENTS AND APPEARANCE
MAETRIA MIRACLE RECIPE PREMIUMSHAMPOO
salicylic acid shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82934-401 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 1 g in 100 mL Inactive Ingredients Ingredient Name Strength PEG-120 METHYL GLUCOSE DIOLEATE (UNII: YM0K64F20V) LYCIUM CHINENSE FRUIT (UNII: TG711Q7A1Q) PRUNELLA VULGARIS (UNII: Y45L13XZ2U) MORINDA CITRIFOLIA LEAF (UNII: 7UOL7P5FF5) ANGELICA DAHURICA ROOT (UNII: 1V63N2S972) LITHOSPERMUM ERYTHRORHIZON ROOT (UNII: 9I70D8O47I) PAEONIA X SUFFRUTICOSA ROOT (UNII: 7M7E9A2C8J) CORNUS OFFICINALIS FRUIT (UNII: 23NL8NQ187) PEA (UNII: W4X7H8GYFM) ACENEURAMIC ACID (UNII: GZP2782OP0) WINE GRAPE (UNII: 3GOV20705G) SODIUM C14-16 OLEFIN SULFONATE (UNII: O9W3D3YF5U) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) PANTHENOL (UNII: WV9CM0O67Z) NIACINAMIDE (UNII: 25X51I8RD4) PUERARIA MONTANA VAR. LOBATA ROOT (UNII: PET93F4I3C) ALANINE (UNII: OF5P57N2ZX) WATER (UNII: 059QF0KO0R) PHELLINUS LINTEUS WHOLE (UNII: YVO92B1UCA) HOUTTUYNIA CORDATA FLOWERING TOP (UNII: RH041UUZ22) ECLIPTA PROSTRATA LEAF (UNII: H86R96580E) SODIUM CHLORIDE (UNII: 451W47IQ8X) GREEN TEA LEAF (UNII: W2ZU1RY8B0) GINKGO (UNII: 19FUJ2C58T) MYRRH OIL (UNII: H74221J5J4) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) RASPBERRY (UNII: 4N14V5R27W) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) BIOTIN (UNII: 6SO6U10H04) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) ECKLONIA CAVA (UNII: UXX2N5V39P) MENTHOL (UNII: L7T10EIP3A) THUJA OCCIDENTALIS LEAF (UNII: 0T0DQN8786) SPARASSIS CRISPA FRUITING BODY (UNII: BOO3RTP9PX) PERILLA FRUTESCENS LEAF (UNII: T4L5881Y68) VITEX TRIFOLIA FRUIT (UNII: Q04E1F5351) POLYGALA TENUIFOLIA ROOT (UNII: 5S7W573MTU) ANGELICA GIGAS ROOT (UNII: 32766B2FHX) SODIUM SULFATE (UNII: 0YPR65R21J) TETRADECENE (UNII: FW23481S7S) HEXADECENE (MIXED ISOMERS) (UNII: 38H8547VP0) CAPRYLYL GLYCOL (UNII: 00YIU5438U) COCO-BETAINE (UNII: 03DH2IZ3FY) GLYCERIN (UNII: PDC6A3C0OX) PROPANEDIOL (UNII: 5965N8W85T) POLYQUATERNIUM-10 (1000 MPA.S AT 2%) (UNII: GMR4PEN8PK) PENTYLENE GLYCOL (UNII: 50C1307PZG) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) ALLANTOIN (UNII: 344S277G0Z) PINUS PALUSTRIS LEAF (UNII: OT6R5143A1) RUBUS COREANUS FRUIT (UNII: 18VM55XVF7) MORUS ALBA ROOT (UNII: CST1G9BZGD) REYNOUTRIA MULTIFLORA ROOT (UNII: AUZ3VD75MC) LONICERA JAPONICA FLOWER (UNII: 4465L2WS4Y) REHMANNIA GLUTINOSA ROOT (UNII: 1BEM3U6LQQ) RESVERATROL (UNII: Q369O8926L) FRAGRANCE FLORAL ORC0902236 (UNII: R66Z4YW3X0) ACETYL TETRAPEPTIDE-3 (UNII: D1HW9N9QBX) SCUTELLARIA BAICALENSIS ROOT (UNII: 7J95K7ID2S) CAPRYLHYDROXAMIC ACID (UNII: UPY805K99W) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) XANTHAN GUM (UNII: TTV12P4NEE) SOPHORA FLAVESCENS ROOT (UNII: IYR6K8KQ5K) PAPRIKA (UNII: X72Z47861V) GENISTEIN (UNII: DH2M523P0H) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82934-401-01 1 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 10/13/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final M032 10/13/2022 Labeler - J 1010 Co., Ltd. (695562861) Registrant - J 1010 Co., Ltd. (695562861) Establishment Name Address ID/FEI Business Operations J 1010 Co., Ltd. 695562861 manufacture(82934-401)