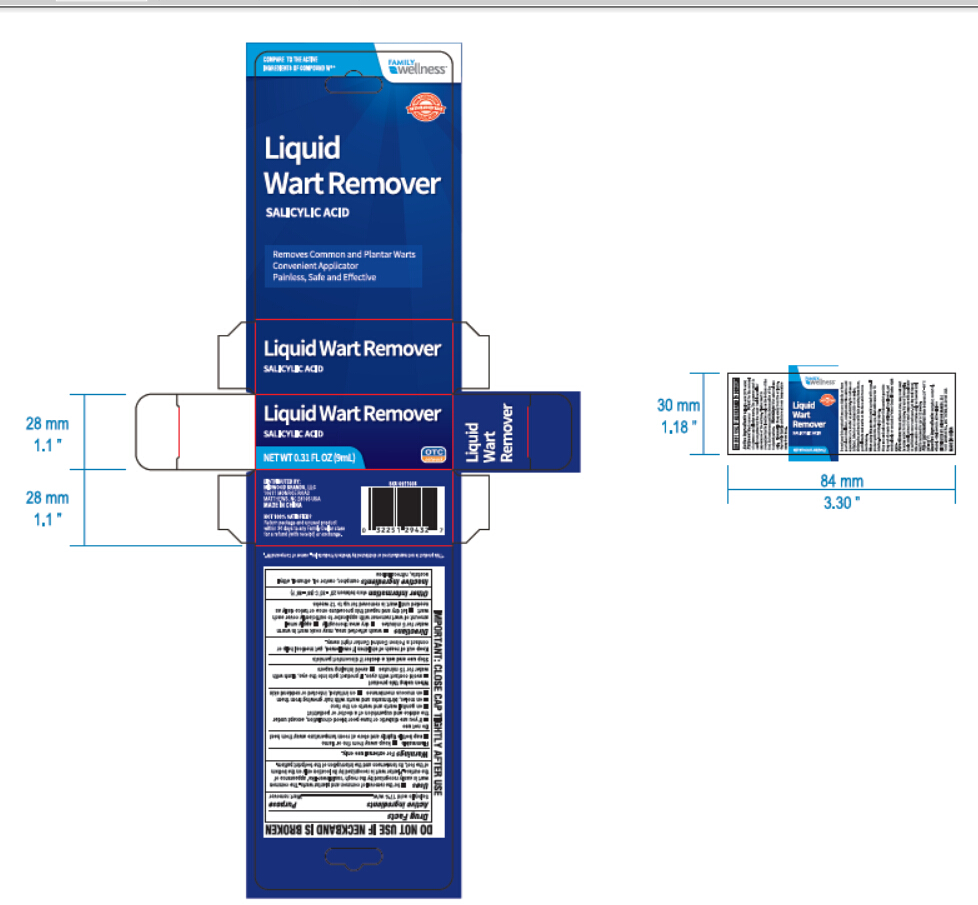
Label: FAMILY WELLNESS LIQUID WART REMOVER- salicylic acid liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 58194-600-01 - Packager: Shanghai Weierya Daily Chemicals Factory
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 25, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings For external use only.
Flammable •keep away from fire or flame
•cap bottle tightly and store at room temperature away from heat
Do not use
•if you are diabetic or have poor blood circulation, except under
the advice and supervision of a doctor or a podiatrist
•on genital warts and warts on the face
•on moles, birthmarks and warts with hair growing from them
•on mucuous membranes •on irritated, infected or reddend skin
When using this product
•avoid contact with eyes. If product gets into the eye, flush with
water for 15 minutes •avoid inhaling vapors
Stop use and ask a doctor if discomfort persists.
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FAMILY WELLNESS LIQUID WART REMOVER
salicylic acid liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58194-600 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 17 g in 100 mL Inactive Ingredients Ingredient Name Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) CASTOR OIL (UNII: D5340Y2I9G) ALCOHOL (UNII: 3K9958V90M) ETHYL ACETATE (UNII: 76845O8NMZ) PYROXYLIN (UNII: KYR8BR2X6O) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58194-600-01 1 in 1 CARTON 06/25/2017 1 9 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part358B 06/25/2017 Labeler - Shanghai Weierya Daily Chemicals Factory (420359333) Establishment Name Address ID/FEI Business Operations Shanghai Weierya Daily Chemicals Factory 420359333 manufacture(58194-600)