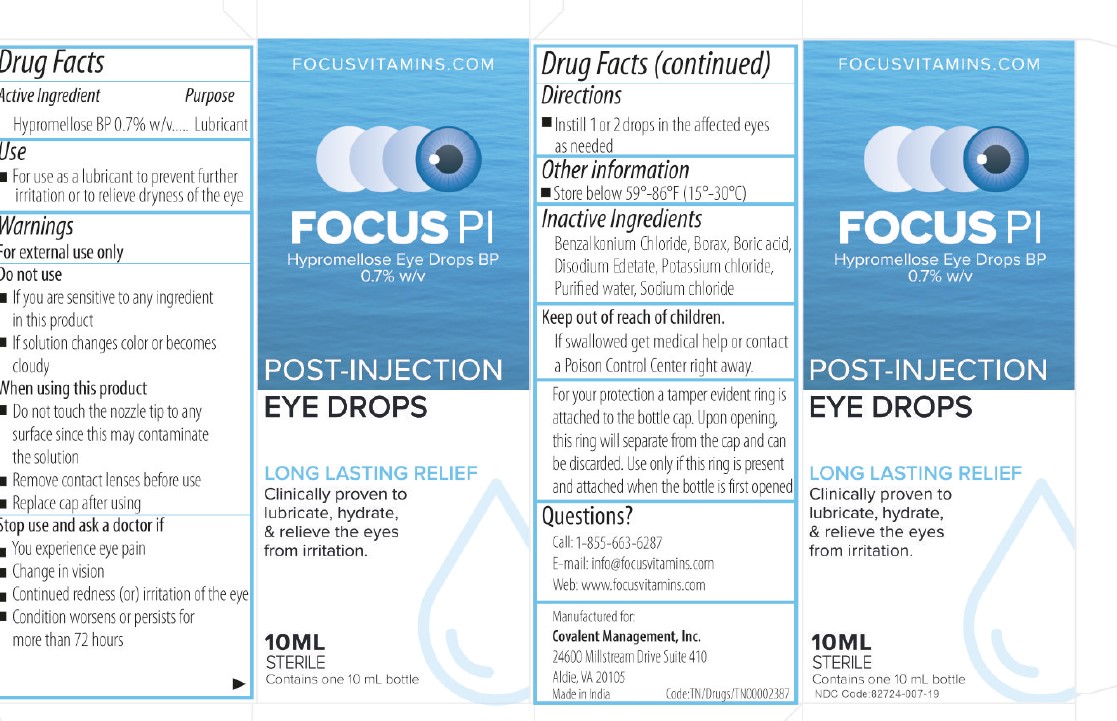
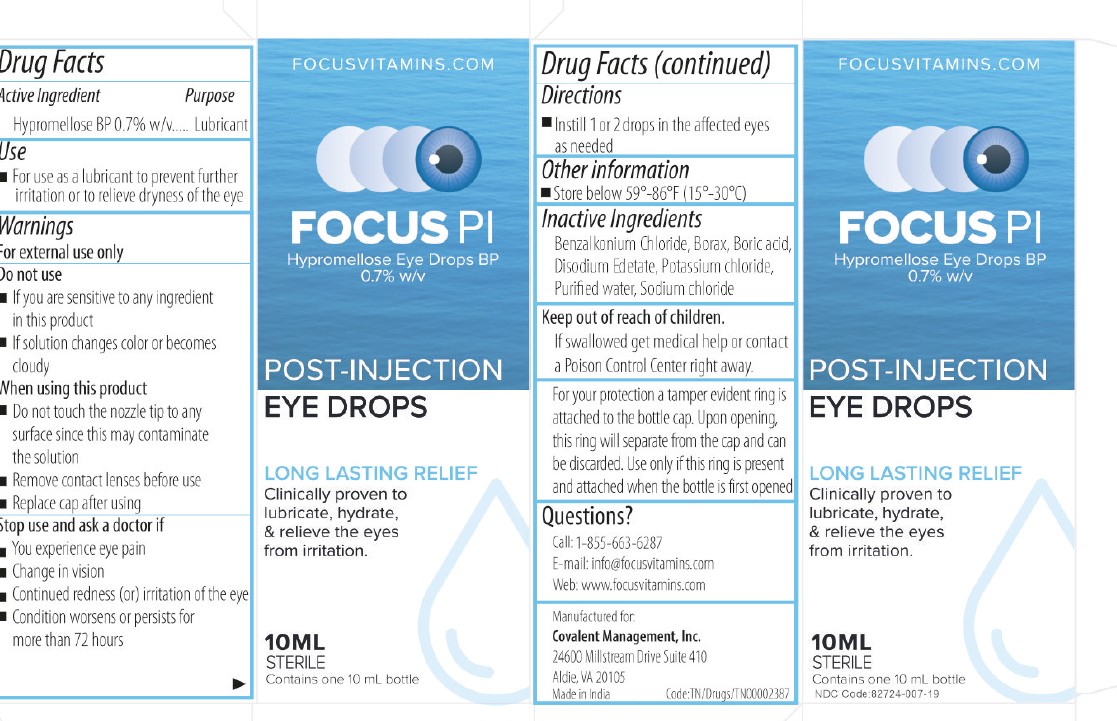
Label: HYPROMELLOSE EYE DROPS 0.7% for solution
- NDC Code(s): 82724-007-19
- Packager: COVALENT MEDICAL
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 19, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- DIRECTIONS FOR USE
- INACTIVE INGREDIENT
- Tamper Protection
- USE
- Questions
- KEEP OUT OF REACH OF CHILDREN
- ASK DOCTOR
- DO NOT USE
- Dosage
- Warnings
- Indications and Usage
- Eye Lubricant
- CARTON LABEL
-
INGREDIENTS AND APPEARANCE
HYPROMELLOSE EYE DROPS 0.7%
hypromellose eye drops 0.7% for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82724-007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYPROMELLOSE 2910 (4000 MPA.S) (UNII: RN3152OP35) (HYPROMELLOSE 2910 (4000 MPA.S) - UNII:RN3152OP35) HYPROMELLOSE 2910 (4000 MPA.S) 7 mg in 1 mL Inactive Ingredients Ingredient Name Strength EDETATE DISODIUM (UNII: 7FLD91C86K) BORIC ACID (UNII: R57ZHV85D4) WATER (UNII: 059QF0KO0R) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) SODIUM BORATE (UNII: 91MBZ8H3QO) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82724-007-19 10 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 09/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M018 09/01/2022 Labeler - COVALENT MEDICAL (112467517) Registrant - AUROLAB (677319965) Establishment Name Address ID/FEI Business Operations AUROLAB 677319965 manufacture(82724-007)