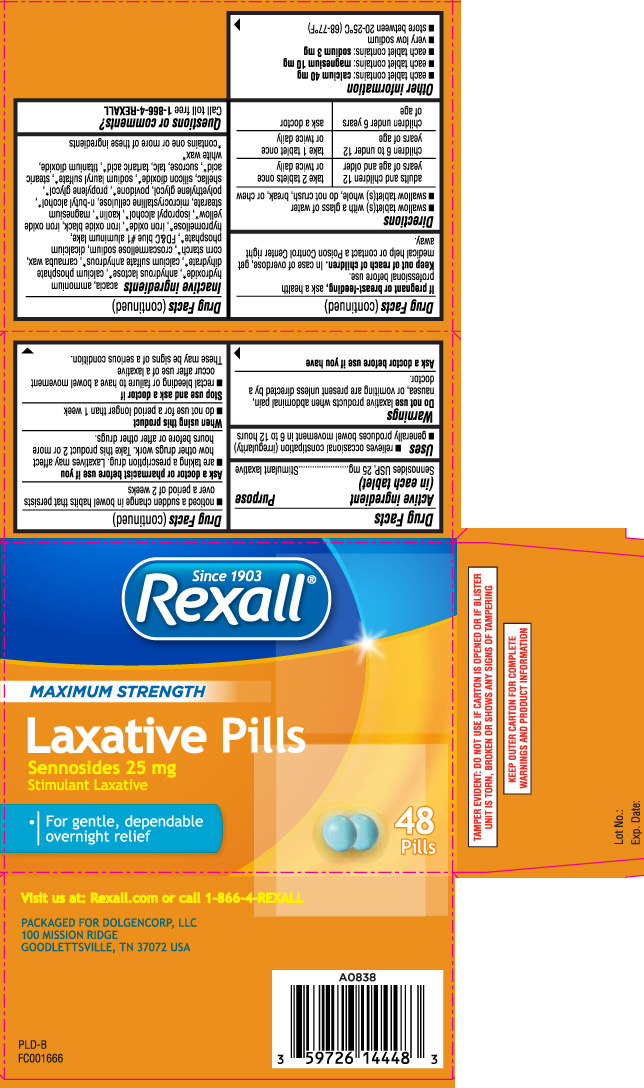
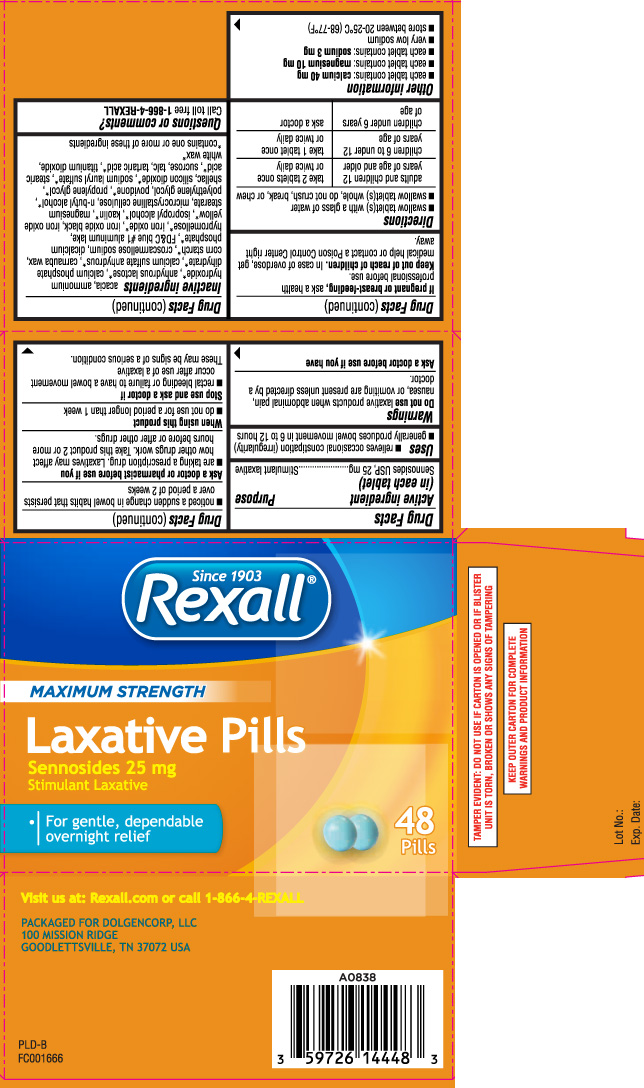
Label: LAXATIVE PILLS MAXIMUM STRENGTH- sennosides tablet
- NDC Code(s): 55910-245-48
- Packager: Dolgencorp, Inc. (DOLLAR GENERAL & REXALL)
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 19, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Do not use
laxative products when abdominal pain, nausea, or vomiting are present unless directed by a doctor.
Ask a doctor before use if you have
- noticed a sudden change in bowel habits that persists over a period of 2 weeks
Ask a doctor or pharmacist before use if you
- are taking a prescription drug. Laxatives may affect how other drugs work. Take this product 2 or more hours before or after other drugs.
- Directions
- Other information
-
Inactive ingredients
acacia, ammonium hydroxide*, anhydrous lactose*, calcium phosphate dihydrate*, calcium sulfate anhydrous*, carnauba wax, corn starch*, croscarmellose sodium, dicalcium phosphate*, FD&C blue #1 aluminum lake, hypromellose*, iron oxide*, iron oxide black, iron oxide yellow*, isopropyl alcohol*, kaolin*, magnesium stearate, microcrystalline cellulose, n-butyl alcohol*, polyethylene glycol, povidone*, propylene glycol*, shellac, silicon dioxide*, sodium lauryl sulfate*, stearic acid*, sucrose, talc, tartaric acid*, titanium dioxide, white wax*
*contains one or more of these ingredients
- Questions or comments?
-
Principal Display Panel
MAXIMUM STRENGTH
Laxative Pills
Sennosides 25 mg
Stimulant Laxative
- For gentle, dependable overnight relief
Visit us at: Rexall.com or call 1-866-4-REXALL
PACKAGED FOR DOLGENCORP, LLC
100 MISSION RIDGE
GOODLETTSVILLE, TN 37072 USA
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION
- Product Label
-
INGREDIENTS AND APPEARANCE
LAXATIVE PILLS MAXIMUM STRENGTH
sennosides tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55910-245 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SENNOSIDES (UNII: 3FYP5M0IJX) (SENNOSIDES - UNII:3FYP5M0IJX) SENNOSIDES 25 mg Inactive Ingredients Ingredient Name Strength ACACIA (UNII: 5C5403N26O) AMMONIA (UNII: 5138Q19F1X) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CALCIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: O7TSZ97GEP) CALCIUM SULFATE ANHYDROUS (UNII: E934B3V59H) CARNAUBA WAX (UNII: R12CBM0EIZ) STARCH, CORN (UNII: O8232NY3SJ) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) CALCIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: L11K75P92J) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) ALUMINUM OXIDE (UNII: LMI26O6933) HYPROMELLOSES (UNII: 3NXW29V3WO) FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) ISOPROPYL ALCOHOL (UNII: ND2M416302) KAOLIN (UNII: 24H4NWX5CO) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) STEARIC ACID (UNII: 4ELV7Z65AP) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WHITE WAX (UNII: 7G1J5DA97F) Product Characteristics Color blue Score no score Shape ROUND Size 11mm Flavor Imprint Code TCL083;S25 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55910-245-48 4 in 1 CARTON 12/30/2013 03/30/2025 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M007 12/30/2013 03/30/2025 Labeler - Dolgencorp, Inc. (DOLLAR GENERAL & REXALL) (068331990)