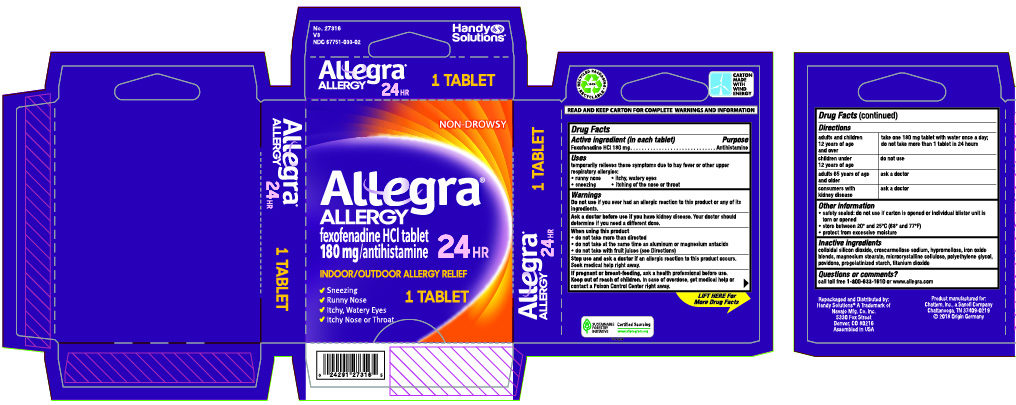
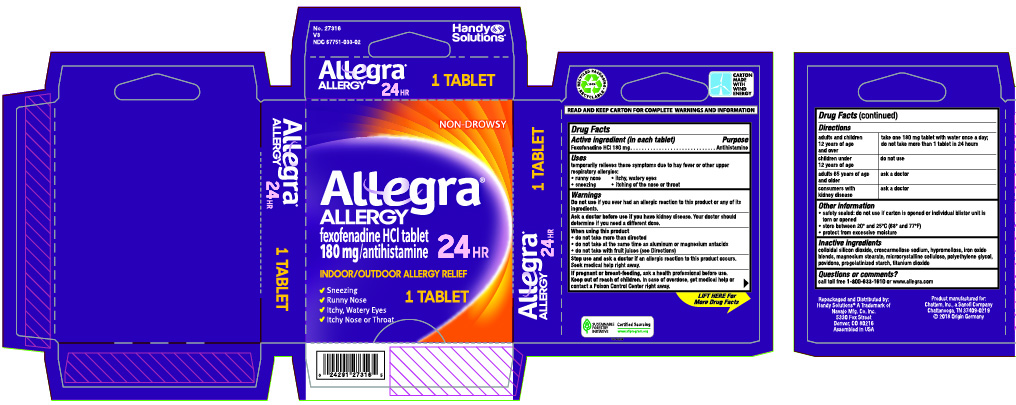
Label: ALLEGRA ALLERGY- fexofenadine hydrochloride tablet, film coated
- NDC Code(s): 67751-033-01, 67751-033-02
- Packager: Navajo Manufacturing Company Inc.
- This is a repackaged label.
- Source NDC Code(s): 41167-4120
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated March 15, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient (in each tablet)
- Uses
-
Warnings
Ask a doctor before use if you have
kidney desease. Your doctor should determine ifyou need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- Product Label
-
INGREDIENTS AND APPEARANCE
ALLEGRA ALLERGY
fexofenadine hydrochloride tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:67751-033(NDC:41167-4120) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FEXOFENADINE HYDROCHLORIDE (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) FEXOFENADINE HYDROCHLORIDE 180 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color orange (peach) Score no score Shape OVAL Size 17mm Flavor Imprint Code 018;E Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67751-033-01 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 05/12/2015 03/31/2016 2 NDC:67751-033-02 1 in 1 CARTON 03/31/2016 2 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020872 05/12/2015 Labeler - Navajo Manufacturing Company Inc. (091917799) Establishment Name Address ID/FEI Business Operations Navajo Manufacturing Company Inc. 136941411 relabel(67751-033) , repack(67751-033)