Label: NYSTATIN tablet, coated
- NDC Code(s): 23155-051-01
- Packager: Heritage Pharmaceuticals Inc. d/b/a Avet Pharmaceuticals Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated May 24, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Nystatin is an antimycotic polyene antibiotic obtained from Streptomyces noursei . Its structural formula:
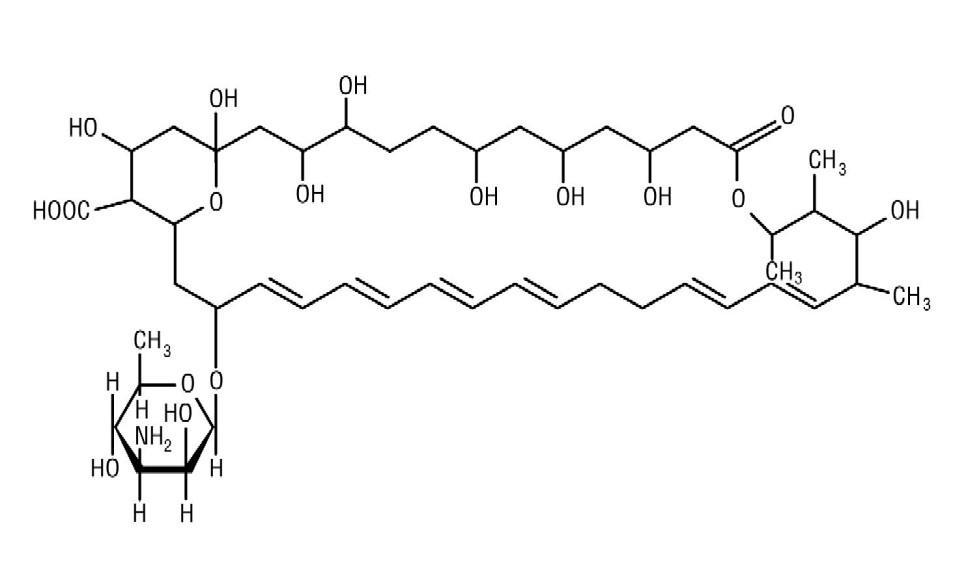
Nystatin tablets are for oral administration and contain 500,000 units of nystatin per tablet.
Nystatin tablets contain the inactive ingredients: corn starch, confectioner sugar, dibasic calcium phosphate, FD&C yellow #6, FD&C red #40, FD&C blue # 2, hydroxypropyl cellulose, hypromellose, microcrystalline cellulose, magnesium stearate, polyethylene glycol, polysorbate 80, talc and titanium dioxide.
-
CLINICAL PHARMACOLOGY
Pharmacokinetics
Gastrointestinal absorption of nystatin is insignificant. Most orally administered nystatin is passed unchanged in the stool. In patients with renal insufficiency receiving oral therapy with conventional dosage forms, significant plasma concentrations of nystatin may occasionally occur.
Microbiology
Nystatin is both fungistatic and fungicidal in vitro against a wide variety of yeasts and yeast-like fungi. Candida albicans demonstrates no significant resistance to nystatin in vitro on repeated subculture in increasing levels of nystatin; other Candida species become quite resistant. Generally, resistance does not develop in vivo. Nystatin acts by binding to sterols in the cell membrane of susceptible Candida species with a resultant change in membrane permeability allowing leakage of intracellular components. Nystatin exhibits no appreciable activity against bacteria, protozoa, or viruses.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
PRECAUTIONS
General
This medication is not to be used for the treatment of systemic mycoses. Discontinue treatment if sensitization or irritation is reported during use.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to evaluate carcinogenic potential. There also have been no studies to determine mutagenicity or whether this medication affects fertility in males or females.
-
ADVERSE REACTIONS
Nystatin is well tolerated even with prolonged therapy. Oral irritation and sensitization have been reported. (See PRECAUTIONS: General.)
Gastrointestinal
Diarrhea (including one case of bloody diarrhea), nausea, vomiting, gastrointestinal upset/disturbances.
-
OVERDOSAGE
Oral doses of nystatin in excess of five million units daily have caused nausea and gastrointestinal upset. There have been no reports of serious toxic effects of superinfections (see CLINICAL PHARMACOLOGY, Pharmacokinetics).
- DOSAGE AND ADMINISTRATION
-
HOW SUPPLIED
Nystatin Tablets USP, 500,000 Units are round brown, film-coated tablets debossed "HP51" on one side and plain on the other side are packaged in:
Bottles of 100: NDC 23155-051-01
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature].
Manufactured by:
Strides Pharma Science Limited
Puducherry- 605 014, India.
PON/DRUGS/16 13 4193
Distributed by:
Avet Pharmaceuticals Inc.
East Brunswick, NJ 08816
1.866.901.DRUG (3784)

Rev: 06/2020
OR
Manufactured by:
Strides Alathur Private Limited
Alathur, Chengalpattu – 603 110,
Tamilnadu, India.M.L. No.: TN00002327
Manufactured for:
Avet Pharmaceuticals Inc.
East Brunswick, NJ 08816
1.866.901.DRUG(3784)

Revised: 01/2024
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NYSTATIN
nystatin tablet, coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:23155-051 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NYSTATIN (UNII: BDF1O1C72E) (NYSTATIN - UNII:BDF1O1C72E) NYSTATIN 500000 [USP'U] Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) SUCROSE (UNII: C151H8M554) HYDROXYPROPYL CELLULOSE (90000 WAMW) (UNII: UKE75GEA7F) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) TALC (UNII: 7SEV7J4R1U) MAGNESIUM STEARATE (UNII: 70097M6I30) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) HYPROMELLOSE 2910 (3 MPA.S) (UNII: 0VUT3PMY82) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYETHYLENE GLYCOL 8000 (UNII: Q662QK8M3B) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) POLYSORBATE 80 (UNII: 6OZP39ZG8H) Product Characteristics Color BROWN Score no score Shape ROUND Size 10mm Flavor Imprint Code HP;51 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:23155-051-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 10/31/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA062474 10/31/2011 Labeler - Heritage Pharmaceuticals Inc. d/b/a Avet Pharmaceuticals Inc. (780779901) Registrant - Heritage Pharmaceuticals Inc. d/b/a Avet Pharmaceuticals Inc. (780779901) Establishment Name Address ID/FEI Business Operations Strides Pharma Science Limited 871402375 MANUFACTURE(23155-051) , ANALYSIS(23155-051) , PACK(23155-051) Establishment Name Address ID/FEI Business Operations Strides Alathur Private Limited 860477684 ANALYSIS(23155-051) , MANUFACTURE(23155-051) , PACK(23155-051)




