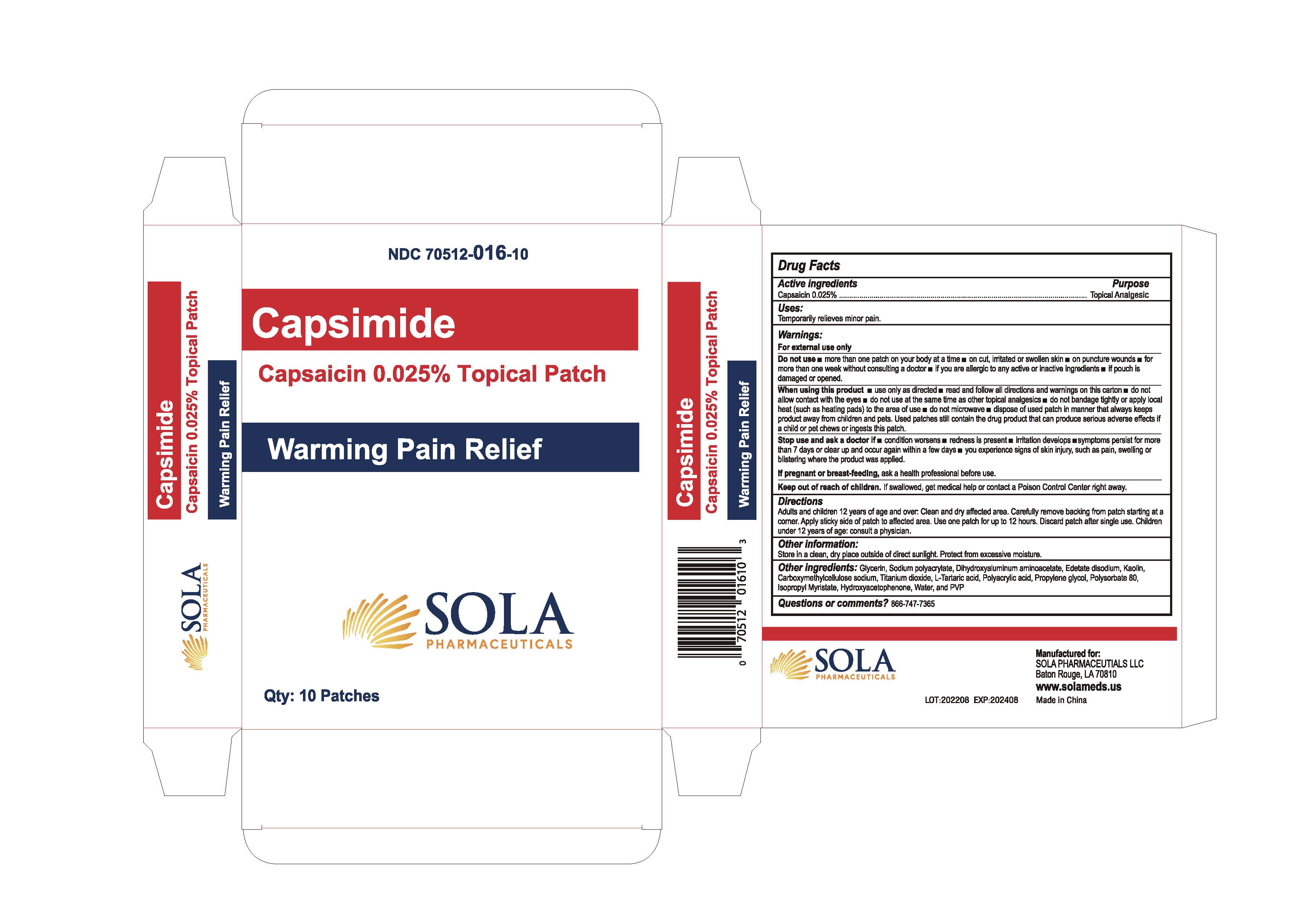
Label: CAPSIMIDE- capsaicin 0.025% topical patch patch
- NDC Code(s): 70512-016-10
- Packager: Sola Pharmaceuticals
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 20, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
- Warnings
- Do not use
-
When using this product
- use only as directed
- read and follow all directions and warnings on this carton
- do not allow contact with the eyes
- do not sue at the same time as other topical analgesics
- do not bandage tightly or apply local heat (such as heating pads) to the area of use
- do not microwave
- dispose of used patch in a manner that always keeps product away from children and pets. Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
- Stop use and ask a doctor if
- If pregnant or breast-feeding
- Keep our of reach of children
- Directions
- Other Information
- Other Ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CAPSIMIDE
capsaicin 0.025% topical patch patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70512-016 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAPSAICIN (UNII: S07O44R1ZM) (CAPSAICIN - UNII:S07O44R1ZM) CAPSAICIN 0.025 g in 100 g Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) WATER (UNII: 059QF0KO0R) DIHYDROXYALUMINUM AMINOACETATE (UNII: DO250MG0W6) EDETATE DISODIUM (UNII: 7FLD91C86K) KAOLIN (UNII: 24H4NWX5CO) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) SODIUM POLYACRYLATE (8000 MW) (UNII: 285CYO341L) POVIDONE (UNII: FZ989GH94E) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TARTARIC ACID (UNII: W4888I119H) POLYACRYLIC ACID (8000 MW) (UNII: 73861X4K5F) POLYSORBATE 80 (UNII: 6OZP39ZG8H) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) GLYCERIN (UNII: PDC6A3C0OX) Product Characteristics Color Score Shape RECTANGLE Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70512-016-10 10 in 1 BOX 09/07/2022 1 9 g in 1 POUCH; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 09/07/2022 Labeler - Sola Pharmaceuticals (080121345)