Label: ALLERGY AND CONGESTION RELIEF NON DROWSY- pseudoephedrine sulfate, loratadine tablet, film coated
-
Contains inactivated NDC Code(s)
NDC Code(s): 42507-724-22 - Packager: HyVee Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 25, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
-
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- sneezing
- itchy, watery eyes
- runny nose
- itching of the nose or throat
- temporarily relieves nasal congestion due to the common cold, hay fever, or other upper respiratory allergies
- reduces swelling of nasal passages
- temporarily relieves sinus congestion and pressure
- temporarily restores freer breathing through the nose
-
Warnings
Do not use
- if you have ever had an allergic reaction to this product or any of its ingredients
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- heart disease
- thyroid disease
- high blood pressure
- diabetes
- trouble urinating due to an enlarged prostate gland
- liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product do not take more than directed.
Taking more than directed may cause drowsiness.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
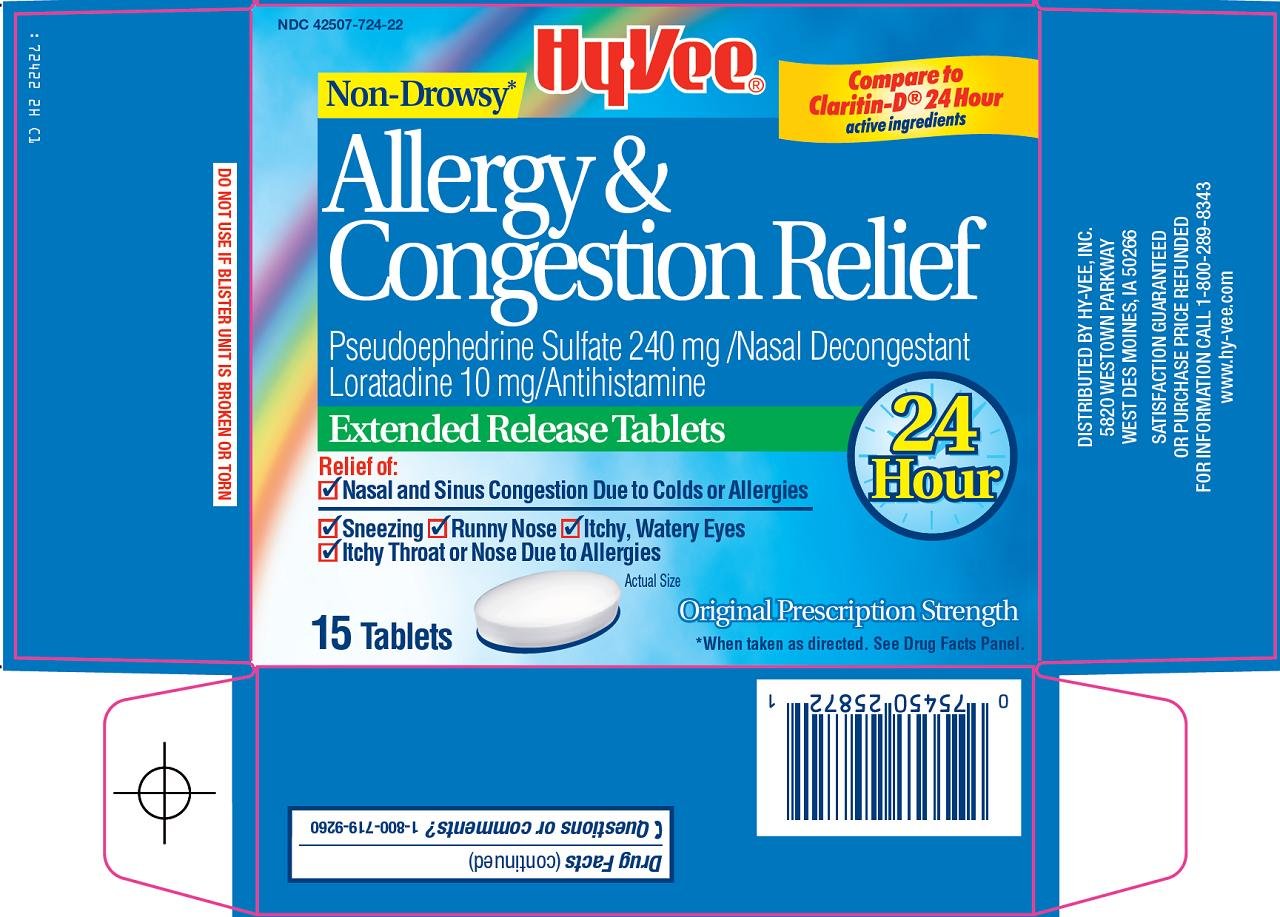
Package/Label Principal Display Panel
Non-Drowsy
Compare to Claritin-D® 24 Hour active ingredients
Allergy & Congestion Relief
Pseudoephedrine Sulfate 240 mg/Nasal Decongestant
Loratadine 10 mg/Antihistamine
Extended Release Tablets
24 hour
Relief of:
Nasal and Sinus Congestion Due to Colds or Allergies
Sneezing
Runny Nose
Itchy, Watery Eyes
Itchy Throat or Nose Due to Allergies
Actual Size
Original Prescription Strength
*When taken as directed. See Drug Facts Panel.
Allergy & Congestion Relief Carton Image 1

Allergy & Congestion Relief Carton Image 2
-
INGREDIENTS AND APPEARANCE
ALLERGY AND CONGESTION RELIEF NON DROWSY
pseudoephedrine sulfate, loratadine tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:42507-724 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 10 mg PSEUDOEPHEDRINE SULFATE (UNII: Y9DL7QPE6B) (PSEUDOEPHEDRINE - UNII:7CUC9DDI9F) PSEUDOEPHEDRINE SULFATE 240 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color WHITE (Off-White) Score no score Shape OVAL Size 18mm Flavor Imprint Code L724 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42507-724-22 3 in 1 CARTON 1 5 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA075989 10/26/2010 Labeler - HyVee Inc (006925671)