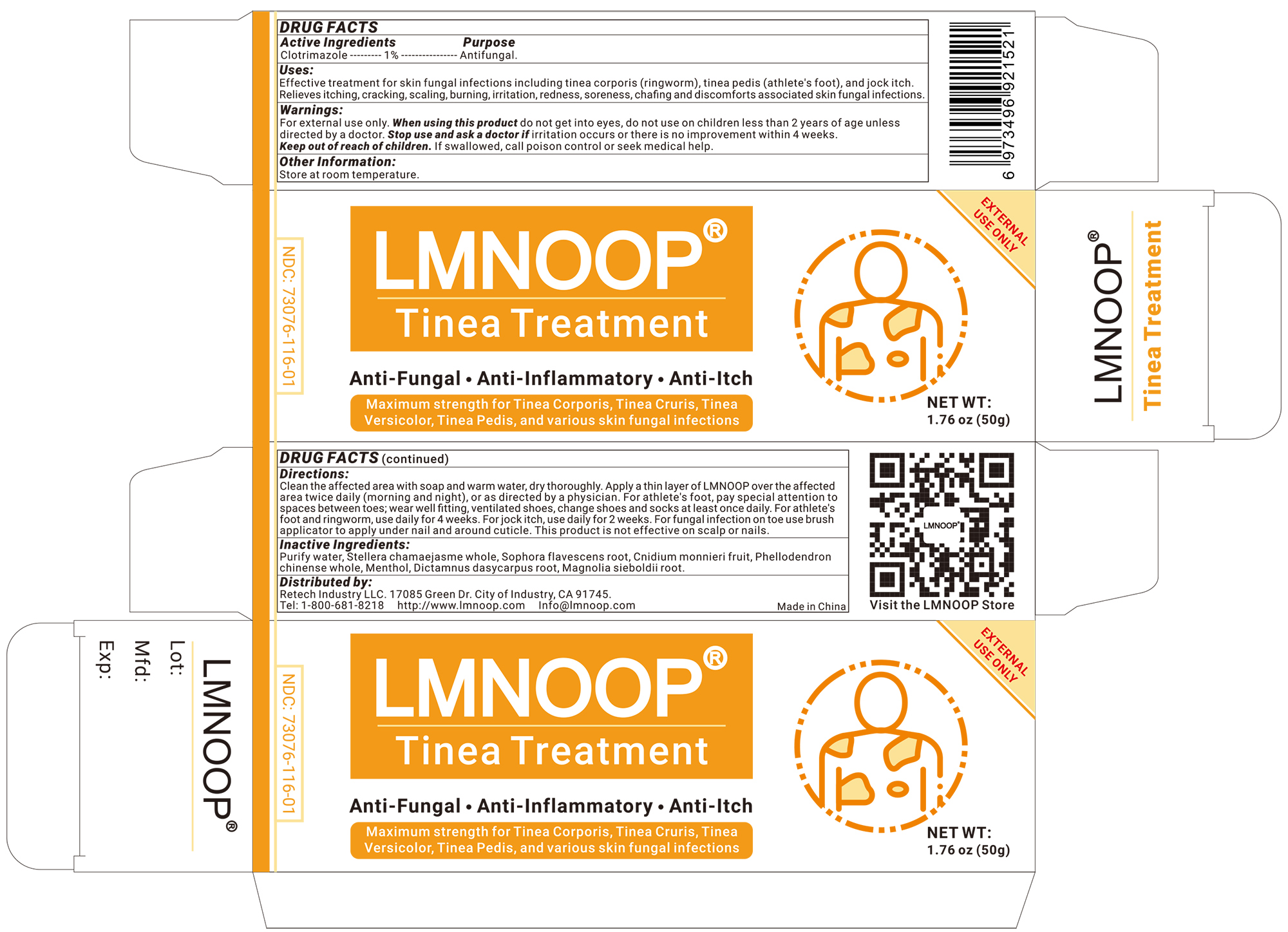
Label: LMNOOP TINEA TREATMENT cream
- NDC Code(s): 73076-116-01, 73076-116-02
- Packager: Shenzhen Ishan Technology Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated October 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Inactive Ingredients
- Keep out of reach of children
- ask a doctor
- Stop use and ask a doctor
- USES
- Other information
-
Directions
Clean the affected area with soap and warm water, dry thoroughly. Apply a thin layer of LMNOOP over the affected area twice daily (morning and night), or as directed by a physician. For athlete's foot, pay special attention to spaces between toes; wear well fitting, ventilated shoes, change shoes and socks at least once daily. For athlete's foot and ringworm, use daily for 4 weeks. For jock itch, use daily for 2 weeks. For fungal infection on toe use brush applicator to apply under nail and around cuticle. This product is not effective on scalp or nails.
- Dosage and administration
- When using this product
-
warning
For external use only. Ask a doctor before use if you have a condition that covers a large area of the body. When Using this product do not get into eyes. Do not use on nails or scalp & near the mouth or the eyes area. Stop use and ask a doctor if condition worsens or does not improve after regular use of this products as directed. Ask a doctor before using on children under 2 years of age. Keep out of reach of children. if swallowed, get medical help or contact a Poison Control Center right away.
- Lable
-
INGREDIENTS AND APPEARANCE
LMNOOP TINEA TREATMENT
lmnoop tinea treatment creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73076-116 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLOTRIMAZOLE (UNII: G07GZ97H65) (CLOTRIMAZOLE - UNII:G07GZ97H65) CLOTRIMAZOLE 1 g in 100 g Inactive Ingredients Ingredient Name Strength SOPHORA FLAVESCENS ROOT (UNII: IYR6K8KQ5K) 3 g in 100 g STELLERA CHAMAEJASME WHOLE (UNII: 80N0P0DD9J) 3 g in 100 g MENTHOL (UNII: L7T10EIP3A) 1 g in 100 g DICTAMNUS DASYCARPUS ROOT (UNII: 6153LEN214) 3 g in 100 g PHELLODENDRON CHINENSE WHOLE (UNII: QKA3ZK8IIE) 3 g in 100 g MAGNOLIA SIEBOLDII ROOT (UNII: F5O3IH7US3) 3 g in 100 g CNIDIUM MONNIERI FRUIT (UNII: V1IA3S3CUS) 3 g in 100 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73076-116-01 50 g in 1 BOTTLE; Type 0: Not a Combination Product 12/01/2022 2 NDC:73076-116-02 100 g in 1 BOTTLE; Type 0: Not a Combination Product 10/17/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 12/01/2022 Labeler - Shenzhen Ishan Technology Co., Ltd (554484192) Establishment Name Address ID/FEI Business Operations Shenzhen Ishan Technology Co., Ltd 554484192 manufacture(73076-116)