Label: SPF 32 DAILY SUSCREEN GHOST DEMOCRACY- zinc oxide cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 81412-001-01 - Packager: Ghost Democracy LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 17, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
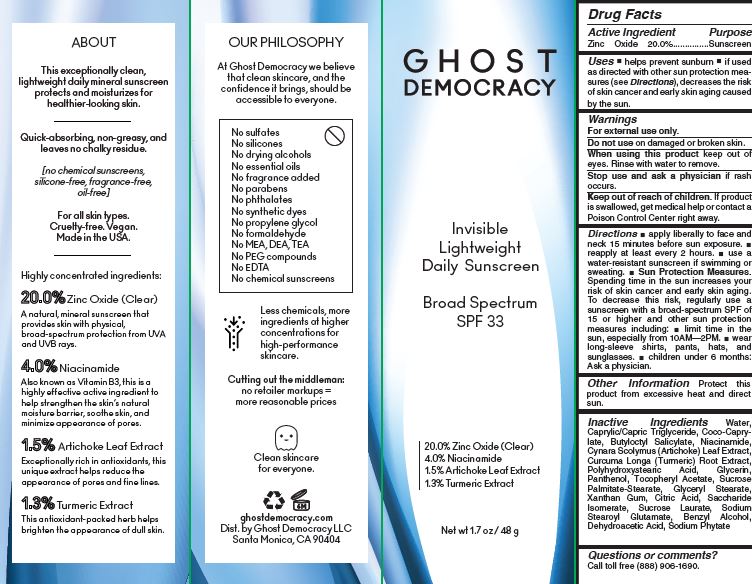
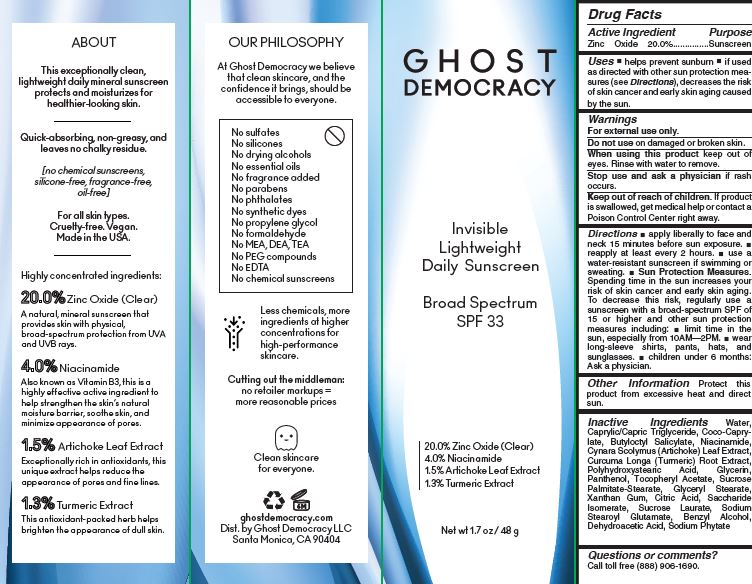
- Active Ingredient Purpose
- PURPOSE
- Uses
- Warnings For external use only.
-
Directions
■ apply liberally to face and neck 15 minutes before sun exposure. ■ reapply at least every 2 hours. ■ use a water-resistant sunscreen if wimming or sweating. ■ Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad-spectrum SPF of 15 or higher and other sun protection measures including: ■ limit time in the
sun, especially from 10AM—2PM. ■ wear long-sleeve shirts, pants, hats, and sunglasses. ■ children under 6 months: Ask a physician. - Other Information
-
Inactive Ingredients
Water,
Caprylic/Capric Triglyceride, Coco-Caprylate,
Butyloctyl Salicylate, Niacinamide,
Cynara Scolymus (Artichoke) Leaf Extract,
Curcuma Longa (Turmeric) Root Extract,
Polyhydroxystearic Acid, Glycerin,
Panthenol, Tocopheryl Acetate, Sucrose
Palmitate-Stearate, Glyceryl Stearate,
Xanthan Gum, Citric Acid, Saccharide
Isomerate, Sucrose Laurate, Sodium
Stearoyl Glutamate, Benzyl Alcohol,
Dehydroacetic Acid, Sodium Phytate - Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SPF 32 DAILY SUSCREEN GHOST DEMOCRACY
zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81412-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 20 g in 100 g Inactive Ingredients Ingredient Name Strength GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) XANTHAN GUM (UNII: TTV12P4NEE) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SACCHARIDE ISOMERATE (UNII: W8K377W98I) SUCROSE LAURATE (UNII: 05Q7CD0E49) SODIUM STEAROYL GLUTAMATE (UNII: 65A9F4P024) BENZYL ALCOHOL (UNII: LKG8494WBH) DEHYDROACETIC ACID (UNII: 2KAG279R6R) PHYTATE SODIUM (UNII: 88496G1ERL) WATER (UNII: 059QF0KO0R) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) COCO-CAPRYLATE (UNII: 4828G836N6) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) NIACINAMIDE (UNII: 25X51I8RD4) ARTICHOKE (UNII: 4F3W47PLBE) TURMERIC (UNII: 856YO1Z64F) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) GLYCERIN (UNII: PDC6A3C0OX) PANTHENOL (UNII: WV9CM0O67Z) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81412-001-01 1 in 1 CARTON 01/12/2021 1 48 g in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 01/12/2021 Labeler - Ghost Democracy LLC (117813286) Registrant - Inspec Solutions LLC (081030372) Establishment Name Address ID/FEI Business Operations Inspec Solutions 081030372 manufacture(81412-001)