Label: ALDAMED- painelimin with collagen cooling gel
- NDC Code(s): 82811-111-11
- Packager: Herbamedicus, s.r.o.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- PURPOSE
- Use
- warnings
- Do not use
- When using this product
- ASK DOCTOR
- Stop use and ask a doctor
- Keep out of reach of children
- Directions
- Other information
-
Inactive ingredient
water, alcohol denat., glycerin, symphytum officinale root extract, aesculus hippocastanum seed extract, eucalyptus globulus leaf oil, abies alba leaf oil, dimethyl sulfone, equisetum arvense extract, mentha piperita oil, magnesium pca, sodium hyaluronate, thymus vulgaris oil, juniperus communis fruit oil, nelumbo nucifera flower water, nymphaea coerulea flower water, arnica montana flower extract, ginkgo biloba leaf extract, hydrolyzed collagen, scutellaria baicalensis extract, pinus pumila twig leaf oil, pinus mugo twig oil, echinacea purpurea extract, agrimonia eupatoria extract, calendula officinalis flower extract, vaccinium myrtillus fruit extract, polygonum bistorta root extract, citrus limon fruit oil, citrus grandis peel oil, rosmarinus officinalis leaf oil, lavandula angustifolia oil, calluna vulgaris extract, ribes nigrum fruit extract, chamomilla recutita flower extract, carbomer, polysorbate 80, phenoxyethanol, sodium hydroxide, ethylhexylglycerin, peg-12 dimethicone
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALDAMED
painelimin with collagen cooling gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82811-111 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 3 mg in 100 mL MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 3 mg in 100 mL Inactive Ingredients Ingredient Name Strength NYMPHAEA CAERULEA FLOWER VOLATILE OIL (UNII: 869334Z0JF) CITRUS MAXIMA FRUIT RIND OIL (UNII: 8U3877WD44) GINKGO (UNII: 19FUJ2C58T) GLYCERIN (UNII: PDC6A3C0OX) LAVENDER OIL (UNII: ZBP1YXW0H8) PEG-12 DIMETHICONE (UNII: ZEL54N6W95) POLYSORBATE 80 (UNII: 6OZP39ZG8H) AGRIMONIA EUPATORIA (UNII: EBU0U94820) CITRUS LIMON FRUIT OIL (UNII: 0HNC1J1YED) PINE NEEDLE OIL (PINUS MUGO) (UNII: 6KF1RLN1CI) CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) ABIES ALBA LEAF OIL (UNII: G49QS877DA) HORSE CHESTNUT (UNII: 3C18L6RJAZ) ARNICA MONTANA FLOWER (UNII: OZ0E5Y15PZ) EUCALYPTUS OIL (UNII: 2R04ONI662) SCUTELLARIA BAICALENSIS ROOT (UNII: 7J95K7ID2S) WATER (UNII: 059QF0KO0R) PHENOXYETHANOL (UNII: HIE492ZZ3T) POLYGONUM BISTORTA ROOT (UNII: X23X6CW68R) SODIUM HYDROXIDE (UNII: 55X04QC32I) COMFREY ROOT (UNII: M9VVZ08EKQ) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) PEPPERMINT OIL (UNII: AV092KU4JH) THYME OIL (UNII: 2UK410MY6B) NELUMBO NUCIFERA FLOWER OIL (UNII: P658Q19EG2) BLACK CURRANT (UNII: 9755T40D11) ROSEMARY OIL (UNII: 8LGU7VM393) HYALURONATE SODIUM (UNII: YSE9PPT4TH) BILBERRY (UNII: 9P2U39H18W) ECHINACEA PURPUREA (UNII: QI7G114Y98) ALCOHOL (UNII: 3K9958V90M) CALLUNA VULGARIS WHOLE (UNII: DTE28NXG2W) CARBOXYPOLYMETHYLENE (UNII: 0A5MM307FC) CHAMOMILE (UNII: FGL3685T2X) DIMETHYL SULFONE (UNII: 9H4PO4Z4FT) EQUISETUM ARVENSE BRANCH (UNII: 1L0VKZ185E) HYDROLYSED MARINE COLLAGEN (ENZYMATIC; 2000 MW) (UNII: 2WID9OCG7P) JUNIPER BERRY OIL (UNII: SZH16H44UY) MAGNESIUM PIDOLATE (UNII: V5PC588N7G) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82811-111-11 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 08/08/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 08/08/2022 Labeler - Herbamedicus, s.r.o. (495275964) Establishment Name Address ID/FEI Business Operations Green Idea s.r.o. 361646523 manufacture(82811-111)

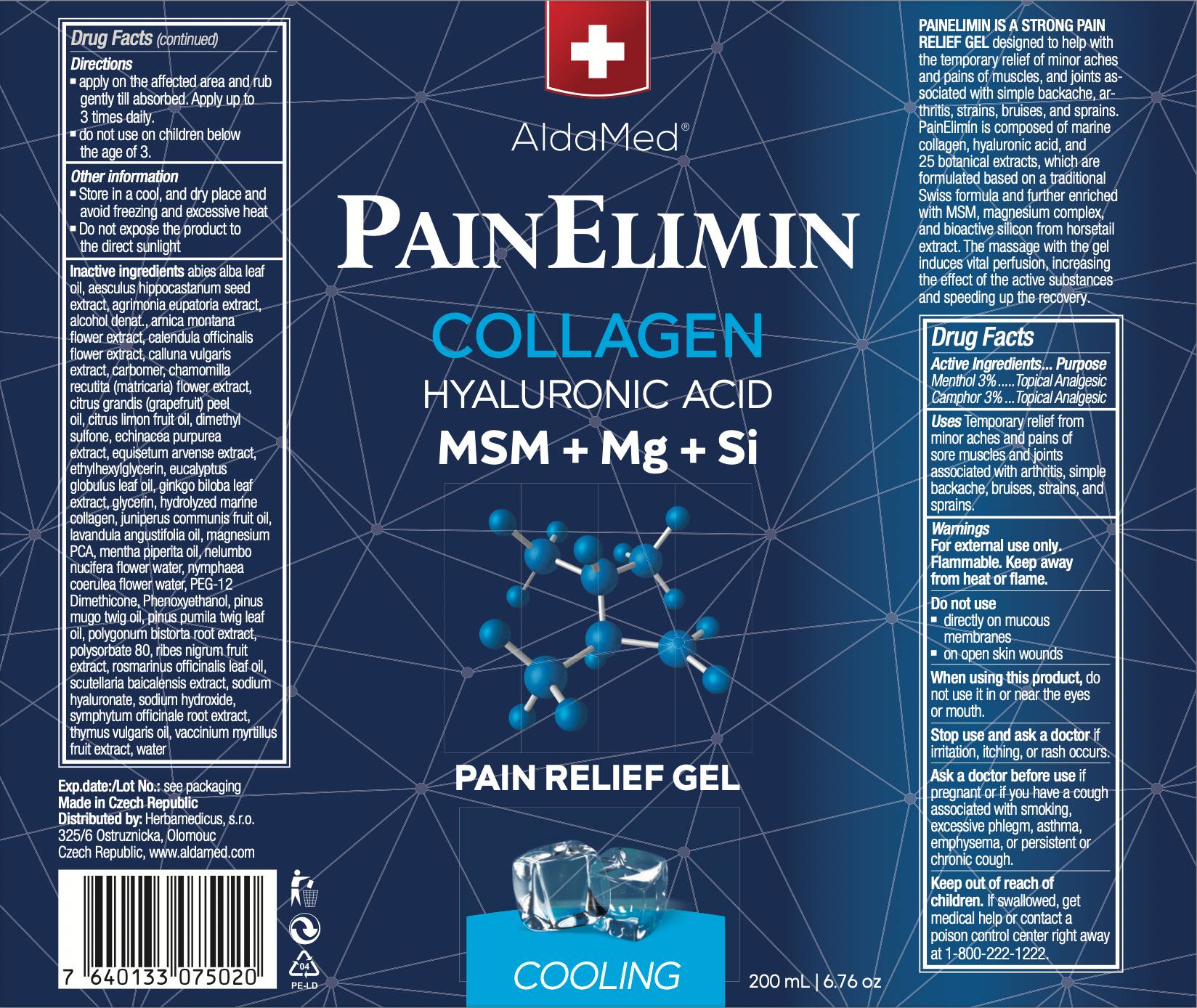
 200 mL, NDC: 82811-111-11
200 mL, NDC: 82811-111-11