Label: UNISHIELD PAIN ACHE- aspirin, caffeine tablet, film coated
- NDC Code(s): 49314-8218-0
- Packager: Unishield
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 17, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
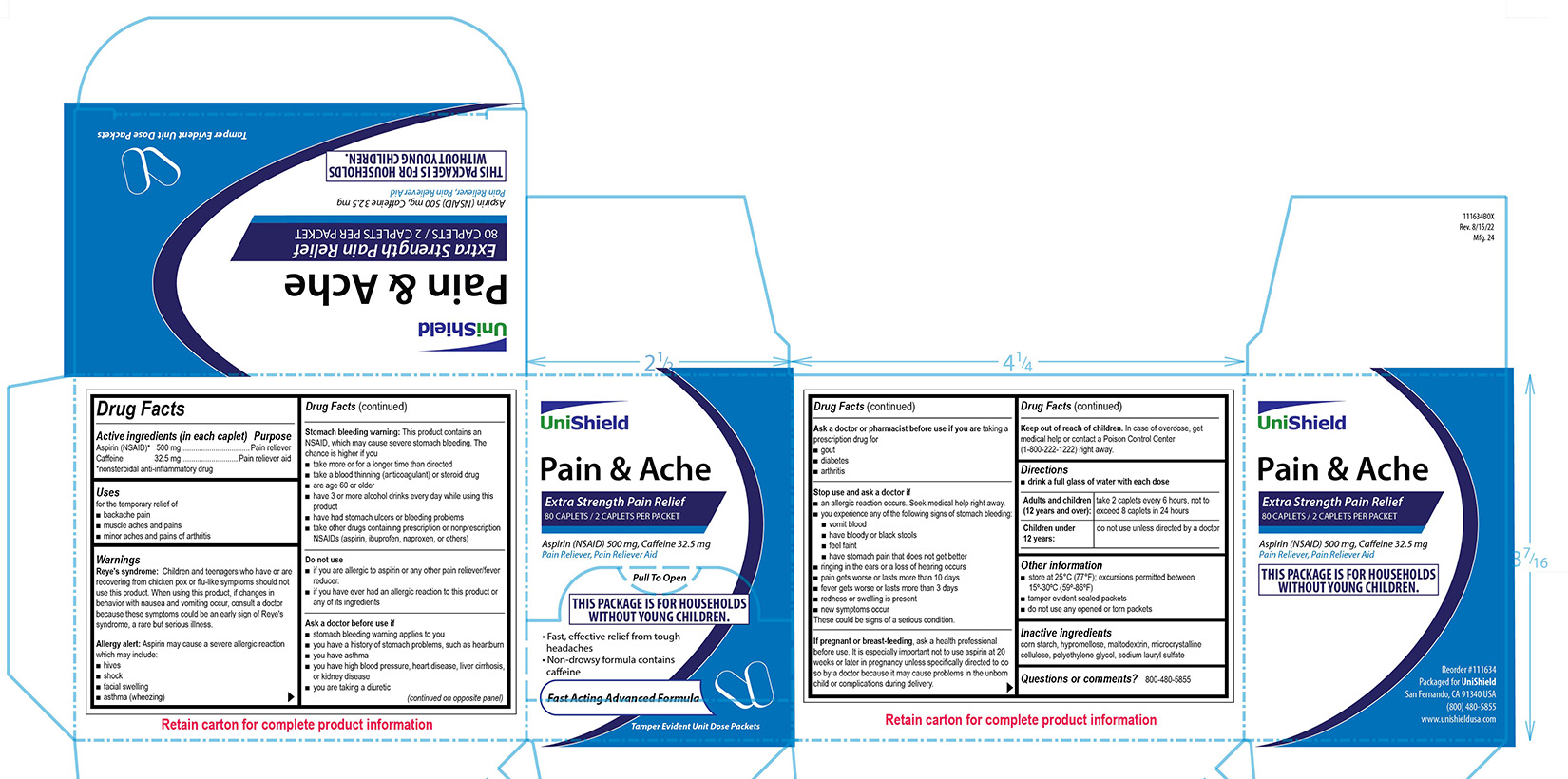
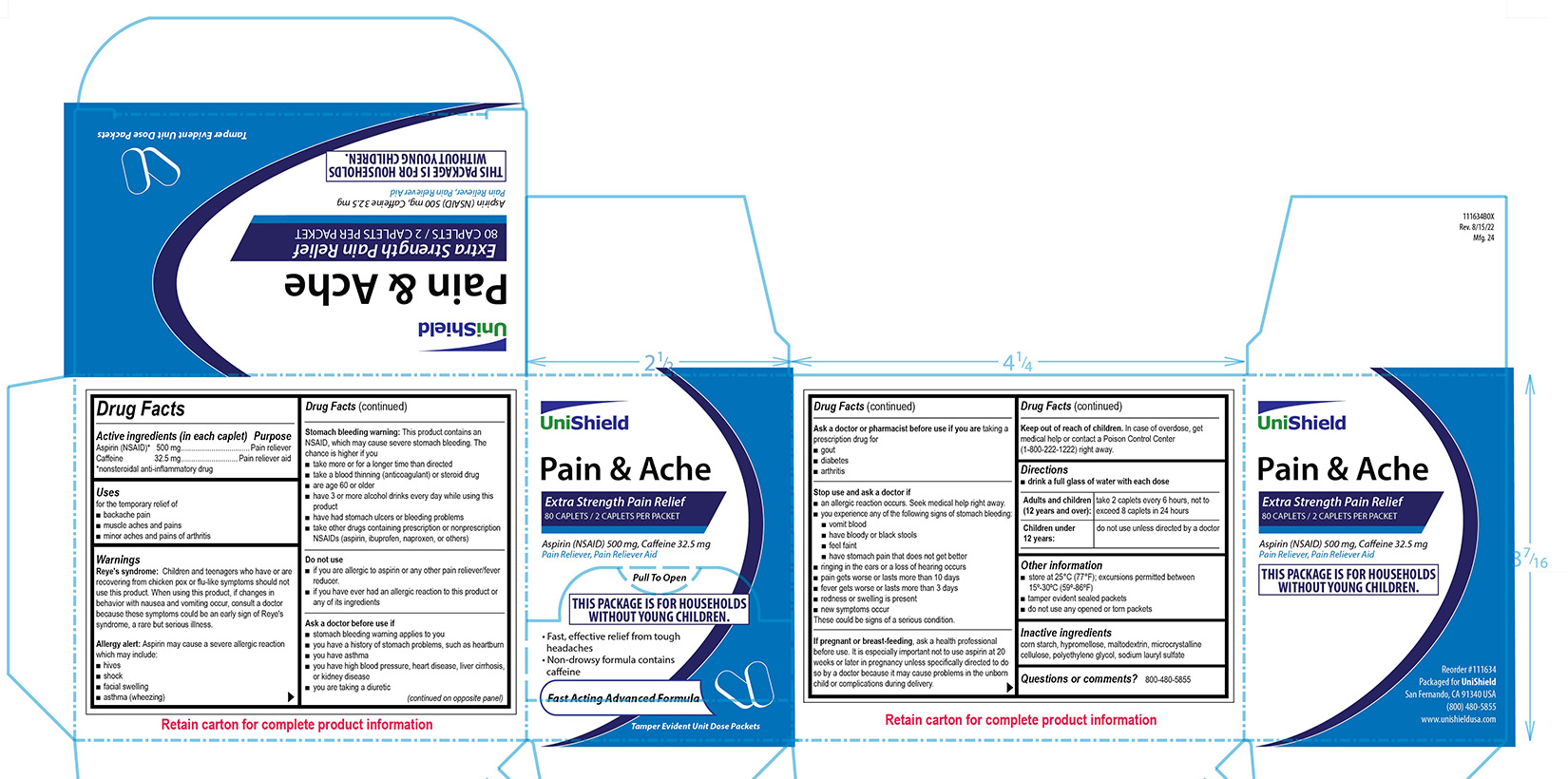
Reye's syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert:
Aspirin may cause a severe allergic reaction which may include:
■ hives
■ shock
■ facial swelling
■ asthma (wheezing)
Stomach bleeding warning:
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
■ take more or for a longer time than directed
■ take a blood thinning (anticoagulant) or steroid drug
■ are age 60 or older
■ have 3 or more alcohol drinks every day while using this product
■ have had stomach ulcers or bleeding problems
■ take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- DO NOT USE
- ASK DOCTOR
- ASK DOCTOR/PHARMACIST
-
STOP USE
Stop use and ask a doctor if
■ an allergic reaction occurs. Seek medical help right away.
■ you experience any of the following signs of stomach bleeding: ■ vomit blood ■ have bloody or black stools ■ feel faint ■ have stomach pain that does not get better
■ ringing in the ears or a loss of hearing occurs
■ pain gets worse or lasts more than 10 days
■ fever gets worse or lasts more than 3 days
■ redness or swelling is present
■ new symptoms occur
These could be signs of a serious condition.
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
-
PRINCIPAL DISPLAY PANEL
Unishield
Pain & Ache
Extra Strength Pain Relief
80 caplets / 2 Caplets Per Packet
Aspirin (NSAID) 500 mg, Caffeine 32.5 mg
Pain Reliever, Pain Reliever Aid
Pull To Open
This package is for Households Without young Children.
• Fast, effective relief from tough headaches
• Non-drowsy formula contains caffeine
Fast Acting Advanced Formula
Tamper Evident Unit Dose Packets
-
INGREDIENTS AND APPEARANCE
UNISHIELD PAIN ACHE
aspirin, caffeine tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49314-8218 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAFFEINE (UNII: 3G6A5W338E) (CAFFEINE - UNII:3G6A5W338E) CAFFEINE 32.5 mg ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 500 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MALTODEXTRIN (UNII: 7CVR7L4A2D) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SODIUM LAURYL SULFATE (UNII: 368GB5141J) STARCH, CORN (UNII: O8232NY3SJ) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) Product Characteristics Color white Score no score Shape CAPSULE (Caplet) Size 17mm Flavor Imprint Code 44;413 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49314-8218-0 40 in 1 BOX 09/26/2022 1 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 09/26/2022 Labeler - Unishield (790677053) Registrant - Unifirst First Aid Corporation (832947092)