Label: SEVERE CONGESTION AND COUGH MAX MAXIUM STRENGTH- dextromethorphan hydrobromide, guaifenesin, phenylephrine hydrochloride liquid
- NDC Code(s): 68016-221-00
- Packager: Chain Drug Consortium, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 29, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Keep out of reach of children
- Uses
-
Warnings
Do not use
- If you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for two weeks after stopping the MAOI drug. If you do not know if your child’s prescription drug contains an MAOI, ask a doctor or a pharmacist before taking this product.
- for children under 12 years of age
- Ask a doctor before use if you have
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast feeding
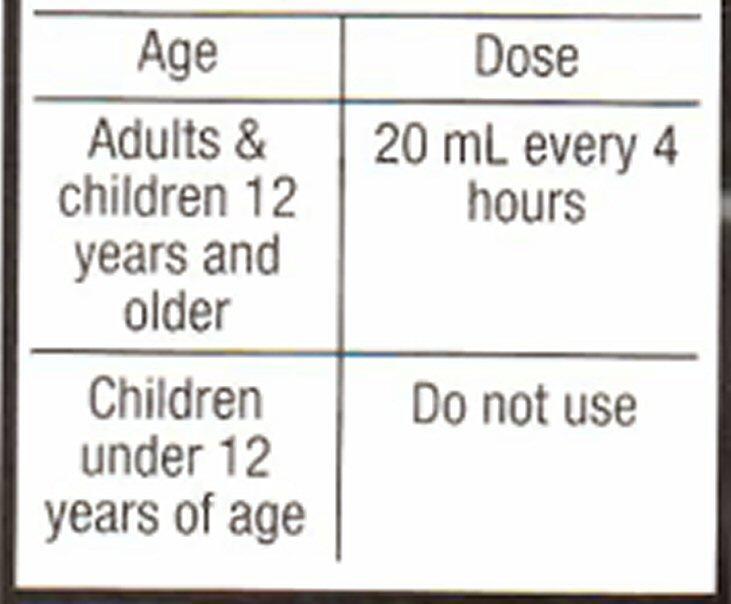
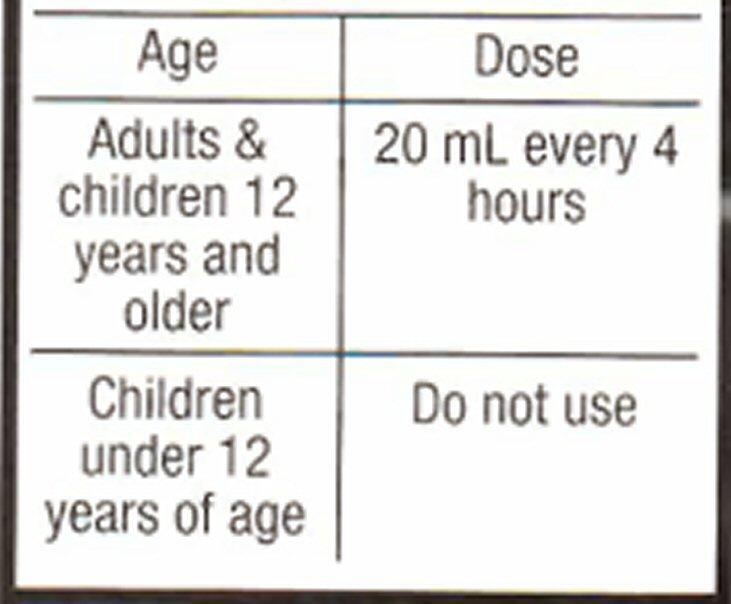
- Directions
- Other information
- Inactive ingredients
- Questions?
-
Product Label
NDC 68016-221-00
*COMPARE TO THE ACTIVE INGREDIENTS IN MUCINEX® FAST-MAX™ SEVERE CONGESTION AND COUGH LF-004
Premier Value®Maximum Strength
Severe Congestion and Cough MAX
Dextromethorphan HBr / Guaifenesin / Phenylephrine HCL
COUGH SUPPRESSANT / EXPECTORANT/ NASAL DECONGESTANT- Controls Cough
- Thins and Loosens Mucus
- Relieves Nasal and Chest Congestion
Adults
For Ages 12 and Over
6 FL OZ (177mL)
INDEPENDENTLY TESTED SATISFACTION GUARANTEED PV
DO NOT USE IF PRINTED SEAL UNDER CAP IS TORN OR MISSING
Peel Corner to read Complete Drug Facts and Information
Lot: Exp:
*This product is not manufactured or distributed by Reckitt Benckiser Inc. distributor of Mucinex® FAST-MAX™ DM MAX
If for Any reason you are not satisfied with this product, lease return it to the store where purchased for a full refund.
DISTRIBUTED BY
CHAIN DRUG CONSORTIUM
3301 NW BOCA RATON BLVD
SUITE 101, BOCA RATON, FL 33431
LF-004


res
-
INGREDIENTS AND APPEARANCE
SEVERE CONGESTION AND COUGH MAX MAXIUM STRENGTH
dextromethorphan hydrobromide, guaifenesin, phenylephrine hydrochloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68016-221 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 20 mg in 20 mL GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 400 mg in 20 mL PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 10 mg in 20 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) EDETATE DISODIUM (UNII: 7FLD91C86K) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) PROPYL GALLATE (UNII: 8D4SNN7V92) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SORBITOL (UNII: 506T60A25R) SUCRALOSE (UNII: 96K6UQ3ZD4) XANTHAN GUM (UNII: TTV12P4NEE) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68016-221-00 177 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/01/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 01/01/2013 Labeler - Chain Drug Consortium, LLC (101668460) Registrant - AptaPharma Inc. (790523323) Establishment Name Address ID/FEI Business Operations AptaPharma Inc. 790523323 manufacture(68016-221)