Label: FOLIVANE-F- ferrous fumarate, iron, folic acid, ascorbic acid, and niacin capsule
- NHRIC Code(s): 13811-538-90
- Packager: Trigen Laboratories, LLC
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated May 30, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
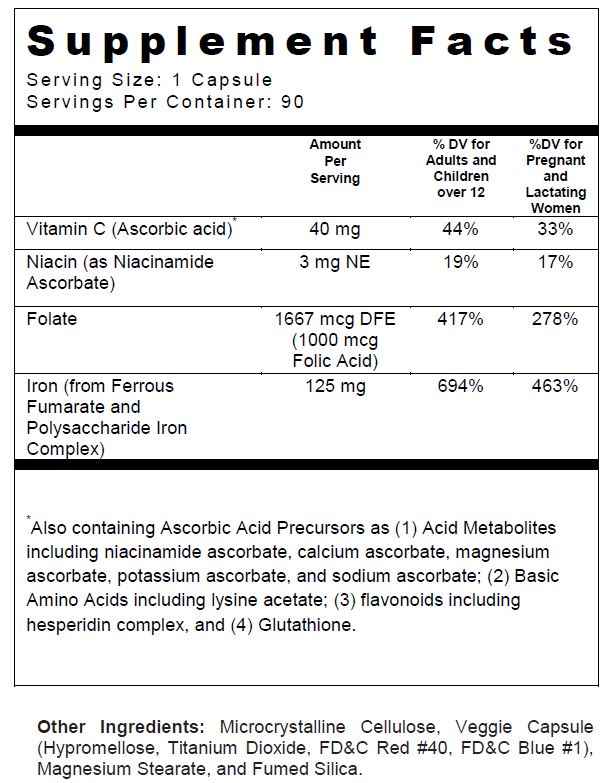
- SUPPLEMENT FACTS
-
CONTRAINDICATIONS
Folivane™-F is contraindicated in patients with a known hypersensitivity to any of the ingredients, also, all iron compounds are contraindicated in patients with hemosiderosis, hemochromatosis, or hemolytic anemias. Pernicious anemia is a contraindication, as folic acid may obscure its signs and symptoms.
- WARNINGS
-
PRECAUTIONS
General: Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where B12 is deficient. Anemia requires appropriate investigation to determine its cause or causes. Periodic clinical and laboratory studies are considered essential. Blood tests including hemoglobin and hematocrit should be done to determine the adequacy of therapy. Folic acid should be used with care in the presence of peptic ulcer disease, regional enteritis, and ulcerative colitis. In doses above 0.1 mg daily, folic acid may obscure the diagnosis of pernicious anemia.
USAGE IN PREGNANCY
Before Folivane™-F is prescribed for megaloblastic anemia in pregnancy, appropriate diagnostic exclusion of Addisonian pernicious anemia (due to faulty or blocked absorption of vitamin B12, or extrinsic factor or either a genetic, immunological or surgical basis) should be carried out.Pediatric Use: Safety and effectiveness of this product have not been established in pediatric patients.
Geriatric Use: Safety and effectiveness of this product have not been established in elderly patients.
-
ADVERSE REACTIONS
Folic Acid: Allergic sensitizations have been reported following both oral and parenteral administration of folic acid.
Ferrous Fumarate: Gastrointestinal disturbances (anorexia, nausea, diarrhea, constipation, heartburn, and vomiting) occur occasionally, but are usually mild and may subside with continuation of therapy. Reducing the dose and administering it with meals will minimize these effects in the sensitive patient. Iron may turn stools black. This is a harmless effect that is a result of unabsorbed iron. Although the absorption of iron is best when taken between meals, giving Folivane™-F after meals may diminish occasional G.I disturbances. Folivane™-F is best absorbed when taken at bedtime.
-
OVERDOSAGE
Acute overdosage of iron may cause abdominal pain, nausea and vomiting and, in severe cases, cardiovascular collapse and death. Other more chronic symptoms include pallor and cyanosis, melena, shock, drowsiness, and coma. The estimated overdose of orally ingested iron is 300 mg/kg body weight. Toxic effects are seen at 10-20 mg/kg elemental iron. When overdoses are ingested by children, severe reactions, including fatalities, have resulted. Folivane™-F should be stored beyond the reach of children to prevent against accidental iron poisoning.
- DESCRIPTION
- DIRECTIONS FOR USE
- HOW SUPPLIED
- STORAGE
-
HEALTH CLAIM
KEEP OUT OF REACH OF CHILDREN.
For use on the order of a healthcare practitioner.
Call your doctor about side effects. To report side effects, call Trigen Laboratories at 1-800-444-5164 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
PLR-FOLF-00001-2 Rev. 05/2023
Manufactured for:
Trigen Laboratories, LLC
Alpharetta, GA 30005
1-770-509-4500

- PRINCIPAL DISPLAY PANEL - 90 Capsule Bottle Label
-
INGREDIENTS AND APPEARANCE
FOLIVANE-F
ferrous fumarate, iron, folic acid, ascorbic acid, and niacin capsuleProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:13811-538 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FERROUS FUMARATE (UNII: R5L488RY0Q) (FERROUS CATION - UNII:GW89581OWR) FERROUS CATION 62.5 mg IRON (UNII: E1UOL152H7) (IRON - UNII:E1UOL152H7) IRON 62.5 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 1 mg ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 40 mg NIACIN (UNII: 2679MF687A) (NIACIN - UNII:2679MF687A) NIACIN 3 mg Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:13811-538-90 90 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 01/01/2010 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color imprint scoring 1 shape size (solid drugs) 20 mm Labeler - Trigen Laboratories, LLC (830479668)