Label: PYRANTEL PAMOATE suspension
- NDC Code(s): 58988-1770-1
- Packager: ProMex LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 25, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
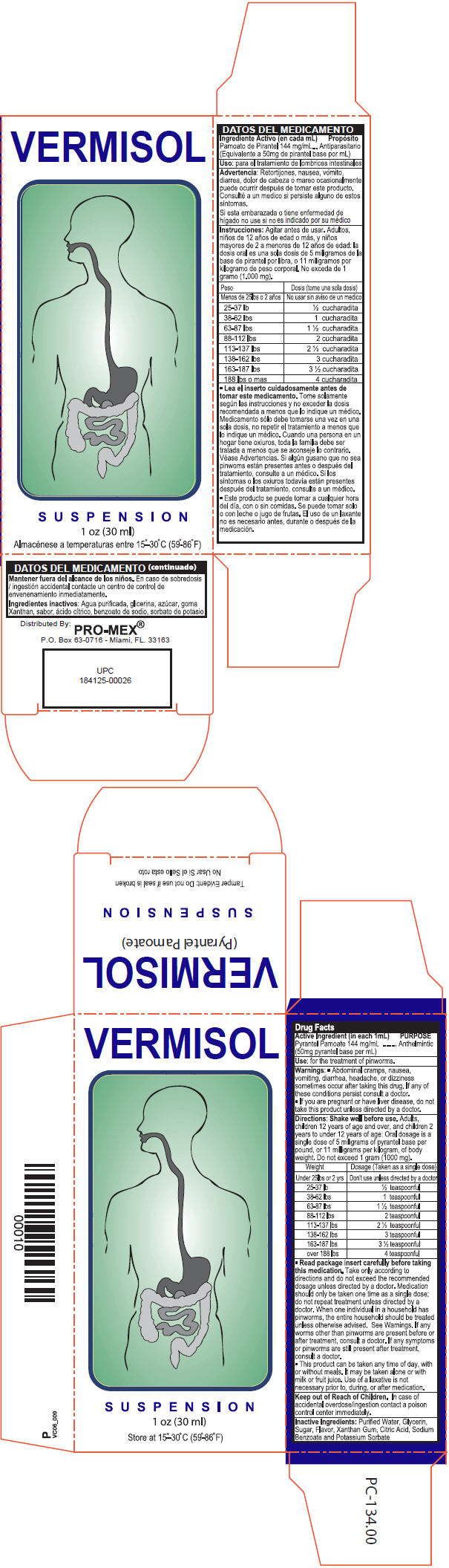
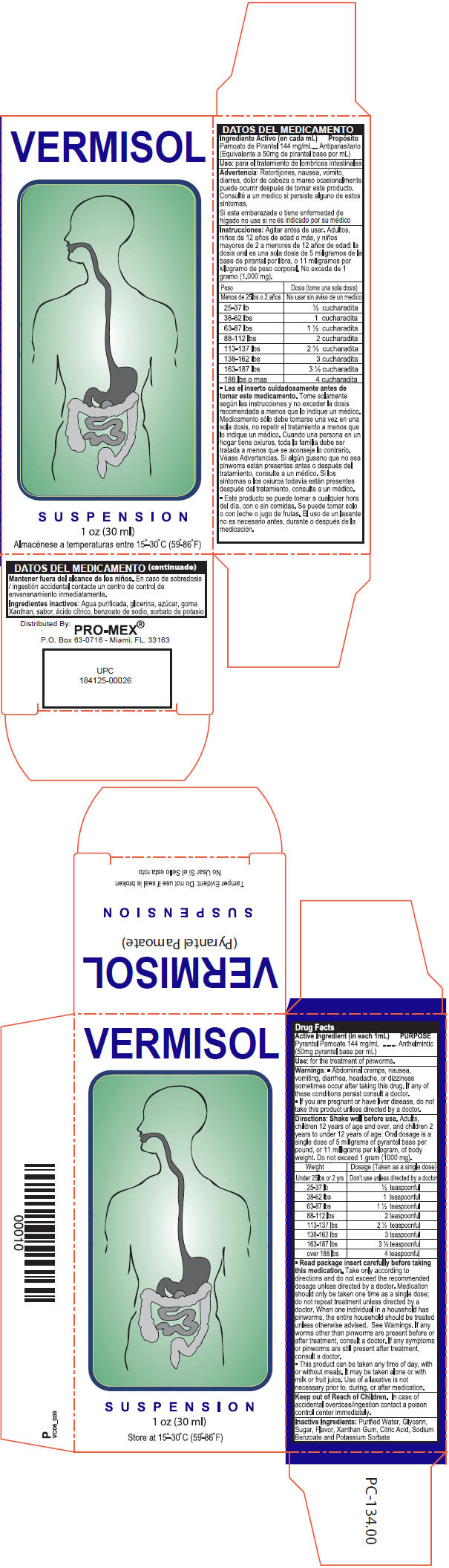
- Active Ingredient (in each 1mL)
- Purpose
- Use
- Warnings
-
Directions
Shake well before use. Adults, children 12 years of age and over, and children 2 years to under 12 years of age: Oral dosage is a single dose of 5 milligrams of pyrantel base per pound, or 11 milligrams per kilogram, of body weight. Do not exceed 1 gram (1000 mg).
Weight Dosage (Taken as a single dose) Under 25 lbs or 2 yrs. Don't use unless directed by a doctor 25-37 lbs ½ teaspoonful 38-62 lbs 1 teaspoonful 63-87 lbs 1 ½ teaspoonful 88-112 lbs 2 teaspoonful 113-137 lbs 2 ½ teaspoonful 138-162 lbs 3 teaspoonful 163-187lbs 3 ½ teaspoonful over 188 lbs 4 teaspoonful *Read package insert carefully before taking this medication. Take only according to directions and do not exceed the recommended dosage unless directed by a doctor. Medication should only be taken one time as a single dose; do not repeat treatment unless directed by a doctor. When one individual in a household has pinworms, the entire household should be treated unless otherwise advised. See Warnings. If any worms other than pinworms are present before or after treatment consult a doctor. If any symptoms or pinworms are still present after treatment, consult a doctor.
*This product can be taken any time of day, with or without meals, it may be taken alone or with milk or fruit juice. Use of a laxative is not necessary prior to, during or after medication.
- KEEP OUT OF REACH OF CHILDREN
- Inactive Ingredients
- PRINCIPAL DISPLAY PANEL - 30 ml Bottle Carton
-
INGREDIENTS AND APPEARANCE
PYRANTEL PAMOATE
pyrantel pamoate suspensionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58988-1770 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Pyrantel Pamoate (UNII: 81BK194Z5M) (Pyrantel - UNII:4QIH0N49E7) Pyrantel 144 mg in 1 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Citric Acid Monohydrate (UNII: 2968PHW8QP) Glycerin (UNII: PDC6A3C0OX) Sucrose (UNII: C151H8M554) Xanthan Gum (UNII: TTV12P4NEE) Sodium Benzoate (UNII: OJ245FE5EU) Potassium Sorbate (UNII: 1VPU26JZZ4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58988-1770-1 1 in 1 CARTON 09/27/2002 1 30 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part357B 09/27/2002 Labeler - ProMex LLC (789974388)