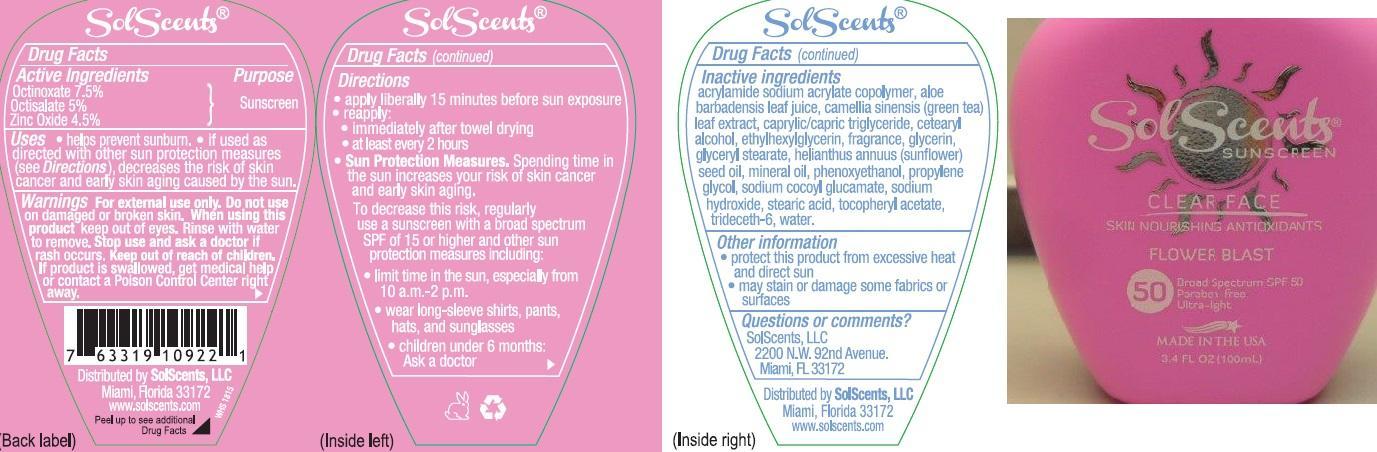
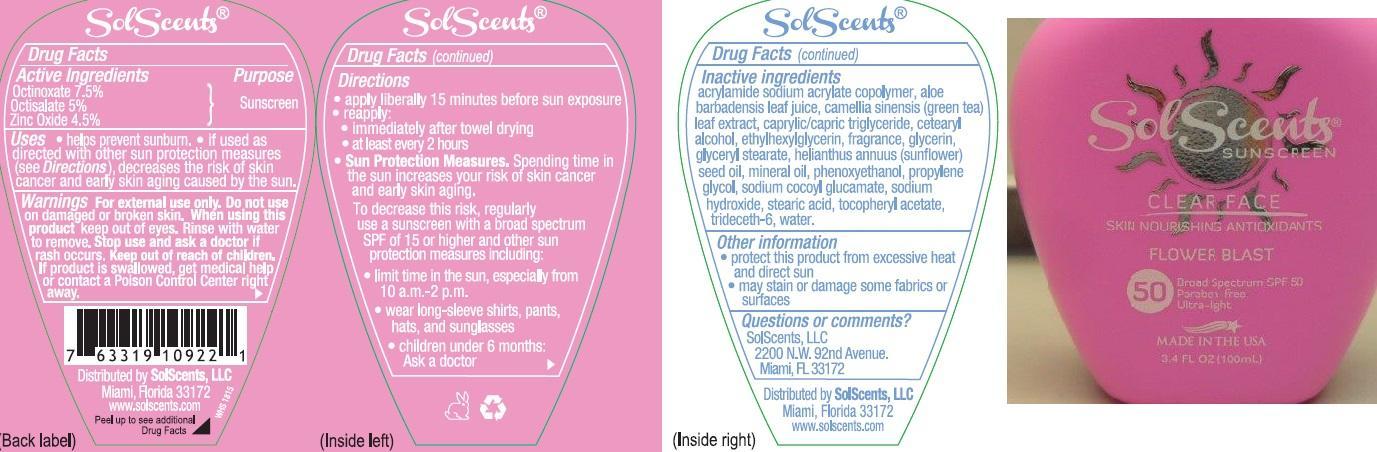
Label: SOLSCENTS CLEAR FACE FLOWER BLAST BROAD SPECTRUM SPF 50- octinoxate, octisalate, and zinc oxide lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 58443-0175-3 - Packager: Prime Enterprises, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 17, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Uses
- Warnings
-
Directions:
- apply liberally 15 minutes before sun exposure
- reapply:
- immediately after towel drying
- at least every 2 hours
- Sun Protection Measures: Spending time in the sun increases your risk of skin cancer & early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher & other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long sleeve shirts, pants, hats, and sunglasses
- Children under 6 months: Ask a doctor
-
Inactive Ingredients
acrylamide sodium acrylate copolymer, aloe barbadensis leaf juice, camellia sinensis (green tea) leaf extract, caprylic/capric tiglyceride, cetearyl alcohol, ethylhexylglycerin, fragrance, glycerin, glyceryl stearate, helianthus annuus (sunflower) seed oil, mineral oil, phenoxyethanol, propylene glycol, sodium cocoyl glucamate, sodium hydroxide, stearic acid, tocopheryl acetate, trideceth-6, water.
- Other information
- Questions or comments?
- PRINCIPAL DISPLAY PANEL - 100 mL Bottle Label
-
INGREDIENTS AND APPEARANCE
SOLSCENTS CLEAR FACE FLOWER BLAST BROAD SPECTRUM SPF 50
octinoxate, octisalate, and zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58443-0175 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 76.5 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 51 mg in 1 mL ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 45.9 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM HYDROXIDE (UNII: 55X04QC32I) STEARIC ACID (UNII: 4ELV7Z65AP) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) TRIDECETH-6 (UNII: 3T5PCR2H0C) WATER (UNII: 059QF0KO0R) CAPRYLIC/CAPRIC MONO/DIGLYCERIDES (UNII: U72Q2I8C85) SODIUM ACRYLOYLDIMETHYLTAURATE-ACRYLAMIDE COPOLYMER (1:1; 90000-150000 MPA.S) (UNII: 5F4963KLHS) ALOE VERA LEAF (UNII: ZY81Z83H0X) GREEN TEA LEAF (UNII: W2ZU1RY8B0) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) SUNFLOWER SEED OIL GLYCERETH-8 ESTERS (UNII: 358X17CAT0) MINERAL OIL (UNII: T5L8T28FGP) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM COCOYL GLUTAMATE (UNII: BMT4RCZ3HG) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58443-0175-3 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 04/01/2015 Labeler - Prime Enterprises, Inc. (101946028) Registrant - Prime Enterprises, Inc. (101946028) Establishment Name Address ID/FEI Business Operations Prime Enterprises, Inc. 101946028 label(58443-0175) , pack(58443-0175) , manufacture(58443-0175) , analysis(58443-0175)