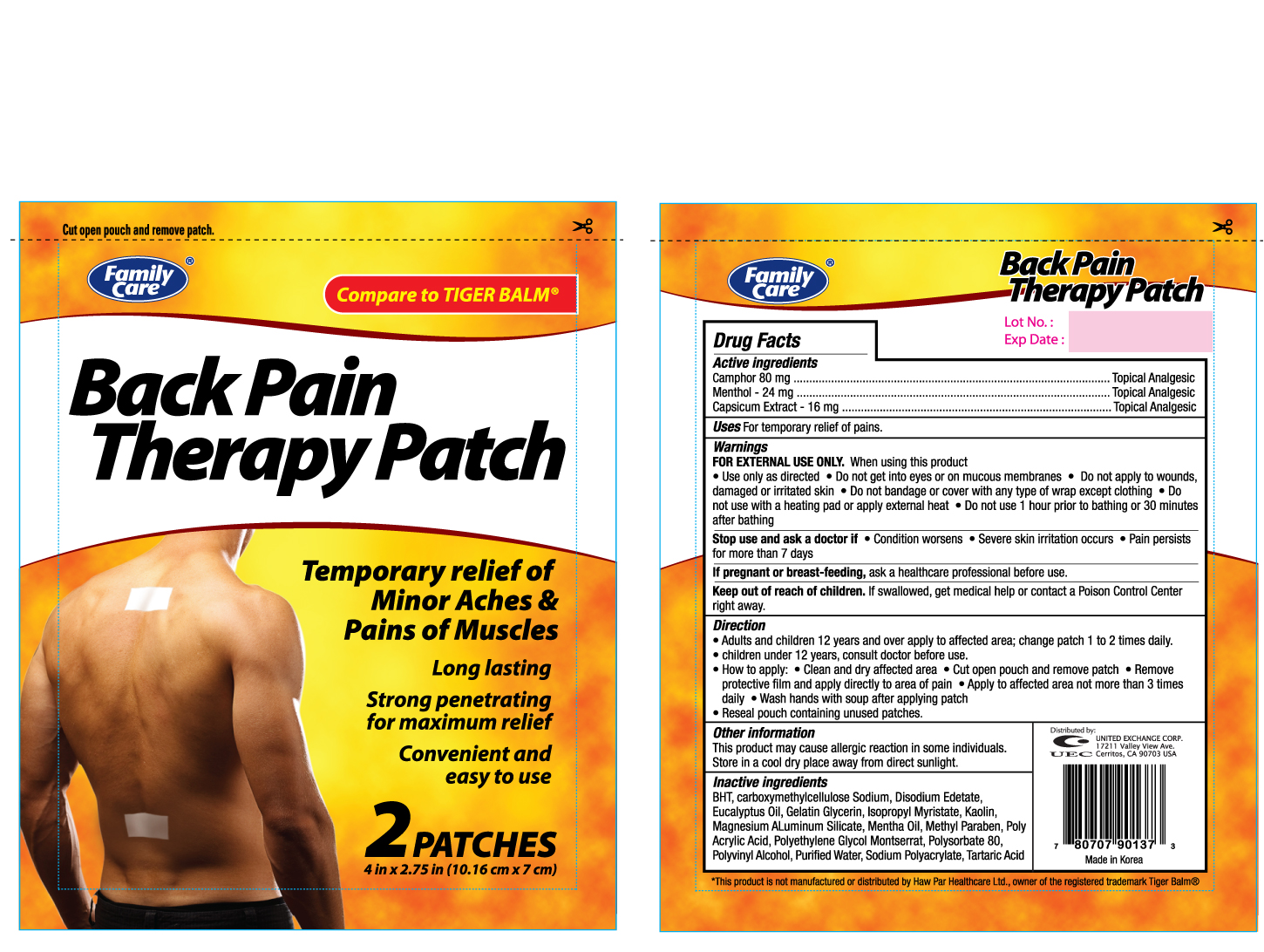
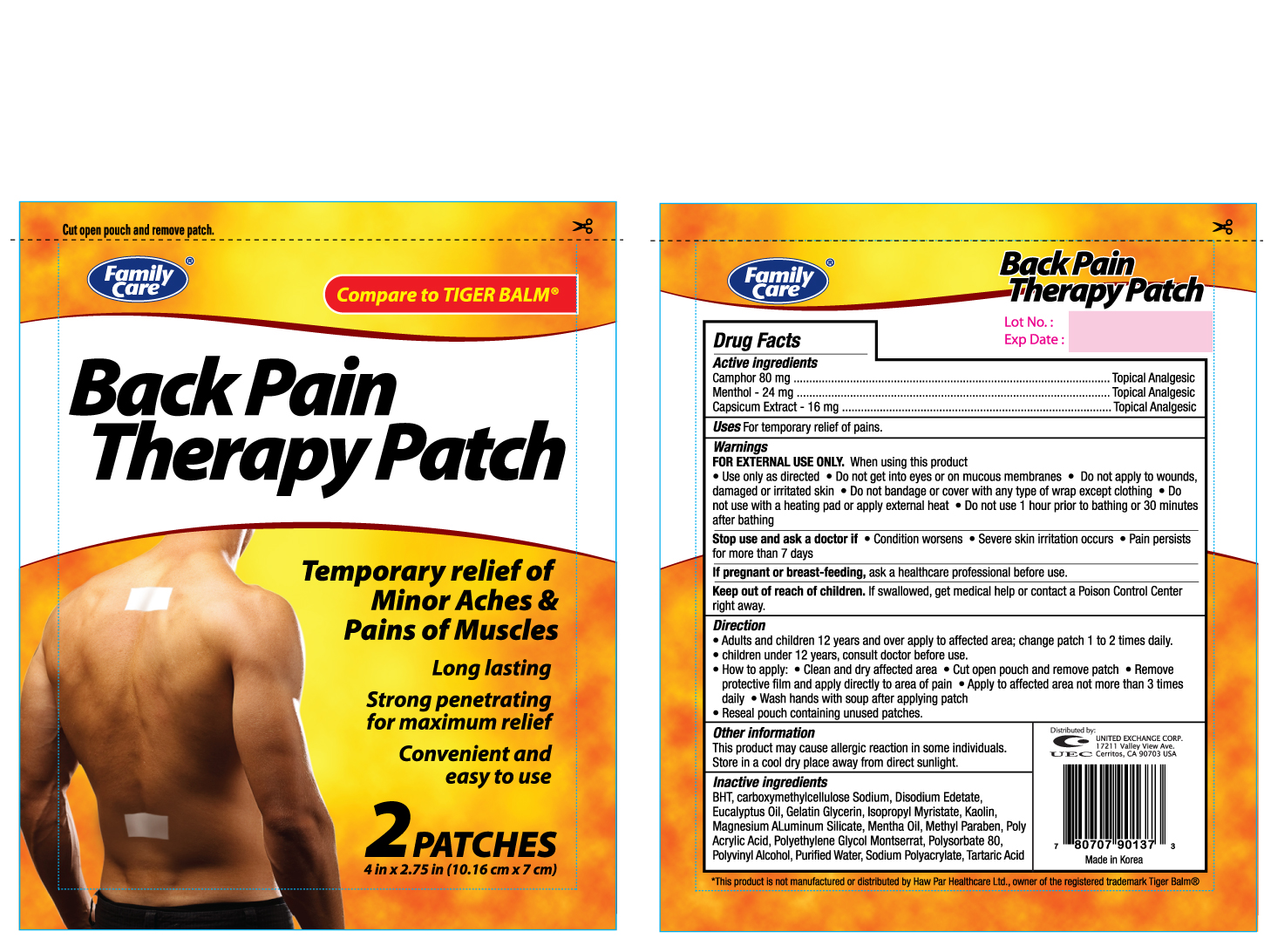
Label: FAMILY CARE BACK PAIN THERAPY (camphor- synthetic, menthol, and capsicum oleoresin patch
-
Contains inactivated NDC Code(s)
NDC Code(s): 65923-901-37 - Packager: UNITED EXCHANGE CORP
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 27, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- WARNINGS
-
WHEN USING
When using this product
- Use only as directed
- Do not get into eyes or on mucous membranes
- Do not apply to wounds, damaged or irritated skin
- Do not bandage or cover with any type of wrap except clothing
- Do not use with a heating pad or apply external heat
- Do not use 1 hour prior to bathing or 30 minutes after bathing
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
Directions
- Adults and children 12 years and over apply to affected area; change patch 1 to 2 times daily
- Children under 12 years, consult doctor before use
- How to apply:
- Clean and dry affected area
- Cut open pouch and remove patch
- Remove protective film and apply directly to area of pain
- Apply to affected area not more than 3 times daily
- Wash hands with soap after applying patch
- Reseal pouch containing unused patches
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FAMILY CARE BACK PAIN THERAPY
camphor (synthetic), menthol, and capsicum oleoresin patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:65923-901 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 80 mg MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 24 mg CAPSICUM OLEORESIN (UNII: UW86K581WY) (CAPSICUM OLEORESIN - UNII:UW86K581WY) CAPSICUM OLEORESIN 16 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) EDETATE DISODIUM (UNII: 7FLD91C86K) EUCALYPTUS OIL (UNII: 2R04ONI662) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) KAOLIN (UNII: 24H4NWX5CO) MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) METHYLPARABEN (UNII: A2I8C7HI9T) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYVINYL ALCOHOL (UNII: 532B59J990) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65923-901-37 1 in 1 BOX 1 2 in 1 PACKET Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 12/27/2011 Labeler - UNITED EXCHANGE CORP (840130579)