Label: ALTREN- altrenogest solution
- NDC Code(s): 51072-108-00
- Packager: Aurora Pharmaceutical, Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: Export only
Drug Label Information
Updated January 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DOSAGE FORMS & STRENGTHS
- FOR VETERINARY USE ONLY
-
DESCRIPTION:
Altren® (altrenogest) Solution 0.22% contains the active synthetic progestin, altrenogest. The chemical name is 17α-allyl-17β-hydroxyestra-4,9,11-trien-3-one. The CAS Registry Number is 850-52-2. The chemical structure is:
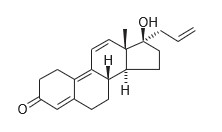
Each mL of Altren® (altrenogest) Solution 0.22% contains 2.2 mg of altrenogest in an oil solution.
- THERAPEUTIC CLASSIFICATION:
-
INDICATIONS:
Horses: Altren® Solution 0.22% is indicated for suppression of estrus. Suppression of estrus allows for a predictable occurrence of estrus following drug withdrawal. This facilitates the attainment of regular cyclicity during the transition from winter anestrus to the physiological breeding season. Suppression of estrus will also facilitate management of prolonged estrus conditions. Suppression of estrus may be used to facilitate scheduled breeding during the physiological breeding season.
Swine: Altren® Solution 0.22% is indicated for suppression of estrus in sexually mature gilts.
Altren® Solution 0.22% mimics the action of progesterone and blocks the cyclical release of gonadotrophins and thus the development of estrus. Following cessation of
treatment, pituitary inhibition ceases and normal development and maturation of the follicles returns, leading to a return to estrus and ovulation. The predictable return to estrus (generally 5-7 days following cessation of treatment) allows for estrus synchronization in a pool of cycling gilts.Altren® Solution 0.22% is indicated for suppression of estrus in sexually mature gilts to:
1. Permit synchronization of estrus in cycling gilts.
2. Improve breeding unit organization by facilitating batch management (all in, all out system) of the breeding gilts.
3. Reduce the pre-breeding gilt pool size. -
DOSAGE AND ADMINISTRATION:
Horses: Administer orally at the rate of 1 ml per 50kg (110 lbs) body weight (0.044mg/kg) once daily for 15 consecutive days. Administer orally by dose syringe or suitable plastic syringe on to the posterior-dorsal surface of the tongue. The recommended dose may alternatively be added to the usual ration.
Dosage Chart:
Approximate Weight Dose in Pounds in mL 770 7 880 8 990 9 1100 10 1210 11 1320 12 -
SPL UNCLASSIFIED SECTION
Extensive clinical trials have demonstrated that estrus will be suppressed in approximately 95% of the mares within three days; however, the post-treatment response depends on the level of ovarian activity when treatment was initiated. Estrus in mares exhibiting regular estrus cycles during the breeding season will be suppressed during treatment; these mares return to estrus four or five days following treatment and continue to cycle normal estrus following withdrawal.
Response in mares in the transition phase between winter anestrus and the summer breeding season depends on the degree of follicular activity. Mares with inactive ovaries and small follicles failed to respond with normal cycles post-treatment.
- DOSAGE & ADMINISTRATION
-
SPECIFIC USES FOR ALTREN® SOLUTION 0.22%:
Horses: Suppression of estrus to:
1. Facilitate attainment of regular cycles during the transition period from winter anestrus to the physiological breeding season.
To facilitate the attainment of regular cycles during the transition phase, mares should be examined to determine the degree of ovarian activity. Estrus in mares with inactive ovaries (no follicles greater than 20 mm in diameter) will be suppressed but these mares may not begin regular cycles following treatment. However, mares with active ovaries (follicles greater than 20 mm in diameter) frequently respond with regular post-treatment estrus cycles.
2. Facilitate management of the mares exhibiting prolonged estrus during the transition period. Estrus will be suppressed in mares exhibiting prolonged behavioural estrus either early or later during the transition period. Again, the post-treatment response depends upon the level of ovarian activity. The mares with greater ovarian activity initiate regular cycles and conceive sooner than the inactive mares. Altren® Solution 0.22% may be administered early in the transition period to suppress estrus in mares with inactive ovaries to aid in the management of these mares or to mares later in the transition period with active ovaries to prepare and schedule the mare for breeding.
3. Permit scheduled breeding of mares during the physiological breeding season. To permit scheduled breeding, mares which are regularly cycling or which have active ovarian function should be given Altren® Solution 0.22% daily for 15 consecutive days, beginning 20 days before the date of the planned estrus. Ovulation will occur 5 to 7 days following the onset of estrus as expected for non- treated mares. Breeding should follow usual procedures for mares in estrus. Mares may be regulated and scheduled either individually or in groups. -
CONTRAINDICATIONS:
Horses: Altren® Solution 0.22% is contraindicated in pregnant mares and in mares having a previous history of uterine inflammation (i.e. a Grade II or greater uterine biopsy result), or current uterine inflammations (i.e. acute, subacute or chronic endometritis). Altren® Solution 0.22% therapy may exacerbate an existing uterine inflammation into a fulminating uterine inflammation. The safety of this drug in pregnant mares has not been established. Various synthetic progestins, including altrenogest, when administered to rats during the embryonic stage of pregnancy at doses many fold greater than the recommended equine dose caused foetal anomalies, specifically of the female genitalia.
Swine: Altren® Solution 0.22% is contraindicated in gestating gilts or sows and is not to be administered to breeding boars since it could interfere with their fertility.
- CAUTIONS:
-
WARNINGS:
Horses: This drug is not to be administered to horses that are intended to be slaughtered for use in food.
Swine: Treated animals must not be slaughtered for use in food for at least 42 days after the latest treatment with this drug.The following individuals should not handle this product (based upon the known effects of progestins used in humans on a chronic basis):
- Pregnant women or women who suspect they are pregnant.
- Women with known or suspected breast, uterine, or endometrial hormone-dependent cancer.
- Women with undiagnosed vaginal bleeding.
- Persons with thrombophlebitis or thromboembolic disorders or with a history of these events.
- Persons with cerebral-vascular or coronary-artery disease.
- Persons with known or suspected estrogen-dependent neoplasia.
- Persons with benign or malignant tumors which developed during the use of oral contraceptives or other estrogen-containing products.
- Persons with liver dysfunction or disease.
Altrenogest is readily absorbed by the skin. Avoid contact with skin or eyes and oral exposure. When handling this product, vinyl, neoprene or nitrile protective gloves must be worn.Women of childbearing age should exercise extreme caution when handling this product. Accidental absorption could lead to a disruption of the menstrual cycle.
The following measures are recommended in case of accidental exposure:
- Skin Exposure: Wash immediately with soap and water.
- Eye Exposure: Immediately flush with plenty of water. Get medical attention.
- If Swallowed: Do not induce vomiting. The product contains an oil. Call a physician. Vomiting should be supervised by a physician because of possible pulmonary damage via aspiration of the oil base. Show the product label to the physician.
Effects of overexposure: There has been no human use of this product. The information in this section is extrapolated from data available on other products of the same pharmacological class that have been used in humans. Effects anticipated are due to the progestational activity of altrenogest. Acute effects after a single exposure are possible; however, continued daily exposure has the potential for more untoward effects such as disruption of the menstrual cycle, uterine or abdominal cramping, increased or decreased uterine bleeding, prolongation of pregnancy and headaches. The oil base may also cause complications if swallowed.
Keep out of reach of children.
- STORAGE:
- SUPPLY:
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALTREN
altrenogest solutionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:51072-108 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALTRENOGEST (UNII: 2U0X0JA2NB) (ALTRENOGEST - UNII:2U0X0JA2NB) ALTRENOGEST 2.2 mg in 1 mL Product Characteristics Color yellow (colorless to yellow) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51072-108-00 1000 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date export only 02/17/2022 Labeler - Aurora Pharmaceutical, Inc. (832848639) Establishment Name Address ID/FEI Business Operations Aurora Pharmaceutical, Inc. 832848639 manufacture


