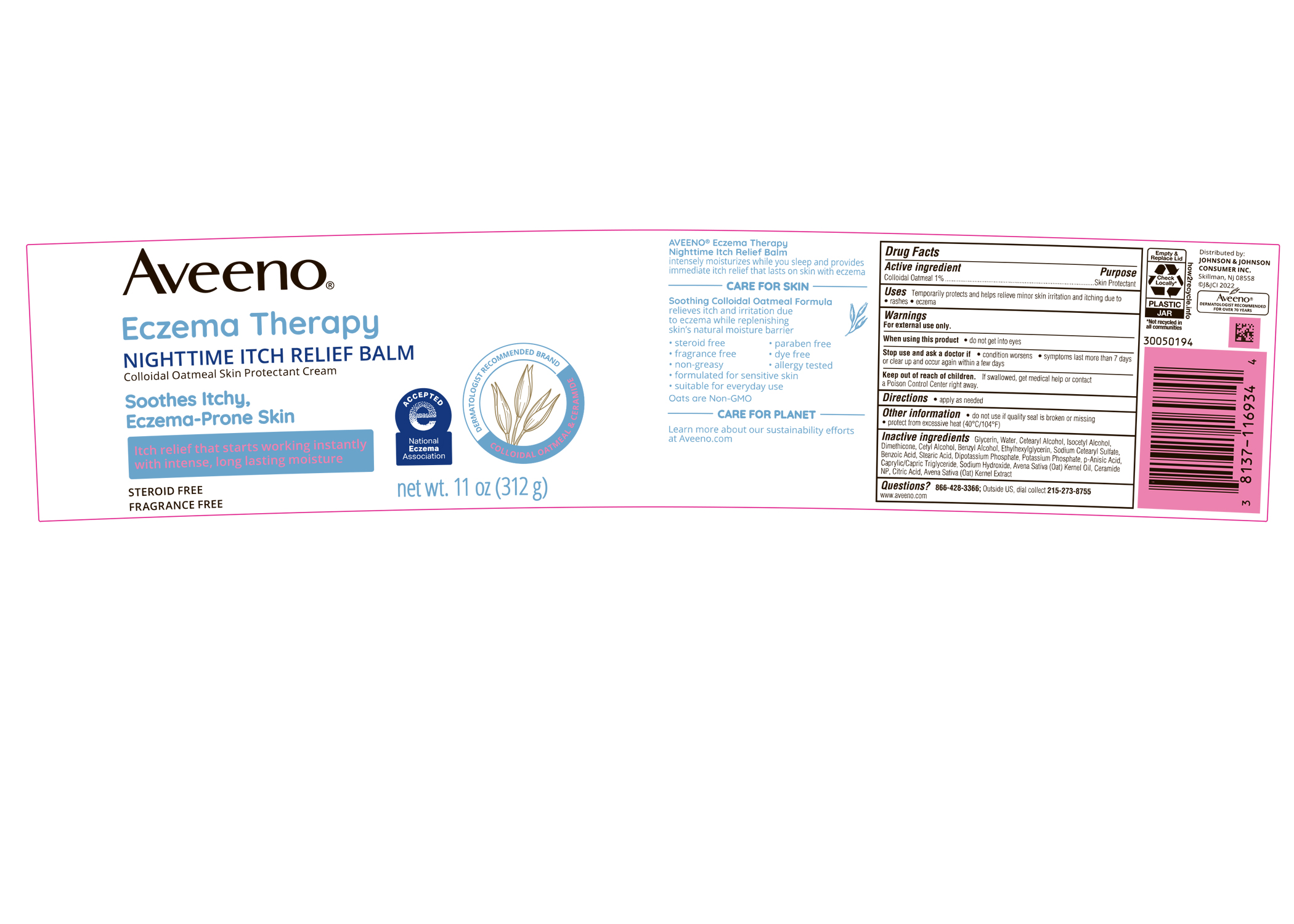
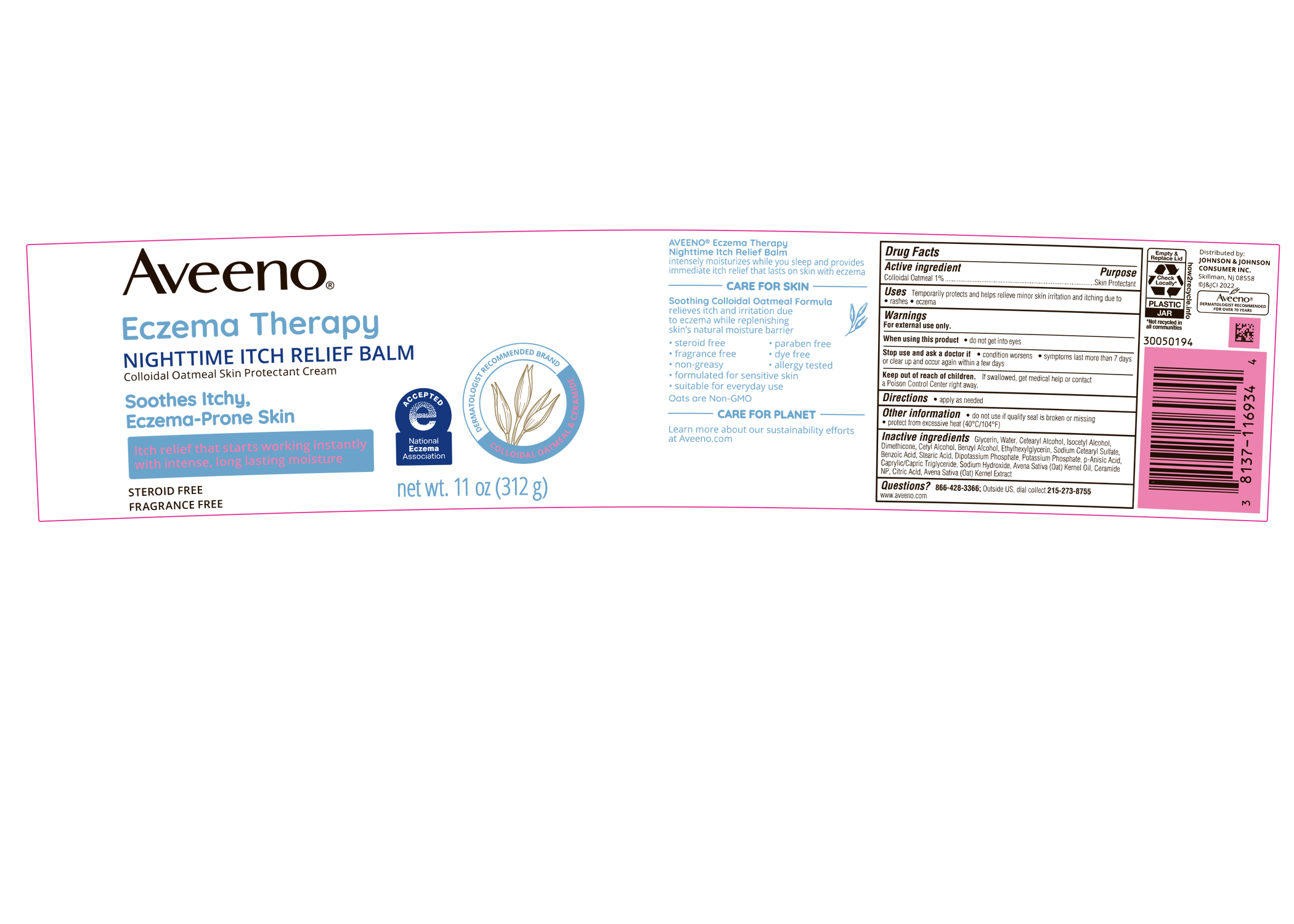
Label: AVEENO ECZEMA THERAPY NIGHTTIME ITCH RELIEF BALM- colloidal oatmeal cream
- NDC Code(s): 69968-0772-1, 69968-0772-2
- Packager: Johnson & Johnson Consumer Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 9, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Uses
- Warnings
- Directions
- Other Information
-
Inactive Ingredients
Glycerin, Water, Cetearyl Alcohol, Isocetyl Alcohol, Dimethicone, Cetyl Alcohol, Benzyl Alcohol, Ethylhexylglycerin, Sodium Cetearyl Sulfate, Benzoic Acid, Stearic Acid, Dipotassium Phosphate, Potassium Phosphate, p-Anisic Acid, Caprylic/Capric Triglyceride, Sodium Hydroxide, Avena Sativa (Oat) Kernel Oil, Ceramide NP, Citric Acid, Avena Sativa (Oat) Kernel Extract
- Questions?
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 312 g Jar
Aveeno®
Eczema Therapy
NIGHTTIME ITCH RELIEF BALM
Colloidal Oatmeal Skin Protectant Cream
Soothes Itchy,
Eczema-Prone Skin
Itch relief that starts working instantly
with intense, long lasting moisture
STEROID FREE
FRAGRANCE FREE
ACCEPTED
e
National
Eczema
Association
DERMATOLOGIST RECOMMENDED BRAND
COLLOIDAL OATMEAL & CERAMIDE
net wt. 11 oz (312 g)
-
INGREDIENTS AND APPEARANCE
AVEENO ECZEMA THERAPY NIGHTTIME ITCH RELIEF BALM
colloidal oatmeal creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69968-0772 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OATMEAL (UNII: 8PI54V663Y) (OATMEAL - UNII:8PI54V663Y) OATMEAL 10 mg in 1 g Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) ISOCETYL ALCOHOL (UNII: 1800H64066) BENZYL ALCOHOL (UNII: LKG8494WBH) SODIUM CETOSTEARYL SULFATE (UNII: 7ZBS06BH4B) BENZOIC ACID (UNII: 8SKN0B0MIM) DIBASIC POTASSIUM PHOSPHATE (UNII: CI71S98N1Z) POTASSIUM PHOSPHATE, UNSPECIFIED FORM (UNII: B7862WZ632) P-ANISIC ACID (UNII: 4SB6Y7DMM3) SODIUM HYDROXIDE (UNII: 55X04QC32I) DIMETHICONE (UNII: 92RU3N3Y1O) CETYL ALCOHOL (UNII: 936JST6JCN) STEARIC ACID (UNII: 4ELV7Z65AP) OAT (UNII: Z6J799EAJK) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) CERAMIDE NP (UNII: 4370DF050B) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) OAT KERNEL OIL (UNII: 3UVP41R77R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69968-0772-2 12 in 1 PACKET 08/01/2022 1 28 g in 1 JAR; Type 0: Not a Combination Product 2 NDC:69968-0772-1 312 g in 1 JAR; Type 0: Not a Combination Product 08/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final M016 08/01/2022 Labeler - Johnson & Johnson Consumer Inc. (118772437)