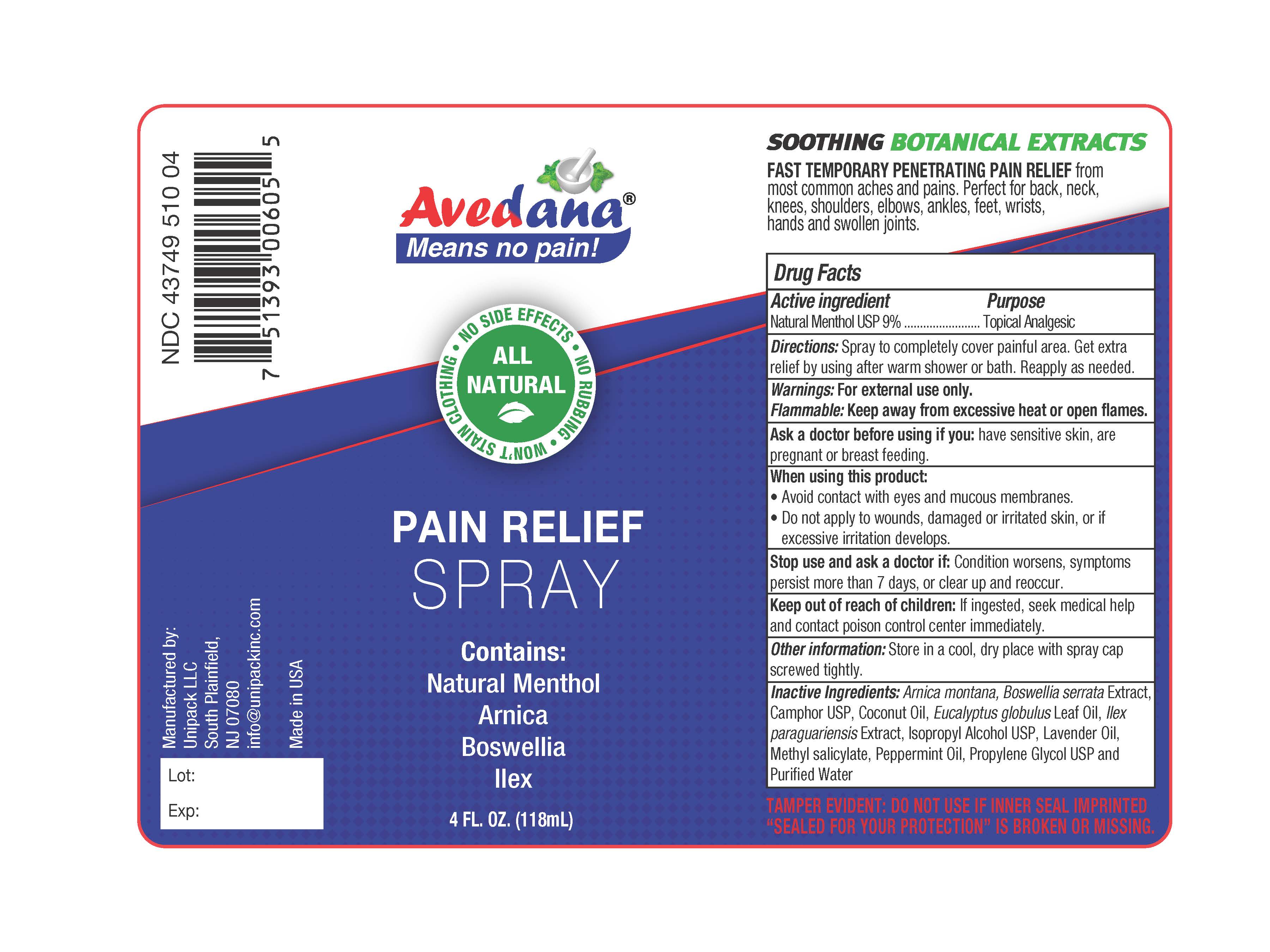
Label: AVEDANA PAIN RELIEF- menthol spray
- NDC Code(s): 43749-510-04
- Packager: Unipack LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 4, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Directions
- Warnings
- Flammable
- Ask a doctor before using if you
- When using this product:
- Stop use and ask a doctor if:
- Keep out of reach of children:
- Other information
- Inactive Ingredients:
- Directions
- Avedana ® Pain Relieving Spray
-
INGREDIENTS AND APPEARANCE
AVEDANA PAIN RELIEF
menthol sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:43749-510 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 9 g in 100 mL Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 1 (UNII: H3R47K3TBD) ISOPROPYL ALCOHOL (UNII: ND2M416302) PEPPERMINT OIL (UNII: AV092KU4JH) COCONUT OIL (UNII: Q9L0O73W7L) METHYL SALICYLATE (UNII: LAV5U5022Y) ARNICA MONTANA (UNII: O80TY208ZW) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) ILEX PARAGUARIENSIS LEAF (UNII: 1Q953B4O4F) LAVENDER OIL (UNII: ZBP1YXW0H8) BOSWELLIA SERRATA RESIN OIL (UNII: 5T1XCE6K8K) WATER (UNII: 059QF0KO0R) CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) EUCALYPTUS OIL (UNII: 2R04ONI662) Product Characteristics Color white (Clear) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:43749-510-04 118 mL in 1 BOTTLE, WITH APPLICATOR; Type 0: Not a Combination Product 09/17/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 09/29/2014 Labeler - Unipack LLC (009248480) Registrant - Unipack LLC (116015769) Establishment Name Address ID/FEI Business Operations Unipack LLC 009248480 manufacture(43749-510)