Label: DICYCLOMINE HYDROCHLORIDE injection
- NDC Code(s): 70436-147-81
- Packager: Slate Run Pharmaceuticals, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 24, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use DICYCLOMINE HYDROCHLORIDE INJECTION safely and effectively. See full prescribing information for DICYCLOMINE HYDROCHLORIDE INJECTION.
DICYCLOMINE HYDROCHLORIDE injection, USP for intramuscular use
Initial U.S. Approval: 1950INDICATIONS AND USAGE
Dicyclomine Hydrochloride is an antispasmodic and anticholinergic (antimuscarinic) agent indicated for the treatment of functional bowel/irritable bowel syndrome (1)
DOSAGE AND ADMINISTRATION
Dosage for dicyclomine hydrochloride must be adjusted to individual patient needs (2).
If a dose is missed, patients should continue the normal dosing schedule (2).
Intramuscular in adults (2.2):
- •
- Intramuscular administration recommended no longer than 1 or 2 days when patients cannot take oral administration
- •
- Recommended dose: 10 mg to 20 mg four times a day
DOSAGE FORMS AND STRENGTHS
- •
- Dicyclomine Hydrochloride Injection, USP 20 mg/2 mL (10 mg/mL) (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- •
- For intramuscular injection only; should not be administered by any other route. Intravenous injection may result in thrombosis or thrombophlebitis and injection site reactions (5.1)
- •
- Cardiovascular conditions: worsening of conditions (5.2)
- •
- Peripheral and central nervous system: heat prostration can occur with drug use (fever and heat stroke due to decreased sweating); drug should be discontinued and supportive measures instituted (5.3)
- •
- Psychosis and delirium have been reported in patients sensitive to anticholinergic drugs (such as elderly patients and/or in patients with mental illness): signs and symptoms resolve within 12 to 24 hours after discontinuation of dicyclomine hydrochloride (5.3)
- •
- Myasthenia Gravis: overdose may lead to muscular weakness and paralysis. Dicyclomine hydrochloride should be given to patients with myasthenia gravis only to reduce adverse muscarinic effects of an anticholinesterase (5.4)
- •
- Incomplete intestinal obstruction: diarrhea may be an early symptom especially in patients with ileostomy or colostomy. Treatment with dicyclomine hydrochloride would be inappropriate and possibly fatal (5.5)
- •
- Salmonella dysenteric patients: due to risk of toxic megacolon (5.6)
- •
- Ulcerative colitis: Dicyclomine hydrochloride should be used with caution in these patients; large doses may suppress intestinal motility or aggravate the serious complications of toxic megacolon (5.7)
- •
- Prostatic hypertrophy: Dicyclomine hydrochloride should be used with caution in these patients; may lead to urinary retention (5.8)
- •
- Hepatic and renal disease: should be used with caution (5.9)
- •
- Geriatric: use with caution in elderly who may be more susceptible to dicyclomine hydrochloride’s adverse events (5.10)
ADVERSE REACTIONS
The most serious adverse reactions include cardiovascular and central nervous system symptoms. The most common adverse reactions (> 5% of patients) are dizziness, dry mouth, vision blurred, nausea, somnolence, asthenia and nervousness (6)
To report SUSPECTED ADVERSE REACTIONS, Slate Run Pharmaceuticals, LLC at 1-888-341-9214 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- •
- Antiglaucoma agents: anticholinergics antagonize antiglaucoma agents and may increase intraocular pressure (7)
- •
- Anticholinergic agents: may affect the gastrointestinal absorption of various drugs; may also increase certain actions or side effects of other anticholinergic drugs (7)
- •
- Antacids: interfere with the absorption of anticholinergic agents (7)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 9/2021
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Intramuscular Dosage and Administration in Adults
2.2 Preparation for Intramuscular Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Inadvertent Intravenous Administration
5.2 Cardiovascular Conditions
5.3 Peripheral and Central Nervous System
5.4 Myasthenia Gravis
5.5 Intestinal Obstruction
5.6 Toxic Dilatation of Intestinemegacolon
5.7 Ulcerative Colitis
5.8 Prostatic Hypertrophy
5.9 Hepatic and Renal Disease
5.10 Geriatric Population
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
6.3 Adverse Reactions Reported with Similar Drugs with Anticholinergic/Antispasmodic Action
7 DRUG INTERACTIONS
7.1 Antiglaucoma Agents
7.2 Other Drugs with Anticholinergic Activity
7.3 Other Gastrointestinal Motility Drugs
7.4 Effect of Antacids
7.5 Effect on Absorption of Other Drugs
7.6 Effect on Gastric Acid Secretion
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
17.1 Inadvertent Intravenous Administration
17.2 Use in Infants
17.3 Use in Nursing Mothers
17.4 Peripheral and Central Nervous System
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
Dosage must be adjusted to individual patient needs.
2.1 Intramuscular Dosage and Administration in Adults
Dicyclomine hydrochloride intramuscular injection must be administered via intramuscular route only. Do not administer by any other route.
The recommended intramuscular dose is 10 mg to 20 mg four times a day [see Clinical Pharmacology (12)].
The intramuscular injection is to be used only for 1 or 2 days when the patient cannot take oral medication.
Intramuscular injection is about twice as bioavailable as oral dosage forms.
2.2 Preparation for Intramuscular Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Aspirate the syringe before injecting to avoid intravascular injection, since thrombosis may occur if the drug is inadvertently injected intravascularly.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Dicyclomine hydrochloride is contraindicated in infants less than 6 months of age [see Use in Specific Populations (8.4)], nursing mothers [see Use in Specific Populations (8.3)], and in patients with:
- •
- unstable cardiovascular status in acute hemorrhage
- •
- myasthenia gravis [see Warnings and Precautions (5.4)]
- •
- glaucoma [see Adverse Reactions (6.3) and Drug Interactions (7.1)]
- •
- obstructive uropathy [see Warnings and Precautions (5.8)]
- •
- obstructive disease of the gastrointestinal tract [see Warnings and Precautions (5.5)]
- •
- severe ulcerative colitis [see Warnings and Precautions (5.7)]
- •
- reflux esophagitis
-
5 WARNINGS AND PRECAUTIONS
5.1 Inadvertent Intravenous Administration
Dicyclomine hydrochloride solution is for intramuscular administration only. Do not administer by any other route. Inadvertent intravenous administration may result in thrombosis, thrombophlebitis, and injection site reactions such as pain, edema, skin color change, and reflex sympathetic dystrophy syndrome [see Adverse Reactions (6.2)].
5.2 Cardiovascular Conditions
Dicyclomine hydrochloride needs to be used with caution in conditions characterized by tachyarrhythmia such as thyrotoxicosis, congestive heart failure and in cardiac surgery, where they may further accelerate the heart rate. Investigate any tachycardia before administration of dicyclomine hydrochloride. Care is required in patients with coronary heart disease, as ischemia and infarction may be worsened, and in patients with hypertension [see Adverse Reactions (6.3)].
5.3 Peripheral and Central Nervous System
The peripheral effects of dicyclomine hydrochloride are a consequence of their inhibitory effect on muscarinic receptors of the autonomic nervous system. They include dryness of the mouth with difficulty in swallowing and talking, thirst, reduced bronchial secretions, dilatation of the pupils (mydriasis) with loss of accommodation (cycloplegia) and photophobia, flushing and dryness of the skin, transient bradycardia followed by tachycardia, with palpitations and arrhythmias, and difficulty in micturition, as well as reduction in the tone and motility of the gastrointestinal tract leading to constipation [see Adverse Reactions (6)].
In the presence of high environmental temperature heat prostration can occur with drug use (fever and heat stroke due to decreased sweating). It should also be used cautiously in patients with fever. If symptoms occur, the drug should be discontinued and supportive measures instituted. Because of the inhibitory effect on muscarinic receptors within the autonomic nervous system, caution should be taken in patients with autonomic neuropathy.
Central nervous system (CNS) signs and symptoms include confusional state, disorientation, amnesia, hallucinations, dysarthria, ataxia, coma, euphoria, fatigue, insomnia, agitation and mannerisms, and inappropriate affect.
Psychosis and delirium have been reported in sensitive individuals (such as elderly patients and/or in patients with mental illness) given anticholinergic drugs. These CNS signs and symptoms usually resolve within 12 to 24 hours after discontinuation of the drug.
Dicyclomine hydrochloride may produce drowsiness, dizziness or blurred vision. The patient should be warned not to engage in activities requiring mental alertness, such as operating a motor vehicle or other machinery or performing hazardous work while taking dicyclomine hydrochloride.
5.4 Myasthenia Gravis
With overdosage, a curare-like action may occur (i.e., neuromuscular blockade leading to muscular weakness and possible paralysis). It should not be given to patients with myasthenia gravis except to reduce adverse muscarinic effects of an anticholinesterase [see Contraindications (4)].
5.5 Intestinal Obstruction
Diarrhea may be an early symptom of incomplete intestinal obstruction, especially in patients with ileostomy or colostomy. In this instance, treatment with this drug would be inappropriate and possibly harmful [see Contraindications (4)].
Rarely development of Ogilvie’s syndrome (colonic pseudo-obstruction) has been reported. Ogilvie’s syndrome is a clinical disorder with signs, symptoms, and radiographic appearance of an acute large bowel obstruction but with no evidence of distal colonic obstruction
5.6 Toxic Dilatation of Intestinemegacolon
Toxic dilatation of intestine and intestinal perforation is possible when anticholinergic agents are administered in patients with Salmonella dysentery.
5.7 Ulcerative Colitis
Caution should be taken in patients with ulcerative colitis. Large doses may suppress intestinal motility to the point of producing a paralytic ileus and the use of this drug may precipitate or aggravate the serious complication of toxic megacolon [see Adverse Reactions (6.3)]. Dicyclomine hydrochloride is contraindicated in patients with severe ulcerative colitis [see Contraindications (4)].
5.8 Prostatic Hypertrophy
Dicyclomine hydrochloride should be used with caution in patients with known or suspected prostatic enlargement, in whom prostatic enlargement may lead to urinary retention [see Adverse Reactions (6.3)].
-
6 ADVERSE REACTIONS
The pattern of adverse effects seen with dicyclomine is mostly related to its pharmacological actions at muscarinic receptors [see Clinical Pharmacology (12)]. They are a consequence of the inhibitory effect on muscarinic receptors within the autonomic nervous system. These effects are dose-related and are usually reversible when treatment is discontinued.
The most serious adverse reactions reported with dicyclomine hydrochloride include cardiovascular and central nervous system symptoms [see Warnings and Precautions (5.2, 5.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure in controlled clinical trials involving over 100 patients treated for functional bowel/irritable bowel syndrome with dicyclomine hydrochloride at initial doses of 160 mg daily (40 mg four times a day).
In these trials most of the side effects were typically anticholinergic in nature and were reported by 61% of the patients. Table 1 presents adverse reactions (MedDRA 13.0 preferred terms) by decreasing order of frequency in a side-by-side comparison with placebo.
Table 1: Adverse reactions experienced in controlled clinical trials with decreasing order of frequency
MedDRA Preferred Term
Dicyclomine Hydrochloride
(40 mg four times a day) %
Placebo %
Dry Mouth
33
5
Dizziness
40
5
Vision Blurred
27
2
Nausea
14
6
Somnolence
9
1
Asthenia
7
1
Nervousness
6
2
Nine percent (9%) of patients were discontinued from dicyclomine hydrochloride because of one or more of these side effects (compared with 2% in the placebo group). In 41% of the patients with side effects, side effects disappeared or were tolerated at the 160 mg daily dose without reduction. A dose reduction from 160 mg daily to an average daily dose of 90 mg was required in 46% of the patients with side effects who then continued to experience a favorable clinical response; their side effects either disappeared or were tolerated.
6.2 Postmarketing Experience
The following adverse reactions, presented by system organ class in alphabetical order, have been identified during post approval use of dicyclomine hydrochloride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- •
- Cardiac disorders: palpitations, tachyarrhythmias
- •
- Eye disorders: cycloplegia, mydriasis, vision blurred
- •
- Gastrointestinal disorders: abdominal distension, abdominal pain, constipation, dry mouth, dyspepsia, nausea, vomiting
- •
- General disorders and administration site conditions: fatigue, malaise
- •
- Immune system disorders: drug hypersensitivity including face edema, angioedema, anaphylactic shock
- •
- Nervous system disorders: dizziness, headache, somnolence, syncope
- •
- Psychiatric disorders: As with the other anti-cholinergic drugs, cases of delirium or symptoms of delirium such as amnesia (or transient global amnesia), agitation, confusional state, delusion, disorientation, hallucination (including visual hallucination) as well as mania, mood altered and pseudodementia, have been reported with the use of dicyclomine. Nervousness and insomnia have also been reported.
- •
- Reproductive system and breast disorders: suppressed lactation
- •
- Respiratory, thoracic and mediastinal disorders: dyspnea, nasal congestion
- •
- Skin and subcutaneous tissue disorder: dermatitis allergic, erythema, rash
Cases of thrombosis, thrombophlebitis and injection site reactions such as local pain, edema, skin color change and even reflex sympathetic dystrophy syndrome have been reported following inadvertent IV injection of dicyclomine hydrochloride.
6.3 Adverse Reactions Reported with Similar Drugs with Anticholinergic/Antispasmodic Action
Gastrointestinal: anorexia
Central Nervous System: tingling, numbness, dyskinesia, speech disturbance, insomnia
Peripheral Nervous System: with overdosage, a curare-like action may occur (i.e., neuromuscular blockade leading to muscular weakness and possible paralysis)
Ophthalmologic: diplopia, increased ocular tension
Dermatologic/Allergic: urticaria, itching, and other dermal manifestations
Genitourinary: urinary hesitancy, urinary retention in patients with prostatic hypertrophy
Cardiovascular: hypertension
Respiratory: apnea
Other: decreased sweating, sneezing, throat congestion, impotence. With the injectable form, there may be temporary sensation of light-headedness. Some local irritation and focal coagulation necrosis may occur following the intramuscular injection of dicyclomine hydrochloride.
-
7 DRUG INTERACTIONS
7.1 Antiglaucoma Agents
Anticholinergics antagonize the effects of antiglaucoma agents. Anticholinergic drugs in the presence of increased intraocular pressure may be hazardous when taken concurrently with agents such as corticosteroids. Use of dicyclomine hydrochloride in patients with glaucoma is not recommended [see Contraindications (4)].
7.2 Other Drugs with Anticholinergic Activity
The following agents may increase certain actions or side effects of anticholinergic drugs including dicyclomine hydrochloride: amantadine, antiarrhythmic agents of Class I (e.g., quinidine), antihistamines, antipsychotic agents (e.g., phenothiazines), benzodiazepines, MAO inhibitors, narcotic analgesics (e.g., meperidine), nitrates and nitrites, sympathomimetic agents, tricyclic antidepressants, and other drugs having anticholinergic activity.
7.3 Other Gastrointestinal Motility Drugs
Interaction with other gastrointestinal motility drugs may antagonize the effects of drugs that alter gastrointestinal motility, such as metoclopramide.
7.4 Effect of Antacids
Because antacids may interfere with the absorption of anticholinergic agents including dicyclomine hydrochloride, simultaneous use of these drugs should be avoided.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Adequate and well-controlled studies have not been conducted with dicyclomine hydrochloride in pregnant women at the recommended doses of 80 to 160 mg/day. However, epidemiologic studies did not show an increased risk of structural malformations among babies born to women who took products containing dicyclomine hydrochloride at doses up to 40 mg/day during the first trimester of pregnancy. Reproduction studies have been performed in rats and rabbits at doses up to 33 times the maximum recommended human dose based on 160 mg/day (3 mg/kg) and have revealed no evidence of harm to the fetus due to dicyclomine. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
8.3 Nursing Mothers
Dicyclomine hydrochloride is contraindicated in women who are breastfeeding. Dicyclomine hydrochloride is excreted in human milk. Because of the potential for serious adverse reactions in breast-fed infants from dicyclomine hydrochloride, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother [see Use in Specific Populations (8.4)].
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Dicyclomine hydrochloride is contraindicated in infants less than 6 months of age [see Contraindications (4)]. There are published cases reporting that the administration of dicyclomine hydrochloride to infants has been followed by serious respiratory symptoms (dyspnea, shortness of breath, breathlessness, respiratory collapse, apnea and asphyxia), seizures, syncope, pulse rate fluctuations, muscular hypotonia, and coma, and death, however; no causal relationship has been established.
8.5 Geriatric Use
Clinical studies of dicyclomine hydrochloride did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range in adults, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
8.6 Renal Impairment
Effects of renal impairment on PK, safety and efficacy of dicyclomine hydrochloride have not been studied. Dicyclomine hydrochloride drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Dicyclomine hydrochloride should be administered with caution in patients with renal impairment.
-
10 OVERDOSAGE
In case of an overdose, patients should contact a physician, poison control center (1-800-222-1222), or emergency room.
The signs and symptoms of overdosage include: headache; nausea; vomiting; blurred vision; dilated pupils; hot, dry skin; dizziness; dryness of the mouth; difficulty in swallowing; and CNS stimulation including convulsion. A curare-like action may occur (i.e., neuromuscular blockade leading to muscular weakness and possible paralysis).
One reported event included a 37-year-old who reported numbness on the left side, cold fingertips, blurred vision, abdominal and flank pain, decreased appetite, dry mouth, and nervousness following ingestion of 320 mg daily (four 20 mg tablets four times daily). These events resolved after discontinuing the dicyclomine.
The acute oral LD50 of the drug is 625 mg/kg in mice.
The amount of drug in a single dose that is ordinarily associated with symptoms of overdosage or that is likely to be life-threatening, has not been defined. The maximum human oral dose recorded was 600 mg by mouth in a 10-month-old child and approximately 1500 mg in an adult, each of whom survived. In three of the infants who died following administration of dicyclomine hydrochloride [see Warnings and Precautions (5.1)], the blood concentrations of drug were 200, 220, and 505 ng/mL.
It is not known if dicyclomine hydrochloride is dialyzable.
Treatment should consist of gastric lavage, emetics, and activated charcoal. Sedatives (e.g., short-acting barbiturates, benzodiazepines) may be used for management of overt signs of excitement. If indicated, an appropriate parenteral cholinergic agent may be used as an antidote.
-
11 DESCRIPTION
Dicyclomine hydrochloride is an antispasmodic and anticholinergic (antimuscarinic) agent available in the following dosage form:
- •
- Dicyclomine Hydrochloride Injection, USP is a sterile, pyrogen-free, aqueous solution for intramuscular injection (NOT FOR INTRAVENOUS USE) supplied as a vial containing 20 mg/2 mL (10 mg/mL). Each mL contains 10 mg dicyclomine hydrochloride USP in sterile water for injection, made isotonic with sodium chloride.
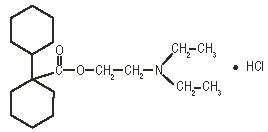
Dicyclomine hydrochloride is [bicyclohexyl]-1- carboxylic acid, 2-(diethylamino) ethyl ester, hydrochloride, with a molecular formula of C19H35NO2•HCl and the following structural formula:
Molecular weight: 345.95
Dicyclomine Hhydrochloride occurs as a fine, white, crystalline, practically odorless powder with a bitter taste. It is soluble in water, freely soluble in alcohol and chloroform, and very slightly soluble in ether.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Dicyclomine relieves smooth muscle spasm of the gastrointestinal tract. Animal studies indicate that this action is achieved via a dual mechanism:
- •
- a specific anticholinergic effect (antimuscarinic) at the acetylcholine-receptor sites with approximately 1/8 the milligram potency of atropine (in vitro, guinea pig ileum); and
- •
- a direct effect upon smooth muscle (musculotropic) as evidenced by dicyclomine’s antagonism of bradykinin- and histamine-induced spasms of the isolated guinea pig ileum.
Atropine did not affect responses to these two agonists. In vivo studies in cats and dogs showed dicyclomine to be equally potent against acetylcholine (ACh)- or barium chloride (BaCl2)-induced intestinal spasm while atropine was at least 200 times more potent against effects of ACh than BaCl2. Tests for mydriatic effects in mice showed that dicyclomine was approximately 1/500 as potent as atropine; antisialagogue tests in rabbits showed dicyclomine to be 1/300 as potent as atropine.
12.2 Pharmacodynamics
Dicyclomine hydrochloride can inhibit the secretion of saliva and sweat, decrease gastrointestinal secretions and motility, cause drowsiness, dilate the pupils, increase heart rate, and depress motor function.
12.3 Pharmacokinetics
Absorption and Distribution
In man, dicyclomine is rapidly absorbed after oral administration, reaching peak values within 60-90 minutes. Mean volume of distribution for a 20 mg oral dose is approximately 3.65 L/kg suggesting extensive distribution in tissues.
Elimination
The metabolism of dicyclomine was not studied. The principal route of excretion is via the urine (79.5% of the dose). Excretion also occurs in the feces, but to a lesser extent (8.4%). Mean half-life of plasma elimination in one study was determined to be approximately 1.8 hours when plasma concentrations were measured for 9 hours after a single dose. In subsequent studies, plasma concentrations were followed for up to 24 hours after a single dose, showing a secondary phase of elimination with a somewhat longer half-life.
- 13 NONCLINICAL TOXICOLOGY
-
14 CLINICAL STUDIES
In controlled clinical trials involving over 100 patients who received drug, 82% of patients treated for functional bowel/irritable bowel syndrome with dicyclomine hydrochloride at initial doses of 160 mg daily (40 mg four times daily) demonstrated a favorable clinical response compared with 55% treated with placebo (p<0.05).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Dicyclomine Hydrochloride Injection, USP is available as a single dose vial in the following size: 20 mg/2 mL (10 mg/mL) injection (NDC 70436-147-80) supplied in boxes of five 20 mg/2 mL vials (10 mg/mL) (NDC 70436-147-81). Store at room temperature, preferably below 86°F (30°C). Protect from freezing.
-
17 PATIENT COUNSELING INFORMATION
17.1 Inadvertent Intravenous Administration
Dicyclomine hydrochloride injection is for intramuscular administration only. Do not administer by any other route. Inadvertent administration may result in thrombosis or thrombophlebitis, and injection site such as pain, edema, skin color change and even reflex sympathetic dystrophy syndrome [see Adverse Reactions (6.2)].
17.2 Use in Infants
Inform parents and caregivers not to administer dicyclomine hydrochloride in infants less than 6 months of age [see Use in Specific Populations (8.4)].
17.3 Use in Nursing Mothers
Advise lactating women that dicyclomine hydrochloride should not be used while breastfeeding their infants [see Use in Specific Populations (8.3, 8.4)].
17.4 Peripheral and Central Nervous System
In the presence of a high environmental temperature, heat prostration can occur with dicyclomine hydrochloride use (fever and heat stroke due to decreased sweating). If symptoms occur, the drug should be discontinued and a physician contacted. Dicyclomine hydrochloride may produce drowsiness or blurred vision. The patient should be warned not to engage in activities requiring mental alertness, such as operating a motor vehicle or other machinery or to perform hazardous work while taking dicyclomine hydrochloride [see Warnings and Precautions (5.3)].
Rx only
Manufactured by:
Hainan Poly Pharm. Co., Ltd., Guilinyang Economic Development Zone, Haikou, Hainan, China 57112Distributed by:
Slate Run Pharmaceuticals, LLC, Columbus, Ohio 43215
10000180/02
Revised 09/2021
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DICYCLOMINE HYDROCHLORIDE
dicyclomine hydrochloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70436-147 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DICYCLOMINE HYDROCHLORIDE (UNII: CQ903KQA31) (DICYCLOMINE - UNII:4KV4X8IF6V) DICYCLOMINE HYDROCHLORIDE 20 mg in 2 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70436-147-81 5 in 1 BOX 09/23/2021 1 2 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA207076 09/23/2021 Labeler - Slate Run Pharmaceuticals, LLC (039452765) Registrant - Slate Run Pharmaceuticals, LLC (039452765) Establishment Name Address ID/FEI Business Operations Olon S.p.A. 433967359 API MANUFACTURE(70436-147)