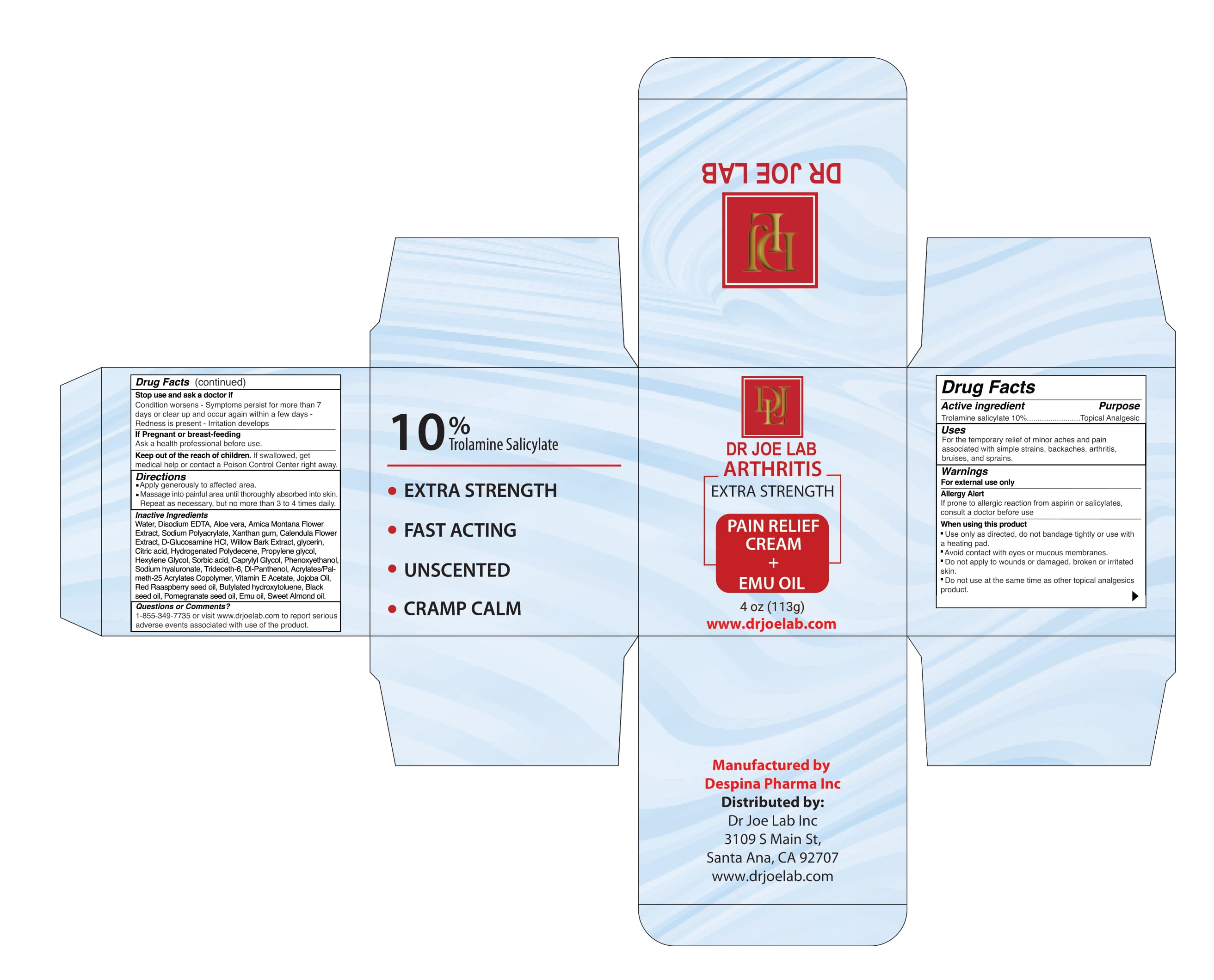
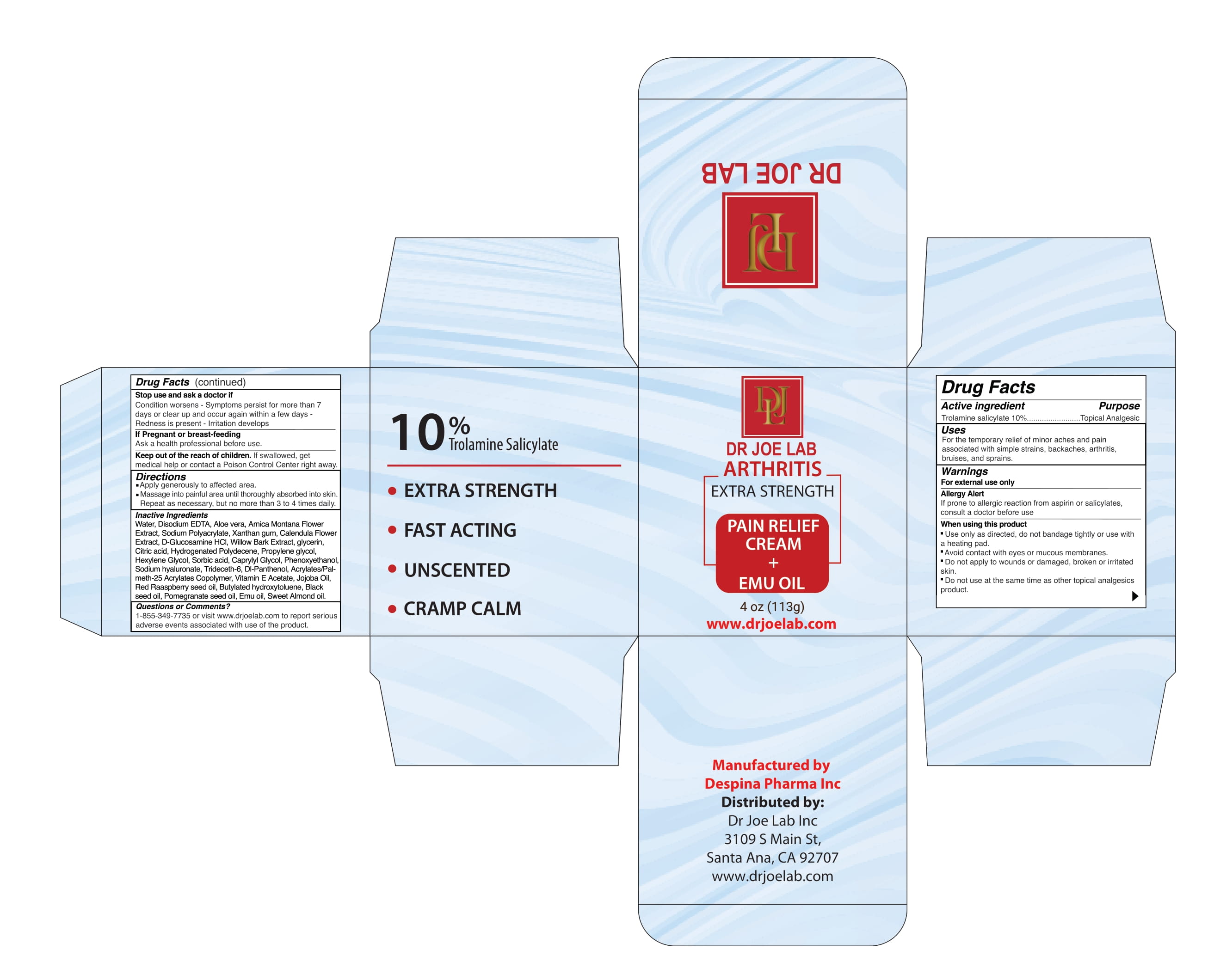
Label: DR JOE LAB ARTHRITIS EXTRA STRENGTH- trolamine salicylate cream
- NDC Code(s): 82330-501-01
- Packager: DESPINA PHARMA INC.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated December 2, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- uses
- WARNINGS
- When Using
- Stop use and ask a doctor if
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- INSTRUCTIONS FOR USE
-
INACTIVE INGREDIENT
Water, Disodium EDTA, Aloe vera, Arnica Montana Flower Extract, Sodium Polyacrylate, Xanthan gum, Calendula Flower Extract, D-Glucosamine HCl, Willow Bark Extract, glycerin, Citric acid, Hydrogenated Polydecene, Propylene glycol, Hexylene Glycol, Sorbic acid, Caprylyl Glycol, Phenoxyethanol, Sodium hyaluronate, Trideceth-6, Dl-Panthenol, Acrylates/Palmeth- 25 Acrylates Copolymer, Vitamin E Acetate, Jojoba Oil, Red Raaspberry seed oil, Butylated hydroxytoluene, Black seed oil, Pomegranate seed oil, Emu oil, Sweet Almond oil.
- QUESTIONS
- DESCRIPTION
- PURPOSE
- DOSAGE & ADMINISTRATION
- label
-
INGREDIENTS AND APPEARANCE
DR JOE LAB ARTHRITIS EXTRA STRENGTH
trolamine salicylate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82330-501 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TROLAMINE SALICYLATE (UNII: H8O4040BHD) (SALICYLIC ACID - UNII:O414PZ4LPZ) TROLAMINE SALICYLATE 10 g in 100 g Inactive Ingredients Ingredient Name Strength METHACRYLIC ACID AND ETHYL ACRYLATE COPOLYMER (UNII: NX76LV5T8J) EDETATE DISODIUM (UNII: 7FLD91C86K) SODIUM POLYACRYLATE (2500000 MW) (UNII: 05I15JNI2J) GLYCERIN (UNII: PDC6A3C0OX) ARNICA MONTANA (UNII: O80TY208ZW) WILLOW BARK (UNII: S883J9JDYX) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) HEXYLENE GLYCOL (UNII: KEH0A3F75J) SORBIC ACID (UNII: X045WJ989B) WATER (UNII: 059QF0KO0R) ALOE VERA LEAF (UNII: ZY81Z83H0X) XANTHAN GUM (UNII: TTV12P4NEE) CALENDULA OFFICINALIS FLOWERING TOP (UNII: 18E7415PXQ) GLUCOSAMINE HYDROCHLORIDE (UNII: 750W5330FY) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) HYDROGENATED POLYDECENE (1500 CST) (UNII: 4YI0729529) HYALURONATE SODIUM (UNII: YSE9PPT4TH) TRIDECETH-6 (UNII: 3T5PCR2H0C) PANTHENOL (UNII: WV9CM0O67Z) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) JOJOBA OIL (UNII: 724GKU717M) RASPBERRY SEED OIL (UNII: 9S8867952A) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) NIGELLA SATIVA SEED OIL (UNII: CS4U38E731) POMEGRANATE SEED OIL (UNII: 0UI45XV0T6) EMU OIL (UNII: 344821WD61) ALMOND OIL (UNII: 66YXD4DKO9) CAPRYLYL GLYCOL (UNII: 00YIU5438U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82330-501-01 113 g in 1 JAR; Type 0: Not a Combination Product 06/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M014 06/01/2022 Labeler - DESPINA PHARMA INC. (112281681) Establishment Name Address ID/FEI Business Operations Despina Pharma Inc 112281681 manufacture(82330-501)