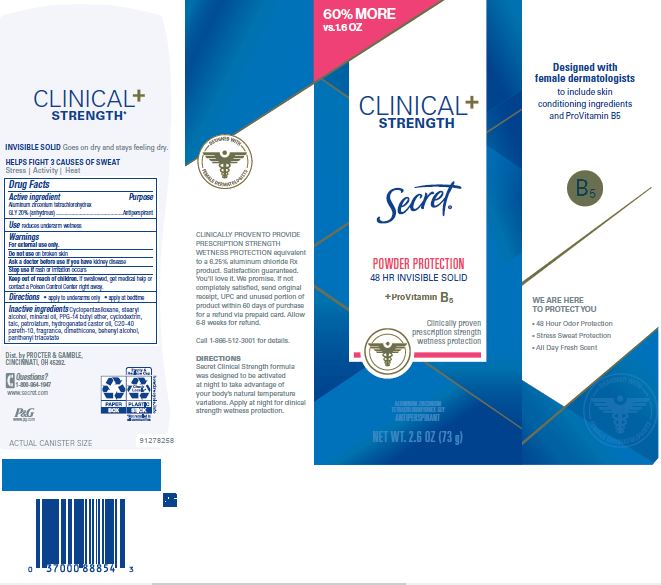
Label: SECRET CLINICAL STRENGTH INVISIBLE PROTECTION- aluminum zirconium tetrachlorohydrex gly stick
- NDC Code(s): 69423-070-14, 69423-070-45, 69423-070-73
- Packager: The Procter & Gamble Manufacturing Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 20, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
- Warnings
- Directions
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 45 g Canister Carton
-
INGREDIENTS AND APPEARANCE
SECRET CLINICAL STRENGTH INVISIBLE PROTECTION
aluminum zirconium tetrachlorohydrex gly stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69423-070 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY (UNII: 8O386558JE) (ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY - UNII:8O386558JE) ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY 20 g in 100 g Inactive Ingredients Ingredient Name Strength BETADEX (UNII: JV039JZZ3A) PANTHENOL TRIACETATE, (+)- (UNII: 1206E8961B) PPG-14 BUTYL ETHER (UNII: R199TJT95T) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) MINERAL OIL (UNII: T5L8T28FGP) TALC (UNII: 7SEV7J4R1U) PETROLATUM (UNII: 4T6H12BN9U) HYDROGENATED CASTOR OIL (UNII: ZF94AP8MEY) C20-40 PARETH-10 (UNII: TE3MZI4V3F) DIMETHICONE (UNII: 92RU3N3Y1O) DOCOSANOL (UNII: 9G1OE216XY) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69423-070-14 1 in 1 CARTON 11/16/2015 02/15/2018 1 14 g in 1 CYLINDER; Type 0: Not a Combination Product 2 NDC:69423-070-45 1 in 1 CARTON 11/16/2015 09/01/2024 2 45 g in 1 CYLINDER; Type 0: Not a Combination Product 3 NDC:69423-070-73 1 in 1 CARTON 11/16/2015 09/01/2024 3 73 g in 1 CYLINDER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M019 11/16/2015 09/01/2024 Labeler - The Procter & Gamble Manufacturing Company (004238200)