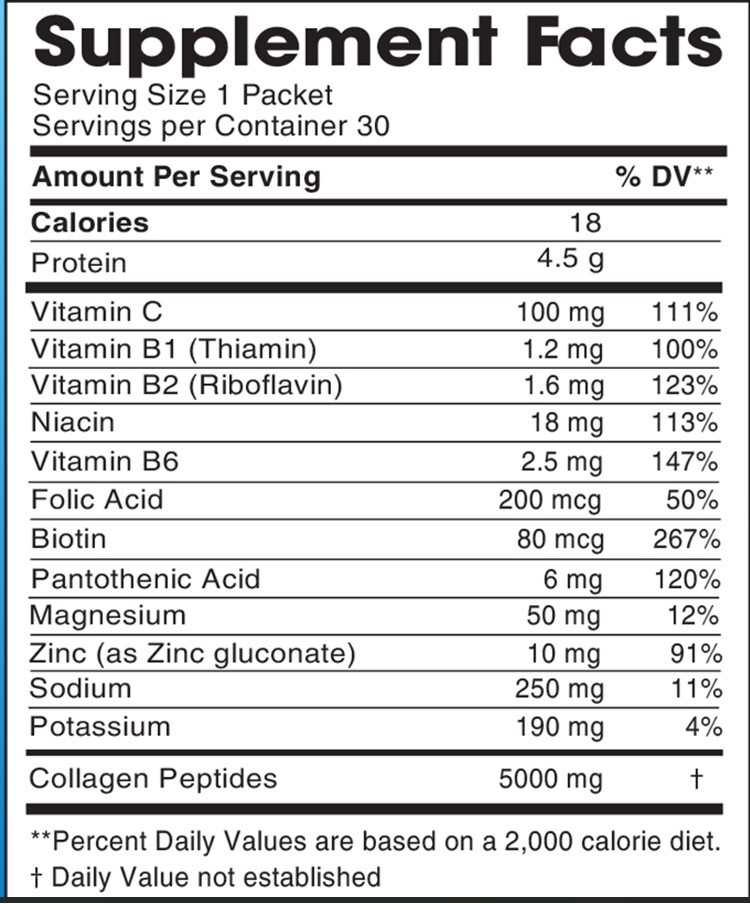
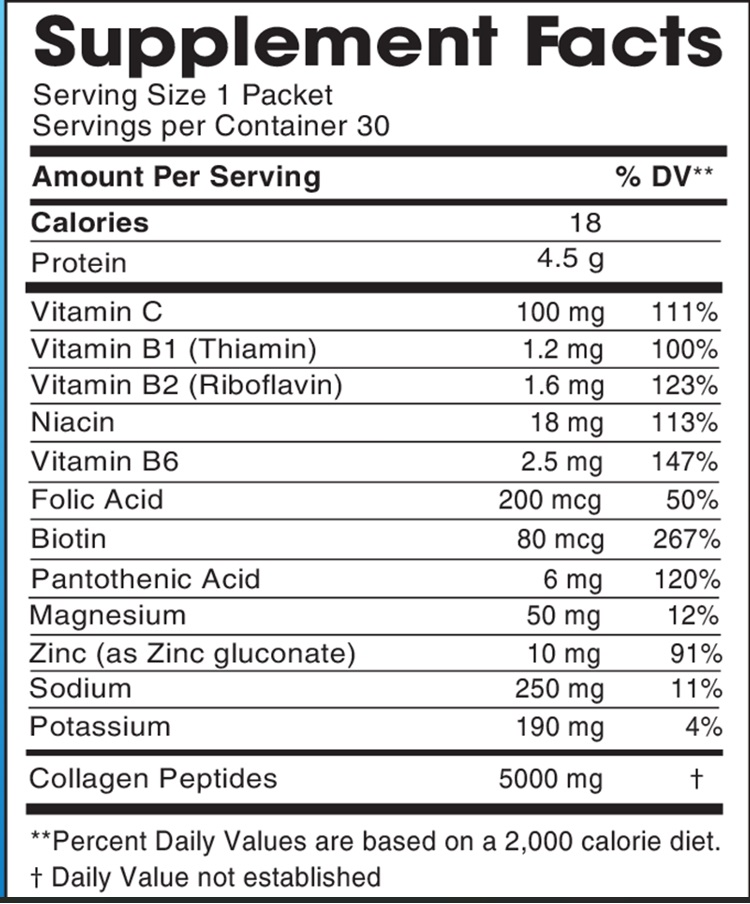
Label: JBA COLLAGEN-ELECTROLYTE MOCKTAIL- vitamin c,thiamin,riboflavin,niacin,vitamin b6,folate,biotin,pantothenic acid,magnesium,zinc,sodium,potassium,bovine collagen peptides powder
- NDC Code(s): 13411-848-05, 13411-848-15, 13411-848-30
- Packager: Advanced Pharmaceutical Services, Inc. Dba Affordable Quality Pharmaceuticals
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated March 25, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Identity
-
Warnings
BEFORE USING this dietary supplement ask your doctor if you
- Are considering using this product as part of a cholesterol lowering program
- Are using medication to control your blood sugar levels
- Have recently experienced abdominal pain, nausea, vomiting or a sudden change in bowel habits persisting for two weeks.
Warning:
Bulk forming fibers like psyllium husk may affect how well medicines work. Take this product at least 2 hours before or after medicines. - Direction
- Safe handling warning
-
HEALTH CLAIM
JBA® Collagen-Electrolyte Mocktail
5,000 mg Collagen Peptides for youthful skin, hair, nail, and joint*
Electrolyte Mix for FAST relief from dehydration caused by:- Exercise
- Travel
- Heat
- Sleep
- Nausea
- Food Poisoning
Collagen-Electrolyte Mocktail
Collagen Meets Hydration – Enjoy Healing in every cup ™Collagen
- Strengthen hair, skin, and nails*
- Supporting muscles and joint health*
- Supporting control blood sugar*
- Supporting immune health*
Electrolyte
- Support rapid hydration and replenish electrolytes*
- Promotes relaxation and rest quality*
- Help ease feeling of stress*
With 5,000 mg collagen peptides and 111% Daily Recommended Value of Vitamin C, acting as an antioxidant to help replenish collagen levels for firmer, plumper, younger-looking skin and stronger, healthier hair, nails and joints.
* These statements have not been evaluated by the Food & Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
- PRECAUTIONS
- INACTIVE INGREDIENT
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- ACTIVE INGREDIENT
- PURPOSE
- Product label
-
INGREDIENTS AND APPEARANCE
JBA COLLAGEN-ELECTROLYTE MOCKTAIL
vitamin c,thiamin,riboflavin,niacin,vitamin b6,folate,biotin,pantothenic acid,magnesium,zinc,sodium,potassium,bovine collagen peptides powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13411-848 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 100 mg in 12 g THIAMINE (UNII: X66NSO3N35) (THIAMINE ION - UNII:4ABT0J945J) THIAMINE 1.2 mg in 12 g RIBOFLAVIN (UNII: TLM2976OFR) (RIBOFLAVIN - UNII:TLM2976OFR) RIBOFLAVIN 1.6 mg in 12 g NIACIN (UNII: 2679MF687A) (NIACIN - UNII:2679MF687A) NIACIN 18 mg in 12 g PYRIDOXINE (UNII: KV2JZ1BI6Z) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE 2.5 mg in 12 g FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 200 ug in 12 g BIOTIN (UNII: 6SO6U10H04) (BIOTIN - UNII:6SO6U10H04) BIOTIN 80 ug in 12 g PANTOTHENIC ACID (UNII: 19F5HK2737) (PANTOTHENIC ACID - UNII:19F5HK2737) PANTOTHENIC ACID 6 mg in 12 g MAGNESIUM (UNII: I38ZP9992A) (MAGNESIUM - UNII:I38ZP9992A) MAGNESIUM 50 mg in 12 g ZINC (UNII: J41CSQ7QDS) (ZINC - UNII:J41CSQ7QDS) ZINC 10 mg in 12 g SODIUM (UNII: 9NEZ333N27) (SODIUM - UNII:9NEZ333N27) SODIUM 250 mg in 12 g POTASSIUM (UNII: RWP5GA015D) (POTASSIUM - UNII:RWP5GA015D) POTASSIUM 190 mg in 12 g Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) POVIDONE K30 (UNII: U725QWY32X) MALTODEXTRIN (UNII: 7CVR7L4A2D) MANNITOL (UNII: 3OWL53L36A) STEVIA REBAUDIUNA LEAF (UNII: 6TC6NN0876) SUCRALOSE (UNII: 96K6UQ3ZD4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13411-848-30 30 in 1 BOX 03/22/2024 1 12 g in 1 PACKET; Type 0: Not a Combination Product 2 NDC:13411-848-15 15 in 1 BOX 03/22/2024 2 12 g in 1 PACKET; Type 0: Not a Combination Product 3 NDC:13411-848-05 5 in 1 BOX 03/22/2024 3 12 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 03/22/2024 Labeler - Advanced Pharmaceutical Services, Inc. Dba Affordable Quality Pharmaceuticals (187498279) Establishment Name Address ID/FEI Business Operations Advanced Pharmaceutical Services, Inc. Dba Affordable Quality Pharmaceuticals 187498279 manufacture(13411-848)