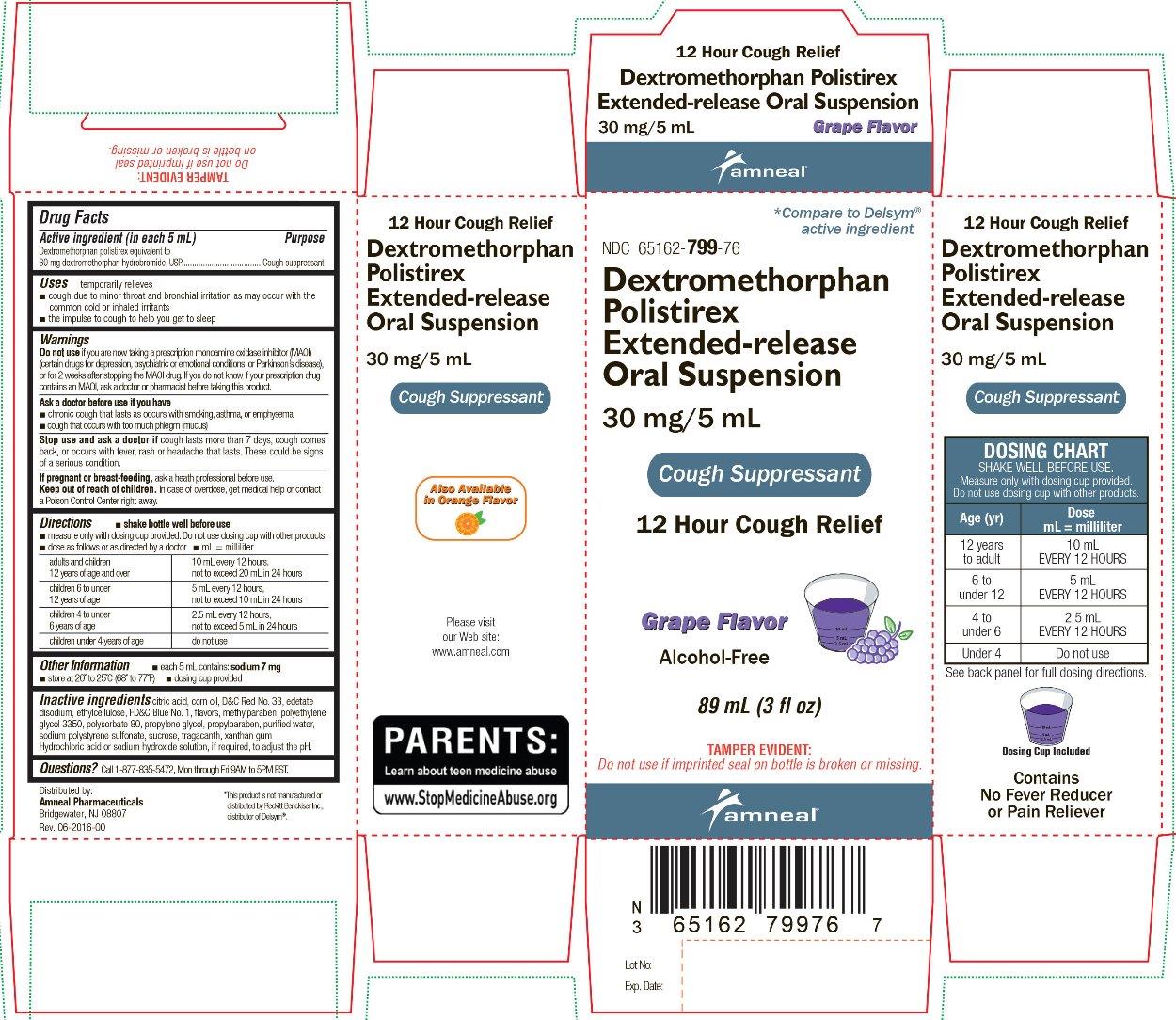
Label: DEXTROMETHORPHAN POLISTIREX EXTENDED-RELEASE- dextromethorphan polistirex suspension
- NDC Code(s): 65162-799-76, 65162-799-77
- Packager: Amneal Pharmaceuticals LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated December 18, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- OTC - ACTIVE INGREDIENT
- OTC - PURPOSE
- INDICATIONS AND USAGE
- WARNINGS
-
OTC - DO NOT USE
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
-
OTC - ASK DOCTOR
Ask a doctor before use if you have
- chronic cough that lasts as occurs with smoking, asthma or emphysema
- cough that occurs with too much phlegm (mucus)
Stop use and ask a doctor if cough lasts more than 7 days, cough comes back, or occurs with fever, rash or headache that lasts. These could be signs of a serious condition.
- OTC - PREGNANCY OR BREAST FEEDING
- OTC - KEEP OUT OF REACH OF CHILDREN
-
DOSAGE AND ADMINISTRATION
Directions
- shake bottle well before use
- measure only with dosing cup provided. Do not use dosing cup with other products.
- dose as follows or as directed by a doctor
adults and children 12 years of age and over 10 mL every 12 hours,
not to exceed 20 mL in 24 hourschildren 6 to under 12 years of age 5 mL every 12 hours,
not to exceed 10 mL in 24 hourschildren 4 to under 6 years of age 2.5 mL every 12 hours,
not to exceed 5 mL in 24 hoursChildren under 4 years of age do not use Other information
- each 5 mL contains: sodium 7 mg
- store at 20° to 25°C (68° to 77°F)
- dosing cup provided
- mL = milliliter
-
INACTIVE INGREDIENT
Inactive ingredients
citric acid, corn oil, D&C Red No. 33, edetate disodium, ethylcellulose, FD&C Blue No. 1, flavors, methylparaben, polyethylene glycol 3350, polysorbate 80, propylene glycol, propylparaben, purified water, sodium polystyrene sulfonate, sucrose, tragacanth, xanthan gum
Hydrochloric acid or sodium hydroxide solution, if required, to adjust the pH.
- OTC - QUESTIONS
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DEXTROMETHORPHAN POLISTIREX EXTENDED-RELEASE
dextromethorphan polistirex suspensionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:65162-799 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 30 mg in 5 mL Inactive Ingredients Ingredient Name Strength SODIUM POLYSTYRENE SULFONATE (UNII: 1699G8679Z) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) CORN OIL (UNII: 8470G57WFM) D&C RED NO. 33 (UNII: 9DBA0SBB0L) EDETATE DISODIUM (UNII: 7FLD91C86K) ETHYLCELLULOSES (UNII: 7Z8S9VYZ4B) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) METHYLPARABEN (UNII: A2I8C7HI9T) POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) POLYSORBATE 80 (UNII: 6OZP39ZG8H) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SUCROSE (UNII: C151H8M554) TRAGACANTH (UNII: 2944357O2O) XANTHAN GUM (UNII: TTV12P4NEE) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Product Characteristics Color purple Score Shape Size Flavor GRAPE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65162-799-76 1 in 1 CARTON 08/01/2017 1 89 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:65162-799-77 1 in 1 CARTON 08/01/2017 2 148 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA203133 08/01/2017 Labeler - Amneal Pharmaceuticals LLC (123797875) Establishment Name Address ID/FEI Business Operations Amneal Pharmaceuticals, LLC 963900878 analysis(65162-799) , label(65162-799) , manufacture(65162-799) , pack(65162-799)