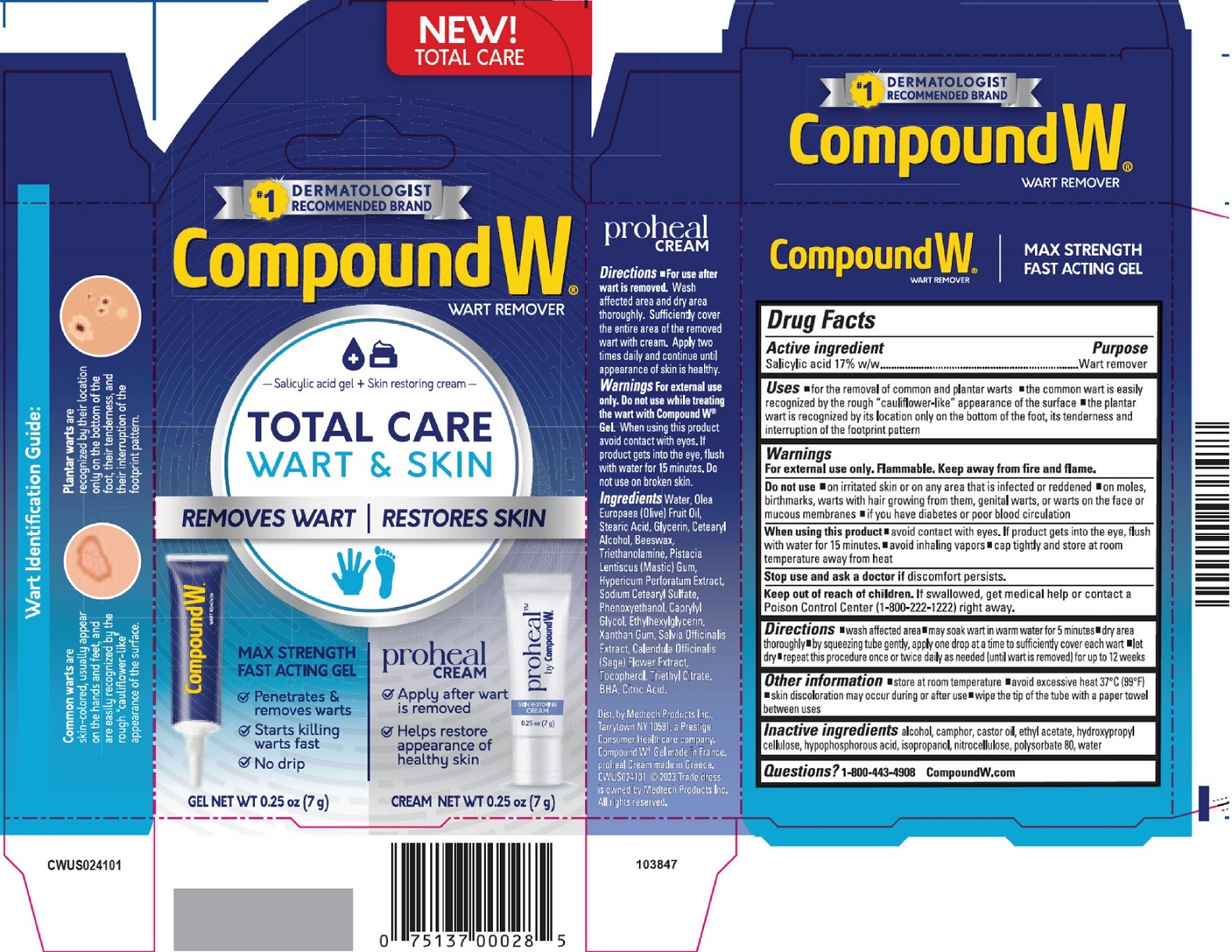
Label: COMPOUND W TOTAL CARE WART AND SKIN KIT- salicylic acid kit
- NDC Code(s): 49354-003-00, 49354-130-00
- Packager: HK Kolmar Canada, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Uses
-
Warnings
For external use only. Flammable. Keep away from fire and flame.
Do not use
- on irritated skin or on any area that is infected or reddened
- on moles, birthmarks, warts with hair growing from them, genital warts or warts on the face or mucous membranes.
- if you have diabetes or poor blood circulation.
- Directions
- Other information
- Inactive ingredients
- Questions?
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
COMPOUND W TOTAL CARE WART AND SKIN KIT
salicylic acid kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49354-130 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49354-130-00 1 in 1 KIT 12/05/2022 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 7 g Part 1 of 1 COMPOUND W WART REMOVER MAXIMUM STRENGTH FAST ACTING
salicylic acid gelProduct Information Item Code (Source) NDC:49354-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 170 mg in 1 g Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) CASTOR OIL (UNII: D5340Y2I9G) ETHYL ACETATE (UNII: 76845O8NMZ) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPOPHOSPHOROUS ACID (UNII: 8B1RL9B4ZJ) ISOPROPYL ALCOHOL (UNII: ND2M416302) PYROXYLIN (UNII: KYR8BR2X6O) POLYSORBATE 80 (UNII: 6OZP39ZG8H) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49354-003-00 1 in 1 CARTON 1 7 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M032 12/05/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M032 12/05/2022 Labeler - HK Kolmar Canada, Inc (243501959)