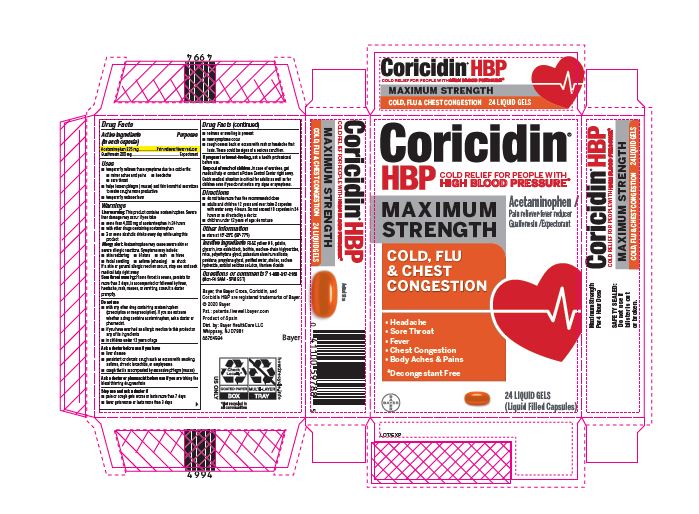
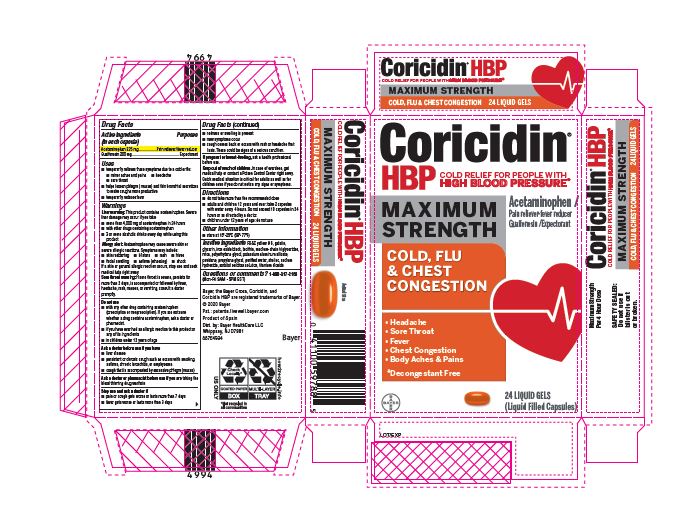
Label: CORICIDIN HBP MAXIMUM STRENGTH COLD FLU AND CHEST CONGESTION- acetaminophen, guaifenesin capsule, gelatin coated
- NDC Code(s): 11523-0094-1
- Packager: Bayer HealthCare LLC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 30, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take • more than 4,000 mg of acetaminophen in 24 hours • with other drugs containing acetaminophen • 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin or severe allergic reactions. Symptoms may include: • skin reddening • blisters • rash • hives • facial swelling • asthma (wheezing) • shock If a skin or general allergic reaction occurs, stop use and seek medical help right away.
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly
- Do not use
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
- ?
- PRINCIPAL DISPLAY PANEL - 24 Liquid Gels
-
INGREDIENTS AND APPEARANCE
CORICIDIN HBP MAXIMUM STRENGTH COLD FLU AND CHEST CONGESTION
acetaminophen, guaifenesin capsule, gelatin coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11523-0094 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 200 mg Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) MICA (UNII: V8A1AW0880) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) SODIUM HYDROXIDE (UNII: 55X04QC32I) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) SHELLAC (UNII: 46N107B71O) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) SORBITAN (UNII: 6O92ICV9RU) POTASSIUM ALUMINUM DISILICATE (UNII: SRB14JRX6C) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) Product Characteristics Color orange Score no score Shape OVAL (Oblong) Size 18mm Flavor Imprint Code CHBPD Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11523-0094-1 2 in 1 CARTON 05/02/2022 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 05/01/2022 Labeler - Bayer HealthCare LLC. (112117283)