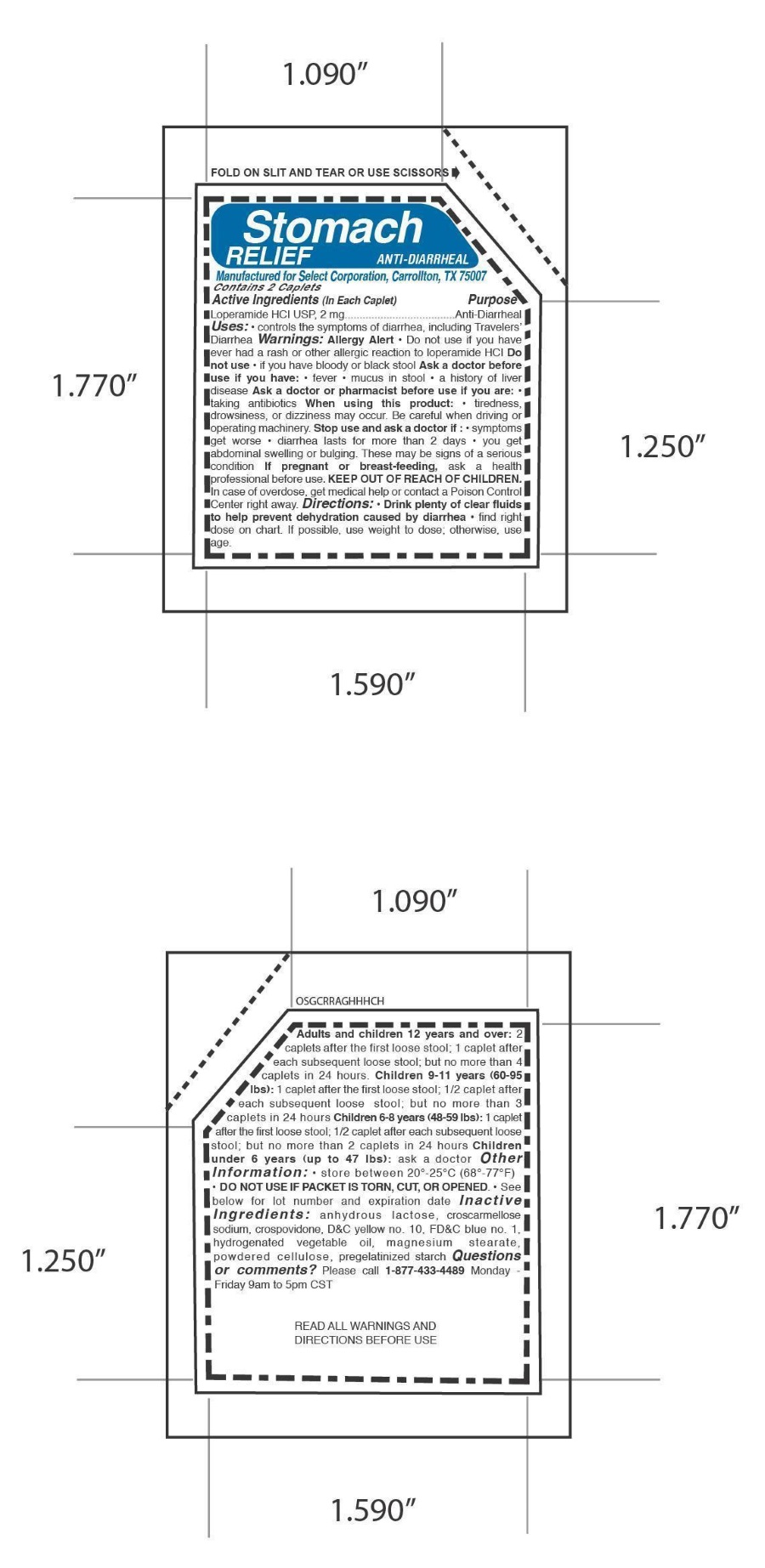
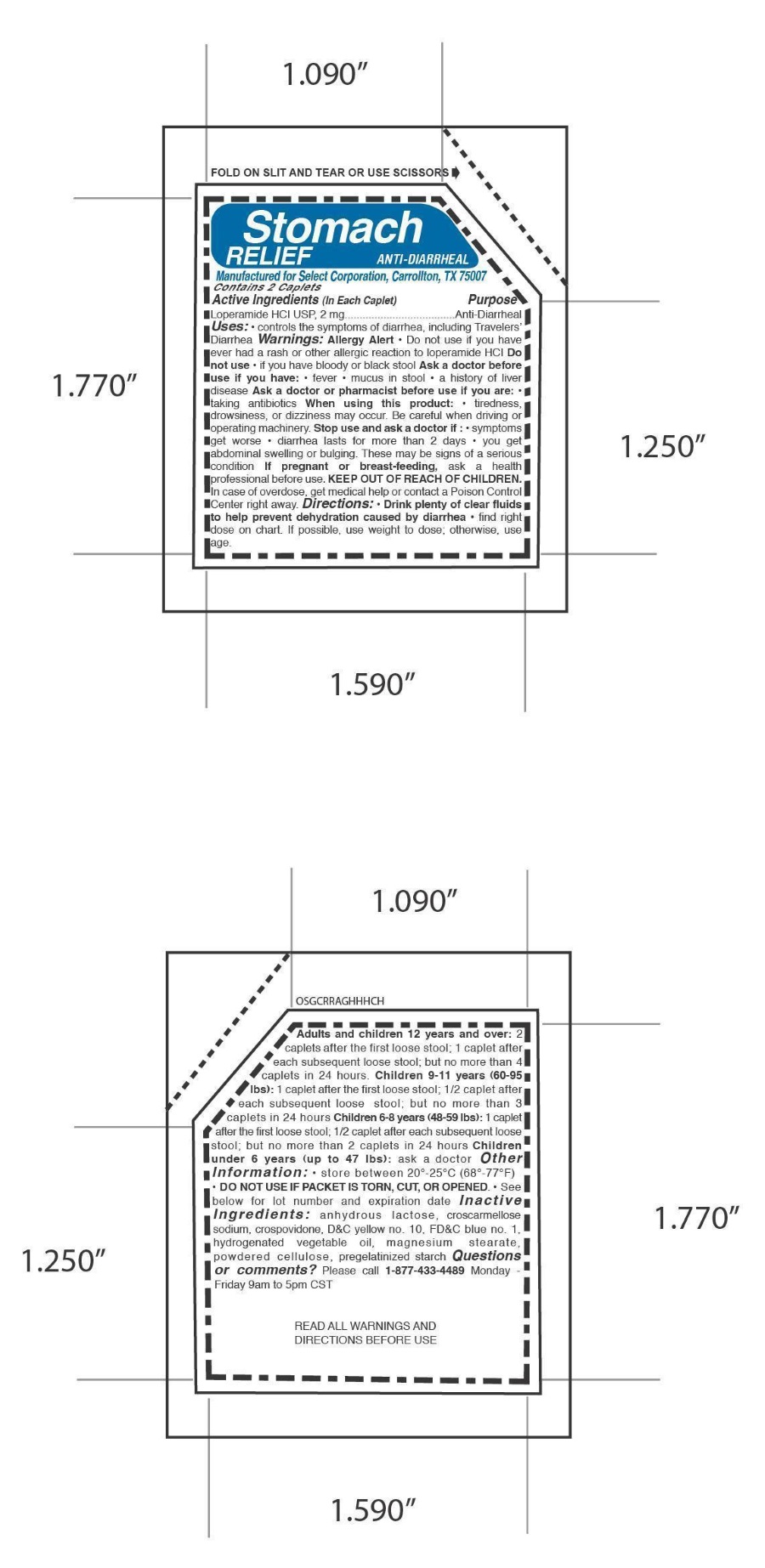
Label: STOMACH RELIEF ANTI DIARRHEAL- loperamide hydrochloride tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 52904-443-02, 52904-443-06, 52904-443-50 - Packager: Select Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 15, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
-
DOSAGE
Directions: • Drink plenty of clear fluids
to help prevent dehydration caused by diarrhea • find right
dose on chart. If possible, use weight to dose; otherwise, use
age.Adults and children 12 years and over: 2
caplets after the first loose stool; 1 caplet after
each subsequent loose stool; but no more than 4
caplets in 24 hours. Children 9-11 years (60-95
lbs): 1 caplet after the first loose stool; 1/2 caplet after
each subsequent loose stool; but no more than 3
caplets in 24 hours Children 6-8 years (48-59 lbs): 1 caplet
after the first loose stool; 1/2 caplet after each subsequent loose
stool; but no more than 2 caplets in 24 hours Children
under 6 years (up to 47 lbs): ask a doctor
- INDICATIONS & USAGE
-
WARNINGS
Allergy Alert
Do not use
• if you have ever had a rash or other allergic reaction to loperamide HCI
• if you have bloody or black stool Ask a doctor before use
• if you have: • fever • mucus in stool • a history of liver disease Ask a doctor or pharmacist before use
• if you are: • taking antibiotics When using this product: • tiredness, drowsiness, or dizziness may occur.
Be careful when driving or operating machinery.
Stop use and ask a doctor if :
• symptoms get worse
• diarrhea lasts for more than 2 days
• you get abdominal swelling or bulging.
These may be signs of a serious condition
- PREGNANCY
- KEEP OUT OF REACH OF CHILDREN
- INACTIVE INGREDIENT
- LABEL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
STOMACH RELIEF ANTI DIARRHEAL
loperamide hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:52904-443 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOPERAMIDE HYDROCHLORIDE (UNII: 77TI35393C) (LOPERAMIDE - UNII:6X9OC3H4II) LOPERAMIDE HYDROCHLORIDE 2 mg Inactive Ingredients Ingredient Name Strength LACTOSE (UNII: J2B2A4N98G) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) CROSPOVIDONE (UNII: 2S7830E561) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) MAGNESIUM STEARATE (UNII: 70097M6I30) POWDERED CELLULOSE (UNII: SMD1X3XO9M) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color green (pea green) Score no score Shape CAPSULE (123) Size 10mm Flavor Imprint Code 123 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52904-443-50 25 in 1 BOX 10/15/2012 1 NDC:52904-443-06 1 in 1 CARTON 1 NDC:52904-443-02 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA074091 10/15/2012 Labeler - Select Corporation (053805599) Establishment Name Address ID/FEI Business Operations Select Corporation 053805599 label(52904-443) Establishment Name Address ID/FEI Business Operations Ohm Laboratories Inc. 184769029 manufacture(52904-443)