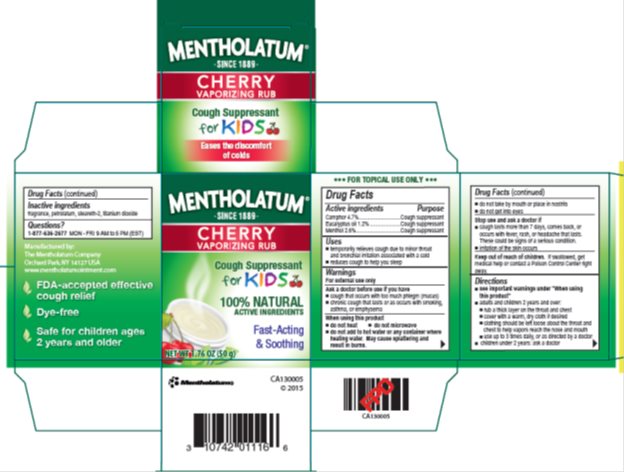
Label: MENTHOLATUM CHERRY VAPORIZING RUB- camphor, eucalyptus oil, menthol ointment
- NDC Code(s): 10742-8731-1
- Packager: The Mentholatum Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 7, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Uses
-
Warnings
For external use only
Ask a doctor before use if you have
- cough that occurs with too much phlegm (mucus)
- chronic cough that lasts or as occurs with smoking, asthma, or emphysema
When using this product
- do not heat
- do not microwave
- do not add to hot water or any container where heating water. May cause splattering and result in burns.
- do not take by mouth or place in nostrils
- do not get into eyes
-
Directions
- see important warnings under "When using this product"
- adults and children 2 years and over:
- rub a thick layer on throat and chest
- cover with a warm, dry cloth if desired
- clothing should be left loose about the throat and chest to help vapors reach the nose and mouth
- use up to 3 times daily, or as directed by a doctor
- children under 2 years: ask a doctor
- Inactive ingredients
- Questions?
- Package/Label Principal Display Panel
-
INGREDIENTS AND APPEARANCE
MENTHOLATUM CHERRY VAPORIZING RUB
camphor, eucalyptus oil, menthol ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10742-8731 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (NATURAL) (UNII: N20HL7Q941) (CAMPHOR (NATURAL) - UNII:N20HL7Q941) CAMPHOR (NATURAL) 47 mg in 1 g EUCALYPTUS OIL (UNII: 2R04ONI662) (EUCALYPTUS OIL - UNII:2R04ONI662) EUCALYPTUS OIL 12 mg in 1 g MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 26 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) STEARETH-2 (UNII: V56DFE46J5) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10742-8731-1 1 in 1 CARTON 02/16/2015 1 50 g in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 02/16/2015 Labeler - The Mentholatum Company (002105757) Registrant - The Mentholatum Company (002105757) Establishment Name Address ID/FEI Business Operations The Mentholatum Company 002105757 manufacture(10742-8731)