Label: CLEAR SINUS AND EAR- pulsatilla vulgaris, euphorbia resinifera resin, calcium sulfate and oyster shell calcium carbonate, crude capsule
- NDC Code(s): 68089-004-01
- Packager: Clear Products, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated November 14, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
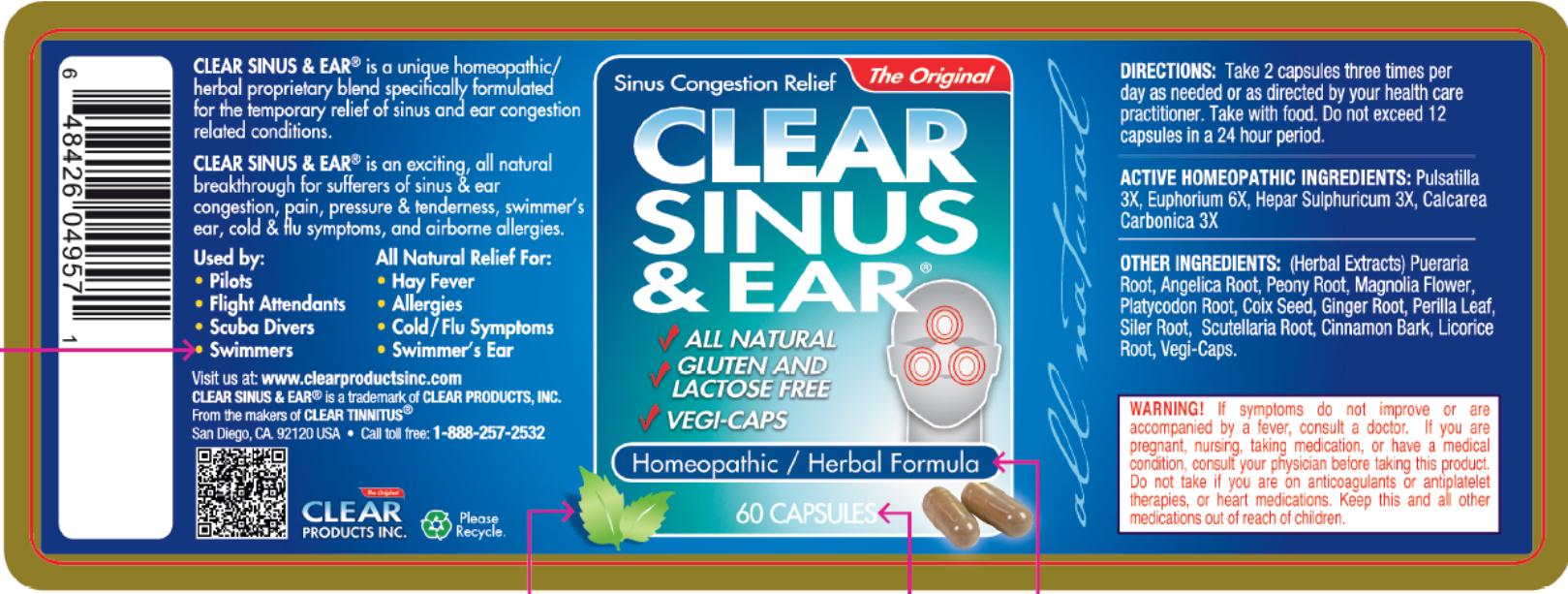
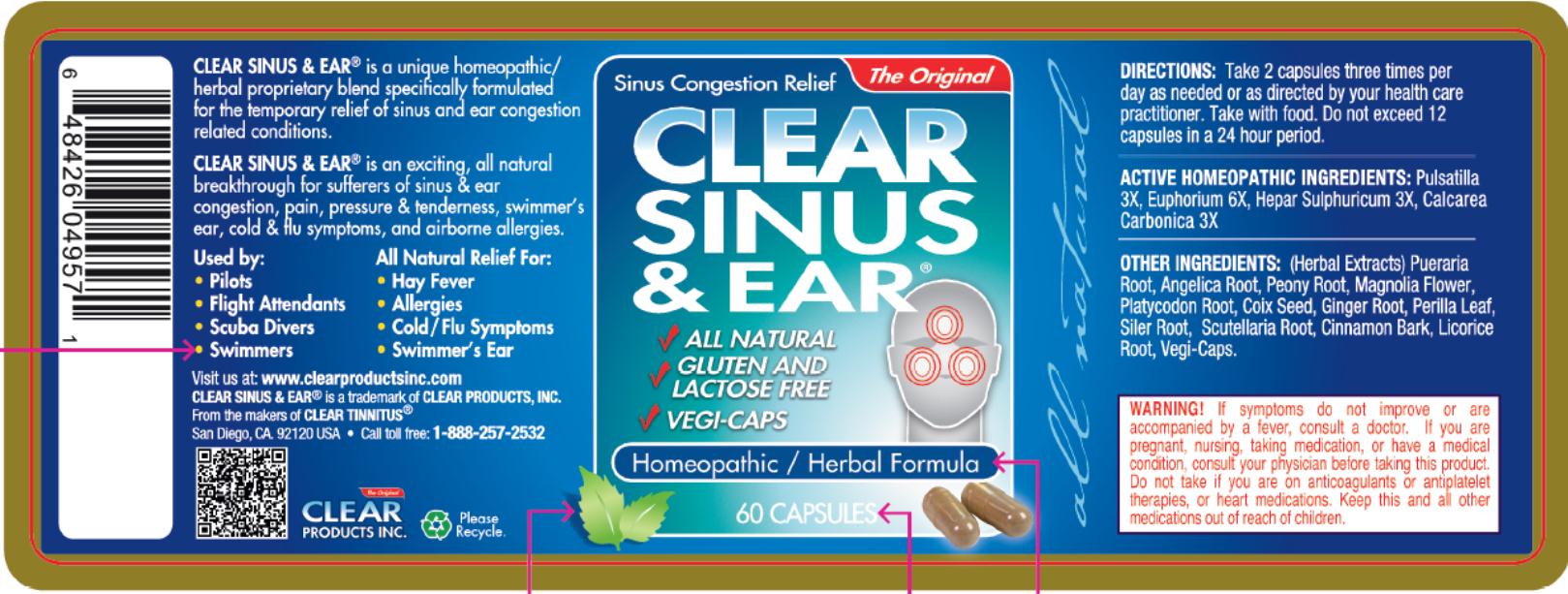
- DIRECTIONS:
- ACTIVE HOMEOPATHIC INGREDIENTS:
- OTHER INGREDIENTS:
- WARNING!
- If you are pregnant, nursing,
- Do not take
- Keep this and all other medications out of reach of children.
-
CLEAR SINUS AND EAR
is a unique homeopathic/herbal proprietary blend specifically formulated for the temporary relief of sinus and ear congestion related conditions.
CLEAR SINUS & EAR® is an exciting, all natural breakthrough for suffers of sinus & ear congestion, pain, pressure & tenderness, swimmer’s ear, cold & flu symptoms, and airborne allergies.
- All Natural Relief for:
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
CLEAR SINUS AND EAR
pulsatilla vulgaris, euphorbia resinifera resin, calcium sulfate and oyster shell calcium carbonate, crude capsuleProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68089-004 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PULSATILLA VULGARIS (UNII: I76KB35JEV) (PULSATILLA VULGARIS - UNII:I76KB35JEV) PULSATILLA VULGARIS 3 [hp_X] EUPHORBIA RESINIFERA RESIN (UNII: 1TI1O9028K) (EUPHORBIA RESINIFERA RESIN - UNII:1TI1O9028K) EUPHORBIA RESINIFERA RESIN 6 [hp_X] CALCIUM SULFATE (UNII: WAT0DDB505) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM SULFATE 3 [hp_X] OYSTER SHELL CALCIUM CARBONATE, CRUDE (UNII: 2E32821G6I) (OYSTER SHELL CALCIUM CARBONATE, CRUDE - UNII:2E32821G6I) OYSTER SHELL CALCIUM CARBONATE, CRUDE 3 [hp_X] Inactive Ingredients Ingredient Name Strength PUERARIA MONTANA VAR. LOBATA ROOT (UNII: PET93F4I3C) ANGELICA ATROPURPUREA ROOT (UNII: 885452N0GZ) PAEONIA LACTIFLORA ROOT (UNII: 3Z3866YW6P) PLATYCODON GRANDIFLORUS ROOT (UNII: 2DF0NS0O2Z) COIX LACRYMA-JOBI SEED (UNII: 4Q4V6NTZ1F) GINGER (UNII: C5529G5JPQ) PERILLA FRUTESCENS LEAF (UNII: T4L5881Y68) SAPOSHNIKOVIA DIVARICATA ROOT (UNII: 8H84LFK2QD) SCUTELLARIA BAICALENSIS ROOT (UNII: 7J95K7ID2S) CINNAMON (UNII: 5S29HWU6QB) LICORICE (UNII: 61ZBX54883) Product Characteristics Color brown (light brown, tannish) Score no score Shape CAPSULE Size 20mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68089-004-01 60 in 1 BOTTLE; Type 0: Not a Combination Product 11/15/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 08/01/2008 Labeler - Clear Products, Inc. (009605580) Establishment Name Address ID/FEI Business Operations HomeoCare Laboratories, Inc. 088248828 manufacture(68089-004)