Label: OHTRUST MOUTH AND NASAL RELIEF- hydroxide ions liquid
- NDC Code(s): 70970-007-01
- Packager: Nanoplus Life Biomedical Technology Co. Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated December 14, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient(s)
- Purpose
- Use
-
Warnings
- Avoid contact with eyes. In case of contact, rinse eyes thoroughly with water immediately.
- Use of this container by more than one person may spread infection.
- Temporary discomforts such as burning, stinging, sneezing or an increase in nasal discharge may occur.
- Stop use if rash or irritation develops. Do not drink it!
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
- Package Label - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
OHTRUST MOUTH AND NASAL RELIEF
hydroxide ions liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70970-007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROXIDE ION (UNII: 9159UV381P) (HYDROXIDE ION - UNII:9159UV381P) HYDROXIDE ION 0.2 g in 100 mL Inactive Ingredients Ingredient Name Strength PEPPERMINT OIL (UNII: AV092KU4JH) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70970-007-01 30 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 04/28/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 04/28/2022 Labeler - Nanoplus Life Biomedical Technology Co. Ltd. (657425219) Establishment Name Address ID/FEI Business Operations Nanoplus Life Biomedical Technology Co. Ltd. 657425219 manufacture(70970-007)


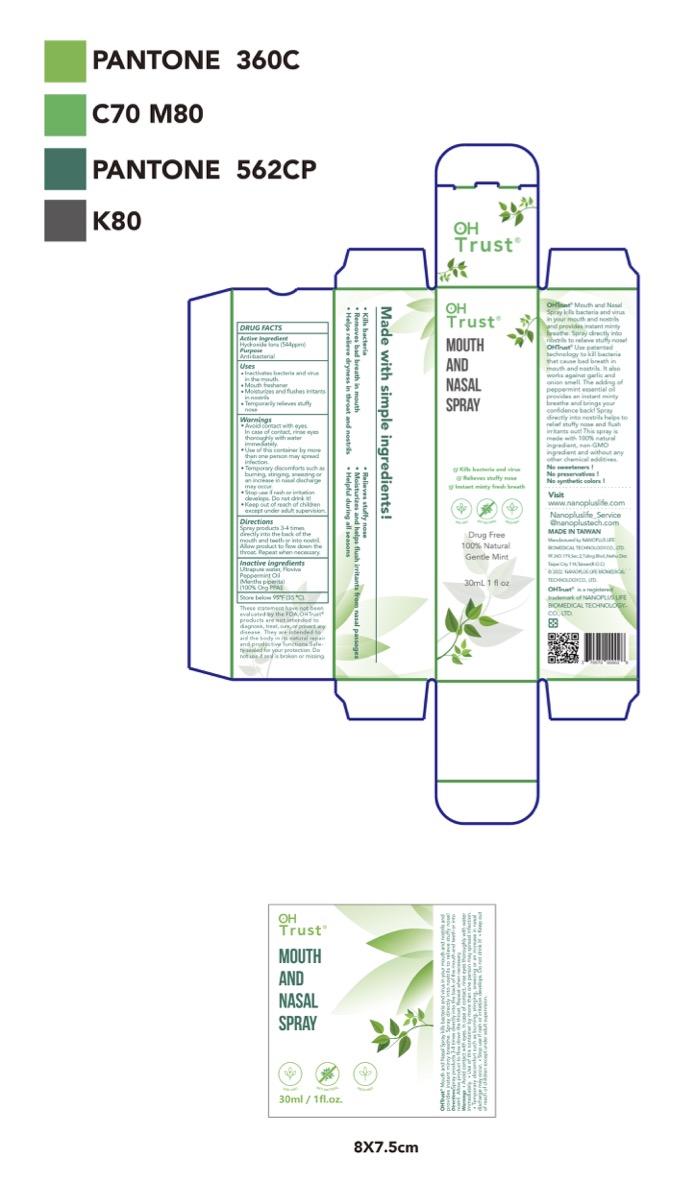 30 mL NDC: 70970-007-01
30 mL NDC: 70970-007-01