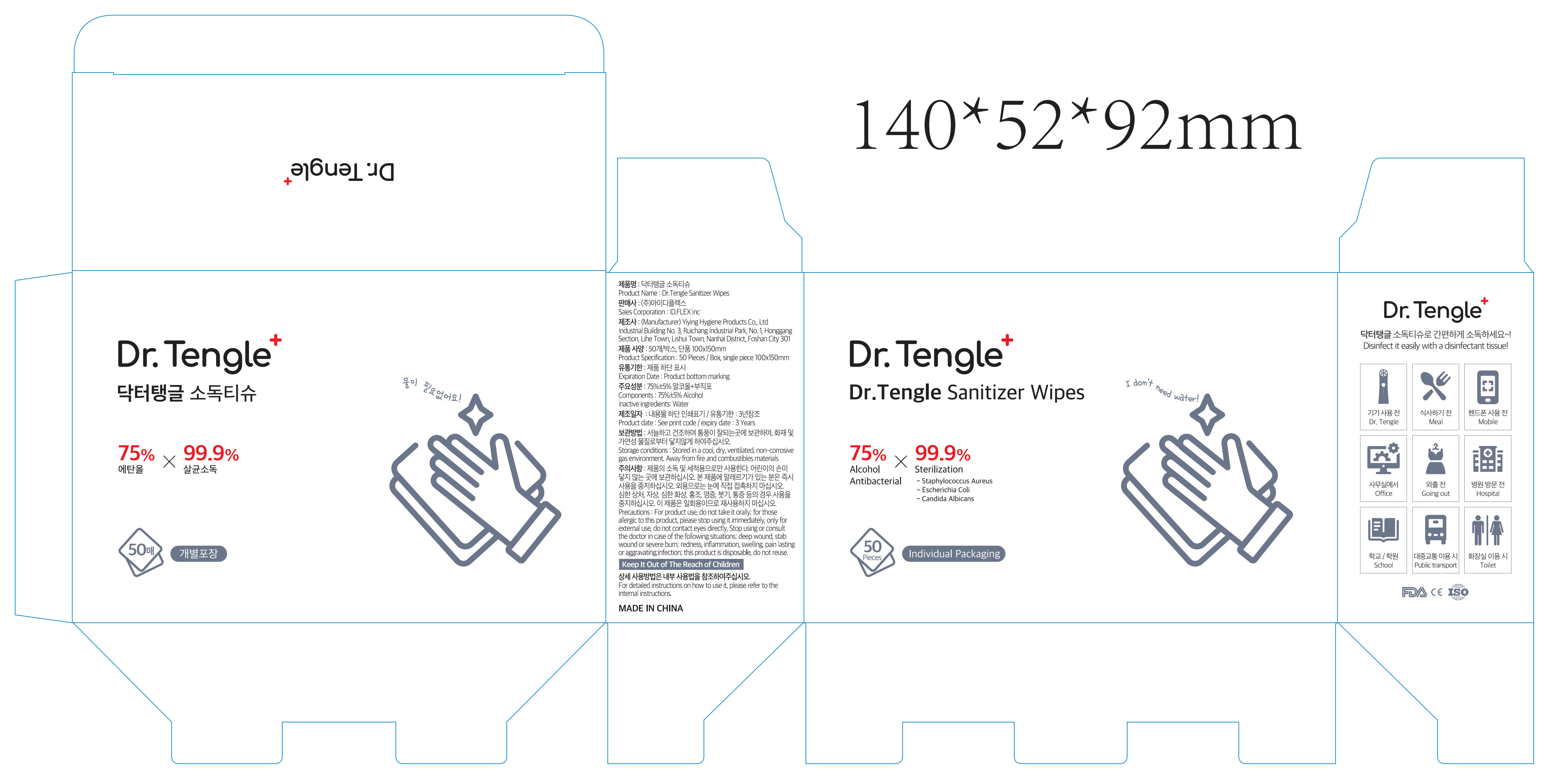
Label: DR.TENGLE SANITIZER WIPES- alcohol cloth
- NDC Code(s): 80139-182-01
- Packager: Foshan Yiying Hygiene Products Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 29, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
-
Precautions
For product use, do not take it orally; keep it out of the reach of children; for those allergic to this product, please stop using it immediately, only for external use, do not contact eyes directly, Stop using or consult the doctor in case of the following situations: deep wound, stab wound or severe burn; redness, inflammation, swelling, pain lasting or aggravation; inflection; this product is disposable, do not reuse.
- DO NOT USE
- When using this product
- Stop use and ask a doctor
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive Ingredient(s)
- Package Label - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
DR.TENGLE SANITIZER WIPES
alcohol clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80139-182 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 2.18 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80139-182-01 50 in 1 BOX; Type 0: Not a Combination Product 04/19/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 04/19/2022 Labeler - Foshan Yiying Hygiene Products Co., Ltd. (554544282) Registrant - Foshan Yiying Hygiene Products Co., Ltd. (554544282) Establishment Name Address ID/FEI Business Operations Foshan Yiying Hygiene Products Co., Ltd. 554544282 manufacture(80139-182)