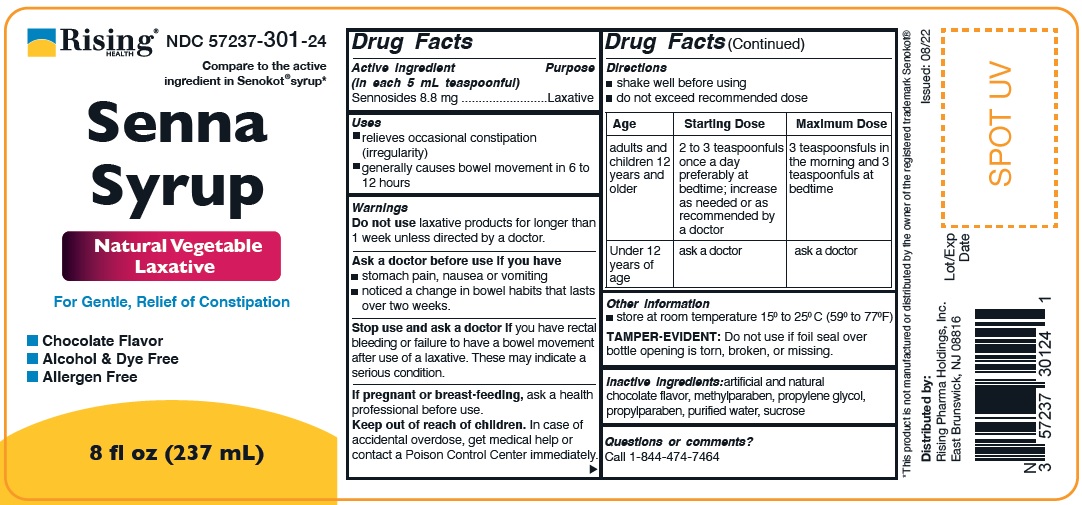
Label: SENNA SYRUP- sennosides liquid
- NDC Code(s): 57237-301-24
- Packager: Rising Health, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 20, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
Do not use laxative products for longer than 1 week unless directed by a doctor.
Ask a doctor before use if you have
- stomach pain, nausea or vomitting
- noticed a change in bowel habits that lasts over two weeks.
Stope use and ask a doctor if you have rectal bleeding or failure to have a bowel movement after use of laxative. These may indicate a serious condition.
- PREGNANCY
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions
- shake well before using
- Do not exceed recommended dose
Age Starting Dose Maximum Dose adults and children 12 years and older 2 to 3 teaspoonfuls once a day preferably at bedtime; increase as needed or as recommended by a doctor 3 teaspoonfuls in the morning and 3 teaspoonfuls at bedtime Under 12 years of age ask a doctor ask a doctor - STORAGE AND HANDLING
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SENNA SYRUP
sennosides liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57237-301 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SENNOSIDES (UNII: 3FYP5M0IJX) (SENNOSIDES - UNII:3FYP5M0IJX) SENNOSIDES 8.8 mg in 5 mL Inactive Ingredients Ingredient Name Strength METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SUCROSE (UNII: C151H8M554) Product Characteristics Color brown (Brown to dark brown) Score Shape Size Flavor CHOCOLATE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57237-301-24 237 mL in 1 BOTTLE; Type 0: Not a Combination Product 09/27/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part334 09/27/2022 Labeler - Rising Health, LLC (080500961) Establishment Name Address ID/FEI Business Operations Invahealth Inc. 116840615 analysis(57237-301) , label(57237-301) , manufacture(57237-301) , pack(57237-301)