Label: CAPLYTA- lumateperone capsule
-
NDC Code(s):
72060-110-07,
72060-110-40,
72060-121-07,
72060-121-40, view more72060-142-07, 72060-142-40
- Packager: Intra-Cellular Therapies, Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated November 14, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use CAPLYTA safely and effectively. See full prescribing information for CAPLYTA.
CAPLYTA® (lumateperone) capsules, for oral use
Initial U.S. Approval: 2019WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS; and SUICIDAL THOUGHTS AND BEHAVIORS
See full prescribing information for complete boxed warning.
- Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. CAPLYTA is not approved for the treatment of patients with dementia-related psychosis. (5.1)
- Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients. Closely monitor all antidepressant-treated patients for worsening and emergence of suicidal thoughts and behaviors. Safety and effectiveness of CAPLYTA have not been established in pediatric patients. (5.2, 8.4)
RECENT MAJOR CHANGES
Indications and Usage, adjunctive treatment of major depressive disorder (1) 11/2025
INDICATIONS AND USAGE
CAPLYTA is an atypical antipsychotic indicated for:
- Treatment of schizophrenia in adults. (1)
- Treatment of depressive episodes associated with bipolar I or II disorder (bipolar depression) in adults, as monotherapy and as adjunctive therapy with lithium or valproate. (1)
- Adjunctive therapy with antidepressants for the treatment of major depressive disorder (MDD) in adults. (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Capsules: 42 mg, 21 mg, 10.5 mg (3)
CONTRAINDICATIONS
CAPLYTA is contraindicated in patients with history of hypersensitivity reaction to lumateperone or any components of CAPLYTA. (4)
WARNINGS AND PRECAUTIONS
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia-Related Psychosis: Increased incidence of cerebrovascular adverse reactions (e.g., stroke and transient ischemic attack). (5.3)
- Neuroleptic Malignant Syndrome: If NMS is suspected, immediately discontinue CAPLYTA and provide intensive symptomatic treatment and monitoring. (5.4)
- Tardive Dyskinesia: If signs and symptoms of TD occur consider discontinuing CAPLYTA treatment. (5.5)
- Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia, and weight gain. (5.6)
- Leukopenia, Neutropenia, and Agranulocytosis: In patients with pre-existing low white blood cell count (WBC) or history of leukopenia or neutropenia, perform complete blood counts (CBC). Consider discontinuing CAPLYTA if clinically significant decline in WBC occurs in absence of other causative factors. Discontinue CAPLYTA in patients with clinically significant neutropenia or ANC < 1000/mm3 and monitor closely until the neutropenia resolve. (5.7)
- Orthostatic Hypotension and Syncope: Monitor heart rate and blood pressure in patients who are vulnerable to hypotension. (5.8)
- Seizures: Use cautiously in patients with a history of seizures or with other conditions that lower seizure threshold. (5.10)
- Potential for Cognitive and Motor Impairment: Use caution when operating machinery and driving a motor vehicle until patients are reasonably certain that therapy with CAPLYTA does not affect them adversely. (5.11)
ADVERSE REACTIONS
Most common adverse reactions in clinical trials (incidence > 5% and greater than twice placebo) were (6.1):
- Schizophrenia: somnolence/sedation and dry mouth.
- Bipolar depression: somnolence/sedation, dizziness, nausea, dry mouth.
- MDD: dizziness, dry mouth, somnolence/sedation, nausea, fatigue, diarrhea.
To report SUSPECTED ADVERSE REACTIONS, contact Intra-Cellular Therapies, Inc. at 1-888-611-4824 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
Pregnancy: May cause extrapyramidal and/or withdrawal symptoms in neonates with third trimester exposure. (8.1)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 11/2025
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS; and SUICIDAL THOUGHTS AND BEHAVIORS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Dosage Recommendations for Concomitant Use with Moderate or Strong CYP3A4 Inhibitors
2.3 Dosage Recommendations for Patients with Hepatic Impairment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Increased Mortality in Elderly Patients with Dementia-Related Psychosis
5.2 Suicidal Thoughts and Behaviors in Pediatric and Young Adult Patients
5.3 Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis
5.4 Neuroleptic Malignant Syndrome
5.5 Tardive Dyskinesia
5.6 Metabolic Changes
5.7 Leukopenia, Neutropenia, and Agranulocytosis
5.8 Orthostatic Hypotension and Syncope
5.9 Falls
5.10 Seizures
5.11 Potential for Cognitive and Motor Impairment
5.12 Body Temperature Dysregulation
5.13 Dysphagia
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Drugs Having Clinically Important Interactions with CAPLYTA
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Schizophrenia
14.2 Depressive Episodes Associated with Bipolar I or II Disorder (Bipolar Depression)
14.3 Adjunctive Treatment of Major Depressive Disorder in Adults
16 HOW SUPPLIED/ STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS; and SUICIDAL THOUGHTS AND BEHAVIORS
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. CAPLYTA is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.1)].
Suicidal Thoughts and Behaviors
Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adults in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.2)]. The safety and effectiveness of CAPLYTA have not been established in pediatric patients [see Use in Specific Populations (8.4)].
-
1 INDICATIONS AND USAGE
CAPLYTA is indicated for:
- Treatment of schizophrenia in adults [see Clinical Studies (14.1)].
- Treatment of depressive episodes associated with bipolar I or II disorder (bipolar depression) in adults, as monotherapy and as adjunctive therapy with lithium or valproate [see Clinical Studies (14.2)].
- Adjunctive therapy with antidepressants for the treatment of major depressive disorder (MDD) in adults [see Clinical Studies (14.3)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended CAPLYTA dosage is 42 mg administered orally once daily with or without food. Dose titration is not needed.
2.2 Dosage Recommendations for Concomitant Use with Moderate or Strong CYP3A4 Inhibitors
The recommended CAPLYTA dosage in patients who receive [see Drug Interactions (7.1)]:
- Strong CYP3A4 inhibitors is 10.5 mg once daily.
- Moderate CYP3A4 inhibitors is 21 mg once daily.
2.3 Dosage Recommendations for Patients with Hepatic Impairment
For patients with moderate hepatic impairment (HI) (Child-Pugh class B) or severe HI (Child-Pugh class C), the recommended CAPLYTA dosage is 21 mg once daily [see Use in Specific Populations (8.6)]. The recommended CAPLYTA dosage in patients with mild HI is the same as those with normal hepatic function.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. In an analysis of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, the risk of death in antipsychotic drug-treated patients was 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the incidence of death in antipsychotic-treated patients was about 4.5%, compared to an incidence of about 2.6% in placebo-treated patients. Although the causes of death varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia).
CAPLYTA is not approved for the treatment of patients with dementia-related psychosis [see Indications and Usage (1)].
5.2 Suicidal Thoughts and Behaviors in Pediatric and Young Adult Patients
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and 4,500 pediatric patients, the incidence of suicidal thoughts and behaviors in antidepressant-treated pediatric and young adult patients was greater than in placebo-treated patients. There were differences in absolute risk of suicidal thoughts and behaviors across the different uses, with the highest incidence in patients with MDD. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1,000 patients treated are provided in Table 1.
Table 1: Risk Differences of the Number of Patients with Suicidal Thoughts and Behaviors in the Pooled Placebo-Controlled Trials of Antidepressants in Pediatric* and Adult Patients Age Range Drug-Placebo Difference in Number of Patients of Suicidal Thoughts
or Behaviors per 1000 Patients Treated
Increases Compared to Placebo <18 years old 14 additional patients 18-24 years old 5 additional patients Decreases Compared to Placebo 25-64 years old 1 fewer patient >65 years old 6 fewer patients *CAPLYTA is not approved for use in pediatric patients.
It is unknown whether the risk of suicidal thoughts and behaviors in pediatric and young adult patients extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that antidepressants delay the recurrence of depression and that depression itself is a risk factor for suicidal thoughts and behaviors.
Monitor all antidepressant-treated patients, especially during the initial few months of anti-depressant drug therapy, and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the health care provider. Consider changing the therapeutic regimen, including possibly discontinuing CAPLYTA, in patients whose depression is persistently worse, or who experience suicidal thoughts or behaviors.
5.3 Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis
In placebo-controlled trials, elderly patients with dementia-related psychosis treated with antipsychotics had a higher incidence of stroke and transient ischemic attack, including fatal stroke compared to those treated with placebo.
CAPLYTA is not approved for the treatment of patients with dementia-related psychosis [see Indications and Usage (1)].
5.4 Neuroleptic Malignant Syndrome
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex, has been reported in association with administration of antipsychotic drugs. Clinical manifestations of NMS include hyperpyrexia, muscle rigidity, delirium, and autonomic instability. Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, immediately discontinue CAPLYTA and provide intensive symptomatic treatment and monitoring.
5.5 Tardive Dyskinesia
Tardive dyskinesia (TD) may develop in patients treated with antipsychotic drugs, including CAPLYTA. TD can develop after a relatively brief treatment period at low dosages and may also occur after discontinuation of treatment. If antipsychotic treatment is discontinued, TD may partially or completely remit. Antipsychotic treatment, however, may suppress or partially suppress the signs and symptoms of TD, and may mask the underlying process. The effect that symptomatic suppression has upon the long-term course of TD is unknown.
The TD risk appears to be highest among the elderly, especially elderly women, but it is not possible to predict which patients are likely to develop TD. The TD risk and the likelihood that TD will become irreversible increase with the duration of antipsychotic drug treatment and the cumulative dosage.
Periodically reassess the need for continued treatment. If signs and symptoms of TD appear in CAPLYTA-treated patients, consider drug discontinuation. However, some patients may require CAPLYTA treatment despite the presence of TD.
5.6 Metabolic Changes
Antipsychotic drugs have caused metabolic changes, including hyperglycemia, diabetes mellitus, dyslipidemia, and weight gain.
Hyperglycemia and Diabetes Mellitus
Hyperglycemia, in some cases extreme and associated with ketoacidosis, hyperosmolar coma or death, has been reported in patients treated with antipsychotics. There have been reports of hyperglycemia in patients treated with CAPLYTA. Assess fasting plasma glucose before or soon after initiation of CAPLYTA and monitor periodically during long-term treatment.
- In pooled data from short-term (4- to 6-week), placebo-controlled trials of adult patients with schizophrenia, mean changes from baseline and the proportion of patients with shifts from normal to greater than normal levels of fasting glucose were similar in CAPLYTA-treated and placebo-treated patients. In an uncontrolled open-label trial of CAPLYTA for up to 1 year in adult patients with stable schizophrenia, the percentages of patients with shifts in fasting glucose and insulin values from normal to high were 8% and 12%, respectively. In this trial, 5% of CAPLYTA-treated patients with normal hemoglobin A1c (<6.5%) at baseline developed elevated levels (≥6.5%) post-baseline.
- In data from short-term (6-week), placebo-controlled monotherapy and adjunctive therapy bipolar depression trials, mean changes from baseline and the proportion of patients with shifts from normal to greater than normal levels of fasting glucose and insulin were similar in CAPLYTA-treated and placebo-treated patients.
- In pooled data from short-term (6-week), placebo-controlled adjunctive therapy MDD trials, mean changes from baseline and the proportion of patients with shifts from normal to greater than normal levels of fasting glucose were similar in CAPLYTA-treated and placebo-treated patients.
Dyslipidemia
Antipsychotics have caused adverse alterations in lipids. Before or soon after initiation of antipsychotic medications, obtain a fasting lipid profile at baseline and monitor periodically during treatment.
- In pooled data from short-term (4- to 6-week), placebo-controlled trials of adult patients with schizophrenia, mean changes from baseline and the proportion of patients with shifts to higher levels of fasting total cholesterol and triglycerides were similar in CAPLYTA-treated and placebo-treated patient. In an uncontrolled open-label trial of CAPLYTA for up to 1 year in patients with stable schizophrenia, the percentages of patients with a shift from normal to high were 8%, 5%, and 4% for total cholesterol, triglycerides, and LDL cholesterol, respectively.
- In data from short-term (6-week), placebo-controlled monotherapy and adjunctive therapy bipolar depression trials, mean changes from baseline and the proportion of patients with shifts to higher levels of fasting total cholesterol and triglycerides were similar in CAPLYTA-treated and placebo-treated patients. In an uncontrolled open-label trial of CAPLYTA for up to 6 months in patients with bipolar depression, the proportion of patients with a shift from normal to high were 10%, 5%, and 2% for total cholesterol, triglycerides, and LDL cholesterol, respectively.
- In pooled data from short-term (6-week), placebo-controlled adjunctive therapy MDD trials, mean changes from baseline and the proportion of patients with shifts to higher levels of fasting total cholesterol and triglycerides were similar in CAPLYTA-treated and placebo-treated patients.
Weight Gain
Weight gain has been observed with use of antipsychotics. Monitor weight at baseline and frequently thereafter.
- In pooled data from placebo-controlled trials of adult patients with schizophrenia, mean changes from baseline and the proportion of patients with an increase in weight ≥7% from baseline to end of study was similar in CAPLYTA-treated and placebo-treated patients. In an uncontrolled open-label trial of CAPLYTA for up to one year in patients with stable schizophrenia, the mean change in body weight was approximately -2 kg (SD 5.6) at Day 175 and approximately - 3.2 kg (SD 7.4) at Day 350.
- In data from short-term (6-week), placebo-controlled monotherapy and adjunctive therapy bipolar depression trials, mean changes from baseline and the proportion of patients with an increase in weight ≥7% from baseline to end of study were similar in CAPLYTA-treated and placebo-treated patients. In an uncontrolled open-label trial of CAPLYTA for up to 6 months in patients with bipolar depression, the mean change in body weight was -0.01 kg (SD 3.1) at Day 175.
- In pooled data from short-term (6-week), placebo-controlled adjunctive therapy MDD trials, mean changes from baseline and the proportion of patients with an increase in weight ≥7% from baseline to end of study were similar in CAPLYTA-treated and placebo-treated patients.
- In a long-term open-label adjunctive therapy MDD trial of CAPLYTA for up to 6 months, the mean change in body weight was -0.16 kg (SD 3.7) at Week 26.
5.7 Leukopenia, Neutropenia, and Agranulocytosis
Leukopenia and neutropenia have been reported during treatment with antipsychotic drugs, including CAPLYTA. Agranulocytosis (including fatal cases) has been reported with other antipsychotic drugs.
Possible risk factors for antipsychotic drug-leukopenia and neutropenia include pre-existing low white blood cell count (WBC) or absolute neutrophil count (ANC) and history of drug-induced leukopenia or neutropenia.
In patients with a pre-existing low WBC or ANC or a history of drug-induced leukopenia or neutropenia, perform complete blood count (CBC) monitoring during the first few months of CAPLYTA therapy. Consider discontinuing CAPLYTA in patients who have a clinically significant decline in WBC in the absence of other causative factors. Discontinue CAPLYTA in patients with clinically significant neutropenia or ANC < 1000/mm3 and monitor closely until the neutropenia resolves.
5.8 Orthostatic Hypotension and Syncope
Atypical antipsychotics cause orthostatic hypotension and syncope. Generally, the risk is greatest during initial dose administration.
Orthostatic vital signs should be monitored in patients who are vulnerable to hypotension (e.g., elderly patients, patients with dehydration, hypovolemia, and concomitant treatment with antihypertensive drugs), patients with known cardiovascular disease (history of myocardial infarction, ischemic heart disease, heart failure, or conduction abnormalities), and patients with cerebrovascular disease. CAPLYTA has not been evaluated in patients with a recent history of myocardial infarction or unstable cardiovascular disease. Such patients were excluded from pre-marketing clinical trials.
- In pooled data from short-term (4- to 6-week), placebo-controlled schizophrenia trials, the frequencies of orthostatic hypotension in CAPLYTA-treated and placebo-treated patients were 0.7% and 0%, respectively. The incidence of syncope for CAPLYTA-treated and placebo-treated patients were 0.2% and 0.2%.
- In data from short-term (6-week), placebo-controlled monotherapy and adjunctive therapy bipolar depression trials, the frequencies of orthostatic hypotension for CAPLYTA-treated and placebo-treated patients were both 0%. The incidence of syncope for CAPLYTA and placebo were 0.3% and 0.5%, respectively in the monotherapy trials, and there were no reports of syncope in the adjunctive therapy trial.
- In pooled data from short-term (6-week), placebo-controlled adjunctive therapy MDD trials, the frequencies of orthostatic hypotension for CAPLYTA-treated and placebo-treated patients were 6.6% and 6.2%, respectively. The incidence of syncope for CAPLYTA-treated and placebo-treated patients were 0.2% and 0%, respectively.
5.9 Falls
Antipsychotics, including CAPLYTA, may cause somnolence, postural hypotension, and motor and sensory instability, which may lead to falls and, consequently, fractures and other injuries.
If patients have a condition (or take concomitant drugs) that could exacerbate these effects, complete fall risk assessments when initiating CAPLYTA treatment and periodically during long-term treatment.
5.10 Seizures
Like other antipsychotic drugs, CAPLYTA may cause seizures. The risk of antipsychotic drug-associated seizures is greatest in patients with a history of seizures or with conditions that lower the seizure threshold. Conditions that lower the seizure threshold may be more prevalent in older patients.
Use CAPLYTA cautiously in patients with a history of seizures or with other conditions that lower seizure threshold.
5.11 Potential for Cognitive and Motor Impairment
CAPLYTA, like other antipsychotics, may cause somnolence and has the potential to impair judgment, thinking, and motor skills. Patients should be cautioned about operating hazardous machinery and motor vehicles until they are reasonably certain that therapy with CAPLYTA does not affect them adversely.
- In short-term (i.e., 4- to 6-week), placebo-controlled clinical trials of patients with schizophrenia, somnolence and sedation were reported in 24% of CAPLYTA-treated patients, compared to 10% of placebo-treated patients.
- In short term (6-week), placebo-controlled monotherapy and adjunctive therapy bipolar depression clinical trials, somnolence and sedation were reported in 13% of CAPLYTA-treated patients, compared to 3% of placebo-treated patients.
- In short term (6-week), placebo-controlled adjunctive therapy MDD trials, somnolence and sedation were reported in 12% of CAPLYTA-treated patients, compared to 2% of placebo-treated patients.
5.12 Body Temperature Dysregulation
Atypical antipsychotics may disrupt the body’s ability to reduce core body temperature. Strenuous exercise, exposure to extreme heat, dehydration, and anticholinergic drugs may contribute to an elevation in core body temperature. Use CAPLYTA with caution in patients who may experience these conditions.
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed in detail in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia-Related Psychosis [see Boxed Warning, Warnings and Precautions (5.1)]
- Suicidal Thoughts and Behaviors [see Boxed Warning, Warnings and Precautions (5.2)]
- Cerebrovascular Adverse Reactions, Including Stroke, in Elderly Patients with Dementia-related Psychosis [see Warnings and Precautions (5.3)]
- Neuroleptic Malignant Syndrome [see Warnings and Precautions (5.4)]
- Tardive Dyskinesia [see Warnings and Precautions (5.5)]
- Metabolic Changes [see Warnings and Precautions (5.6)]
- Leukopenia, Neutropenia, and Agranulocytosis [see Warnings and Precautions (5.7)]
- Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.8)]
- Falls [see Warnings and Precautions (5.9)]
- Seizures [see Warnings and Precautions (5.10)]
- Potential for Cognitive and Motor Impairment [see Warnings and Precautions (5.11)]
- Body Temperature Dysregulation [see Warnings and Precautions (5.12)]
- Dysphagia [see Warnings and Precautions (5.13)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of CAPLYTA has been evaluated in placebo-controlled clinical trials that included 3575 adult patients with schizophrenia, bipolar depression, or major depressive disorder, exposed to one or more CAPLYTA doses. A total of 852 CAPLYTA-treated patients had at least 6 months of treatment and 108 had at least 1 year of treatment with the 42-mg once daily dosage.
Adverse Reactions in Patients with Schizophrenia
The following adverse reactions are based on the pooled short-term (4- to 6-week), placebo-controlled studies in adult patients with schizophrenia in which CAPLYTA was administered at a dosage of 42 mg once daily (N=406) [see Clinical Studies (14.1)].
There was no single adverse reaction that led to discontinuation that occurred at a rate of >2% in CAPLYTA-treated patients. The most common adverse reactions (incidence of at least 5% of CAPLYTA-treated patients and greater than twice the rate of placebo-treated patients) were somnolence/sedation and dry mouth. Adverse reactions (incidence of at least 2% in CAPLYTA-treated patients and greater than in placebo-treated patients) are shown in Table 2.
Table 2: Adverse Reactions Reported in ≥2% of CAPLYTA-Treated Patients and Greater Incidence Than in Placebo-Treated Patients in 4- to 6-week Schizophrenia Trials CAPLYTA
42 mg
(N=406)Placebo
(N=412)Somnolence/Sedation 24% 10% Nausea 9% 5% Dry Mouth 6% 2% Dizziness1 5% 3% Creatine Phosphokinase Increased 4% 1% Fatigue 3% 1% Vomiting 3% 2% Hepatic Transaminases Increased2 2% 1% Decreased Appetite 2% 1% 1 Dizziness, dizziness postural
2 ALT, AST, “hepatic enzymes” increased, or liver function test abnormalAdverse Reactions in Patients with Bipolar Depression (CAPLYTA Monotherapy)
The following adverse reactions are based on the pooled short-term (6-week), placebo-controlled monotherapy bipolar depression studies in adult patients treated with CAPLYTA 42 mg once daily (N=372) [see Clinical Studies (14.2)].
There was no single adverse reaction leading to discontinuation that occurred at a rate of >2% in CAPLYTA-treated patients.
The most common adverse reactions (incidence of at least 5% of CAPLYTA-treated patients and greater than twice the rate in placebo-treated patients) were somnolence/sedation, dizziness, nausea, and dry mouth.
Adverse reactions associated with CAPLYTA (incidence of at least 2% in CAPLYTA-treated patients and greater than placebo-treated patients) are shown in Table 3.
Table 3: Adverse Reactions Reported in ≥2% of CAPLYTA-Treated Patients and Greater Incidence than in Placebo-Treated Patients in Pooled 6-week Monotherapy Bipolar Depression Trials CAPLYTA
42 mg
(N=372)Placebo
(N=374)Headache 14% 8% Somnolence/Sedation 13% 3% Dizziness1 8% 4% Nausea 8% 3% Dry mouth 5% 1% Diarrhea 4% 2% Vomiting 4% 0% Abdominal pain2 2% 1% Upper respiratory tract infection 2% 1% 1 Dizziness, dizziness postural
2 Abdominal discomfort, abdominal pain, abdominal pain upper and lowerAdverse Reactions in Patients with Bipolar Depression (Concomitant Treatment with CAPLYTA and Lithium or Valproate)
The adverse reactions below are based on a 6-week, placebo-controlled adjunctive therapy bipolar depression study in adult patients treated with CAPLYTA 42 mg once daily + lithium or valproate OR placebo + lithium or valproate (N=177) [see Clinical Studies (14.2)].
There was no single adverse reaction leading to discontinuation that occurred at a rate of >2% in patients in the CAPLYTA + lithium or valproate group.
The most common adverse reactions (incidence of at least 5% in patients treated with CAPLYTA + lithium or valproate and greater than twice the rate of patients treated with placebo + lithium or valproate) were somnolence/sedation, dizziness, nausea, and dry mouth.
Adverse reactions associated with CAPLYTA + lithium or valproate (incidence of at least 2% in patients treated with CAPLYTA + lithium or valproate and greater than with patients treated with placebo + lithium or valproate) are shown in Table 4.
Table 4: Adverse Reactions Reported in ≥2% of CAPLYTA-Treated Patients and that Occurred at Greater Incidence than in the Placebo-Treated Patients in a 6-Week Adjunctive Therapy Bipolar Depression Trial CAPLYTA
42 mg + lithium
or valproate
(N=177)Placebo + lithium
or valproate
(N=175)Somnolence/Sedation 13% 3% Dizziness1 11% 2% Nausea 9% 4% Dry mouth 5% 1% Vomiting 4% 0% Diarrhea 3% 2% Upper respiratory tract infection 3% 1% Blurred vision 3% 1% Increased blood prolactin 2% 0% 1 Dizziness, dizziness postural
Adverse Reactions in Studies for Adjunctive Treatment of Major Depressive Disorder
The following adverse reactions are based on pooled short-term (6-week), placebo-controlled adjunctive therapy MDD studies in adult patients treated with CAPLYTA 42 mg orally once daily and background antidepressant therapy (ADT) or placebo and background ADT (N=483) [see Clinical Studies (14.3)].
There was no single adverse reaction leading to discontinuation that occurred at an incidence of >2% in patients treated with CAPLYTA and background ADT.
The most common adverse reactions (incidence of at least 5% of patients treated with CAPLYTA and background ADT and greater than twice the incidence in patients treated with placebo and background ADT) were dizziness, dry mouth, somnolence/sedation, nausea, fatigue, and diarrhea.
Adverse reactions in the adjunctive therapy MDD studies (incidence of at least 2% of patients treated with CAPLYTA and background ADT and greater than in patients treated with placebo and background ADT) are shown in Table 5.
Table 5: Adverse Reactions Reported in ≥2% of Patients Treated with CAPLYTA + Background ADT and at Greater Incidence than in Patients Treated with Placebo + Background ADT in Pooled 6-Week Adjunctive MDD Trials CAPLYTA
42 mg + ADT
(N=483)Placebo + ADT
(N=481)Headache1 19% 13% Dizziness2 17% 5% Dry Mouth 13% 3% Somnolence/Sedation 12% 2% Nausea 9% 4% Fatigue 8% 2% Diarrhea 5% 1% Tremor 4% <1% Vomiting 3% 2% Vertigo 3% <1% Insomnia 3% 2% 1Headache, migraine, tension headache
2Dizziness, postural dizzinessAdditional Adverse Reactions
Dystonia: Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. Although these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and higher doses of first-generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
Extrapyramidal Symptoms: In the short-term, placebo-controlled schizophrenia, bipolar depression, and adjunctive MDD studies, extrapyramidal (EPS) symptom data was objectively collected on the Simpson-Angus Scale (SAS) for EPS (total score ranges from 0 to 40), the Barnes Akathisia Rating Scale (BARS) for akathisia (total score ranges from 0 to 14), and the Abnormal Involuntary Movement Scale (AIMS) for dyskinesia (total score ranges from 0 to 28).
- In the 4- to 6-week, placebo-controlled schizophrenia trials, the frequency of EPS-related reported events including akathisia, extrapyramidal disorder, muscle spasms, restlessness, musculoskeletal stiffness, dyskinesia, dystonia, muscle twitching, tardive dyskinesia, tremor, drooling, and involuntary muscle contractions was 6.7% for the CAPLYTA-treated patients and 6.3% for placebo-treated patients. In these trials, the mean changes from baseline for CAPLYTA-treated patients and placebo-treated patients were 0.1 and 0 for the SAS, -0.1 and 0 for the BARS, and 0.1 and 0 for the AIMS, respectively.
- In the 6-week, monotherapy bipolar depression trials, the frequency of reported EPS-related reactions including muscle spasms, dyskinesia, extrapyramidal disorder, movement disorder, tremor, restlessness, and akathisia was 1.3% for CAPLYTA-treated patients and 1.1% for placebo-treated patients. In these trials, the mean changes from baseline for CAPLYTA-treated patients and placebo-treated patients were 0 and 0 for the SAS, -0.1 and -0.1 for the BARS, and 0 and 0 for the AIMS, respectively.
- In a 6-week, adjunctive therapy bipolar depression trial, the frequency of reported EPS-related reactions, including tremor, muscle spasms, akathisia, extrapyramidal disorder, gait disturbance, and restlessness was 4% for CAPLYTA-treated patients and 2.3% for placebo-treated patients. In the 6-week, monotherapy bipolar depression trials, the mean changes from baseline for CAPLYTA-treated patients and placebo-treated patients were 0 and 0 for the SAS, -0.1 and -0.1 for the BARS, and 0 and 0 for the AIMS, respectively. In this trial, the mean changes from baseline for CAPLYTA-treated patients and placebo-treated patients were 0 and 0 for the SAS, 0 and -0.1 for the BARS, and 0 and 0 for the AIMS, respectively.
- In the 6-week, adjunctive MDD trials, the frequency of reported EPS-related adverse reactions (tremor, bradykinesia, muscle spasms, gait disturbance, tongue spasm, muscle tightness and dyskinesia), excluding akathisia and restlessness, was 5% for CAPLYTA-treated patients and 0.8% for placebo-treated patients. The combined incidence of akathisia and restlessness was 1% for CAPLYTA-treated patients and 0.8% for placebo-treated patients. In these trials, the mean changes from baseline for CAPLYTA-treated patients and placebo-treated patients were 0 and 0 for the SAS, 0 and -0.1 for the BARS, and 0 and 0 for the AIMS, respectively.
6.2 Postmarketing Experience
The following adverse reaction has been identified during post-approval use of CAPLYTA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure.
Central and Peripheral Nervous System Disorders: burning sensation, including skin burning sensation
-
7 DRUG INTERACTIONS
7.1 Drugs Having Clinically Important Interactions with CAPLYTA
Clinically important drug interactions with CAPLYTA are presented in Table 6.
Table 6: Clinically Important Drug Interactions with CAPLYTA CYP3A4 Inducers* Prevention or Management Avoid concomitant use of CAPLYTA with CYP3A4 inducers. Clinical Impact Concomitant use of CAPLYTA with CYP3A4 inducers decreases the exposure of lumateperone [see Clinical Pharmacology (12.3)]. Moderate or Strong CYP3A4 Inhibitors* Prevention or Management Reduce the CAPLYTA dosage when used concomitantly with moderate or strong CYP3A4 inhibitors [see Dosage and Administration (2.2)]. Clinical Impact Concomitant use of CAPLYTA with moderate or strong CYP3A4 inhibitors increases lumateperone exposure [see Clinical Pharmacology (12.3)], which may increase the risk of adverse reactions. Serotonin Reuptake Inhibitors Prevention or Management Increased monitoring for SRI- associated adverse reactions is recommended. Clinical Impact Although no clinically significant drug interactions with adjunctive SSRI/SNRIs in MDD were observed in CAPLYTA clinical trials, CAPLYTA’s moderate serotonin transporter (SERT) activity may increase the risk of SRI-associated adverse reactions (e.g., serotonin syndrome, hyponatremia). * See www.fda.gov/CYPandTransporterInteractingDrugs for examples of CYP3A4 Inducers and Moderate or Strong CYP3A4 Inhibitors
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to atypical antipsychotics, including CAPLYTA, during pregnancy. Healthcare providers are encouraged to advise patients to register by calling the National Pregnancy Registry for Atypical Antipsychotics at 1-866-961-2388 or visiting online at https://womensmentalhealth.org/research/pregnancyregistry/.
Risk Summary
Neonates exposed to antipsychotic drugs during the third trimester are at risk for extrapyramidal and/or withdrawal symptoms following delivery (see Clinical Considerations). Available data from case reports on CAPLYTA use in pregnant women are insufficient to establish any drug associated risks for birth defects, miscarriage, or adverse maternal or fetal outcomes. There are risks to the mother associated with untreated schizophrenia and with exposure to antipsychotics, including CAPLYTA, during pregnancy (see Clinical Considerations). In animal reproduction studies, no malformations were observed with oral administration of lumateperone to pregnant rats and rabbits during organogenesis at doses up to 2.4 and 9.7 times, respectively, the maximum recommended human dose (MRHD) of 42 mg/day on a mg/m2 basis. When pregnant rats were administered lumateperone during the period of organogenesis through lactation, the number of perinatal deaths of pups was increased at 4.9 times the MRHD, with no adverse effects on pups at 2.4 times the MRHD (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease Associated Maternal and/or Embryo/fetal Risk: There is risk to the mother from untreated schizophrenia, including increased risk of relapse, hospitalization, and suicide. Schizophrenia is associated with increased adverse perinatal outcomes, including preterm birth. It is not known if this is a direct result of the illness or other comorbid factors.
Fetal/neonatal Adverse Reactions: Extrapyramidal and/or withdrawal symptoms, including agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress, and feeding disorder have been reported in neonates who were exposed to antipsychotic drugs during the third trimester of pregnancy. These symptoms have varied in severity. Monitor neonates for extrapyramidal and/or withdrawal symptoms and manage symptoms appropriately. Some neonates recovered within hours or days without specific treatment; others required prolonged hospitalization.
Data
Animal Data
Pregnant rats were treated with oral doses of 3.5, 10.5, 21, and 63 mg/kg/day lumateperone (0.8, 2.4, 4.9, and 14.6 times the MRHD on a mg/m2 basis) during the period of organogenesis. No malformations were observed with lumateperone at doses up to 2.4 times the MRHD. Findings of decreased body weight were observed in fetuses at 4.9 and 14.6 times the MRHD. Findings of incomplete ossification and increased incidences of visceral and skeletal variations were recorded in fetuses at 14.6 times the MRHD, a dose that induced maternal toxicity.
Pregnant rabbits were treated with oral doses of 2.1, 7, and 21 mg/kg/day lumateperone (1.0, 3.2, and 9.7 times the MRHD on a mg/m2 basis) during the period of organogenesis. Lumateperone did not cause adverse developmental effects at doses up to 9.7 times the MRHD.
In a study in which pregnant rats were administered oral doses of 3.5, 10.5, and 21 mg/kg/day lumateperone (0.8, 2.4, and 4.9 times the MRHD on a mg/m2 basis) during the period of organogenesis and through lactation, the number of live-born pups was decreased at 2.4 and 4.9 times the MRHD, and early postnatal deaths increased at a dose 4.9 times the MRHD. Impaired nursing and decreased body weight gain in pups were observed at 4.9 times, but not at 2.4 times, the MRHD.
Pregnant rats were treated with a human metabolite of lumateperone (reduced ketone metabolite) at oral doses of 15, 60, and 100 mg/kg/day (1.2, 19, and 27 times the exposure to this metabolite at the MRHD of lumateperone based on AUC plasma exposure) during the period of organogenesis. This metabolite did not cause adverse developmental effects at a dose 1.2 times the exposure at the MRHD of lumateperone; however, it caused an increase in visceral malformations (cleft palate) at 27 times and skeletal malformations at 19 times the exposure at the MRHD of lumateperone, a dose that induced maternal toxicity.
8.2 Lactation
Risk Summary
Lumateperone and its metabolites are present in human breast milk in low amounts. In a clinical lactation study, lumateperone was detected in human milk at an estimated daily infant dose 0.0004 mg/kg, with a relative infant dose (RID) of 0.06% the maternal weight-adjusted dosage. Several major circulating metabolites were similarly detected in breast milk in low amounts; however, aniline metabolites were not present in milk or maternal plasma at quantifiable levels (see Data). There are no data on the effects of lumateperone on the breastfed infant or the effects on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for CAPLYTA and any potential adverse effects on the breastfed child from CAPLYTA or from the underlying maternal condition.
Data
A lactation study in 17 lactating women evaluated the concentrations of lumateperone and its metabolites in plasma and mature breast milk following a single dose of 42 mg CAPLYTA. The estimated daily infant dose of lumateperone in human milk was 0.0004 mg/kg/day (with assumed average milk consumption of 200 ml/kg/day). The mean relative infant dose (RID) (with assumed mean milk consumption of 200 mL/kg/day and average maternal weight of 71 kg) was 0.06% of the maternal weight-adjusted dosage. Several major circulating metabolites were also present in breast milk at estimated daily infant dose of 0.0004 mg/kg/day. Aniline metabolites were not present in milk or maternal plasma at quantifiable levels.
8.3 Females and Males of Reproductive Potential
Infertility
Based on findings from animal studies, lumateperone may impair male and female fertility [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
Safety and effectiveness of CAPLYTA have not been established in pediatric patients.
Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric patients [see Boxed Warning, Warnings and Precautions (5.2)].
8.5 Geriatric Use
Controlled clinical studies of CAPLYTA for the treatment of schizophrenia and as adjunctive therapy with antidepressants for the treatment of MDD did not include any patients aged 65 or older to determine whether or not they respond differently from younger adult patients.
Among the CAPLYTA-treated patients in clinical studies for the treatment of depressive episodes associated with bipolar depression, 20 (6%) were 65 to 74 years of age, and none were 75 years of age or older [see Clinical Studies (14.2)]. These clinical studies did not include sufficient numbers of patients aged 65 years of age or older to determine whether or not they respond differently from younger adult patients.
Antipsychotic drugs increase the risk of death in elderly patients with dementia-related psychosis. CAPLYTA is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions (5.1) and (5.3)].
Elderly patients with dementia-related psychosis treated with antipsychotics have an increased risk of cerebrovascular adverse reactions (e.g., stroke, transient ischemic attack) including fatalities, compared to those treated with placebo [see Warnings and Precautions (5.1)].
Antipsychotic drugs increase the risk of tardive dyskinesia and this risk appears to be highest among the elderly, particularly elderly women [see Warnings and Precautions (5.5)].
The use of serotonin reuptake inhibitors (SRIs) has been associated with clinically significant hyponatremia in geriatric patients, who may be at greater risk for this adverse reaction. The concomitant use of CAPLYTA with an SRI may increase this risk [see Drug Interactions (7.1)].
8.6 Hepatic Impairment
Patients with moderate hepatic impairment (HI) (Child-Pugh class B) and severe HI (Child-Pugh class C) generally had higher exposure to lumateperone than patients with normal hepatic function; therefore, the recommended CAPLYTA dosage in patients with moderate or severe HI is lower than those with normal hepatic function [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
The recommended dosage in patients with mild HI (Child-Pugh class A) is the same as those with normal hepatic function.
-
10 OVERDOSAGE
No specific antidotes for CAPLYTA are known. In managing an CAPLYTA overdose, provide supportive care, including close medical supervision and monitoring and consider the possibility of multiple drug involvement. Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
-
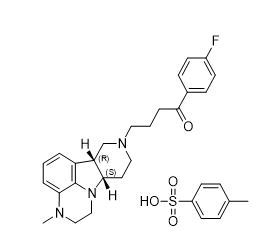
11 DESCRIPTION
Lumateperone is an atypical antipsychotic present as lumateperone tosylate salt with the chemical name 4-((6bR,10aS)-3-methyl-2,3,6b,9,10,10a-hexahydro-1H,7H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxalin-8-yl)-1-(4-fluoro-phenyl)-butan-1-one 4-methylbenzenesulfonate. Its molecular formula is C31H36FN3O4S, and its molecular weight is 565.71 g/mol with the following structure:
CAPLYTA (lumateperone) capsules are for oral administration. Each capsule contains:
- 42 mg of lumateperone (equivalent to 60 mg of lumateperone tosylate), or
- 21 mg of lumateperone (equivalent to 30 mg of lumateperone tosylate), or
- 10.5 mg of lumateperone (equivalent to 15 mg of lumateperone tosylate).
The capsules include the following inactive ingredients: croscarmellose sodium, gelatin, magnesium stearate, mannitol, and talc. Colorants include FD&C blue #1 and red #3 (42 mg), FDA/E172 black iron oxide, FDA/E172 red iron oxide and FD&C red #3 (10.5 mg), and titanium dioxide (42 mg, 21 mg and 10.5 mg).
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism of action of lumateperone for the treatment of schizophrenia in adults, for the treatment of depressive episodes associated with bipolar depression (as monotherapy or as adjunctive therapy with lithium or valproate), and as adjunctive therapy with antidepressants for the treatment of MDD is unknown. However, the mechanism of action of lumateperone for these uses could be mediated through a combination of antagonist activity at central serotonin 5-HT2A receptors, and partial agonist activity at central dopamine D2 receptors.
12.2 Pharmacodynamics
Lumateperone has high binding affinity for serotonin 5-HT2A receptors (Ki = 0.54 nM) and moderate binding affinity for dopamine D2 (Ki = 32 nM) receptors. Lumateperone has moderate binding affinity for dopamine D1 (Ki = 41 nM) and D4 and adrenergic alpha1A and alpha1B receptors (Ki ≤ 100 nM), serotonin transporters (Ki = 33 nM), and inhibits uptake of serotonin in cells expressing the human SERT (IC50 = 150 nM). Lumateperone has low binding affinity (less than 50% inhibition at 100 nM) for muscarinic and histaminergic receptors.
In healthy human volunteers the mean cortical 5-HT2A receptor occupancy and the mean striatal D2 receptor occupancy were greater than 80% and approximately 12%, respectively, after a single 7 mg dose of CAPLYTA. D2 receptor occupancy increased with a dose from 7 mg to 28 mg. In patients with schizophrenia treated with CAPLYTA 42 mg for 2 weeks, mean dorsal striatal D2 receptor occupancy was 39%.
Cardiac Electrophysiology
QTcF interval was evaluated in a randomized, placebo- and active- (moxifloxacin 400 mg) controlled, four-arm crossover study utilizing concentration-QTc effect modeling in 33 patients with schizophrenia. The placebo-corrected change from baseline QTcF (90% two-sided upper confidence interval) values of 4.9 (8.9) and 15.8 (19.8) ms for a CAPLYTA 42 mg once daily oral dosage for five days and a CAPLYTA 126 mg once daily oral dosage (three times the recommended daily dosage) for five days, respectively.
12.3 Pharmacokinetics
Following once daily oral administration of CAPLYTA, lumateperone steady state is reached in about 5 days. Lumateperone steady-state exposure is approximately dose-proportional in the range of 3.5 mg to 56 mg (0.08 to 1.3 times the approved recommended daily dosage). A large inter-subject variability in lumateperone PK parameters was observed, with coefficients of variation for Cmax (peak plasma concentration) and AUC (area under the concentration vs time curve) ranging from 68% to 97% at steady state.
Absorption
After CAPLYTA dosing, the absolute bioavailability of lumateperone is about 4.4%. Cmax of lumateperone is reached approximately 1-2 hours after CAPLYTA dosing.
Effect of Food: Ingestion of a high-fat meal with CAPLYTA lowered lumateperone mean Cmax by 33% and increased mean AUC by 9%. Median Tmax was delayed about 1 hour (from 1 hour at fasted state to 2 hours in the presence of food).
Distribution
Protein binding of lumateperone is 97.4% at 5 µM (about 70-fold higher than therapeutic concentrations) in human plasma. The volume of distribution of lumateperone following intravenous administration is about 4.1 L/kg.
Elimination
The clearance of lumateperone is approximately 27.9 L/hour and the terminal half-life is about 18 hours after intravenous administration.
Metabolism: Lumateperone is extensively metabolized with more than twenty metabolites identified in vivo. After a single 14C-labeled oral dose, lumateperone and glucuronidated metabolites represent about 2.8% and 51% of the total plasma radioactivity, respectively. In vitro studies show that multiple enzymes, including but not limited to uridine 5'-diphospho-glucuronosyltransferases (UDP-glucuronosyltransferase, UGT) 1A1, 1A4, and 2B15, aldoketoreductase (AKR)1C1, 1B10, and 1C4, and cytochrome P450 (CYP) 3A4, 2C8, and 1A2, are involved in the metabolism of lumateperone.
Excretion: In a human mass‑balance study, 58% and 29% of the radioactive dose was recovered in the urine and feces, respectively. Less than 1% of the dose was excreted as unchanged lumateperone in the urine.
Specific Populations
No clinically significant differences in the pharmacokinetics of lumateperone were observed based on age, sex, or race.
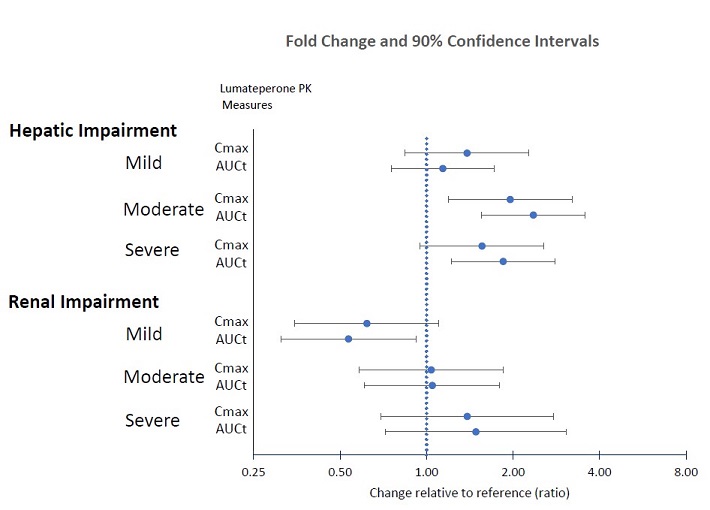
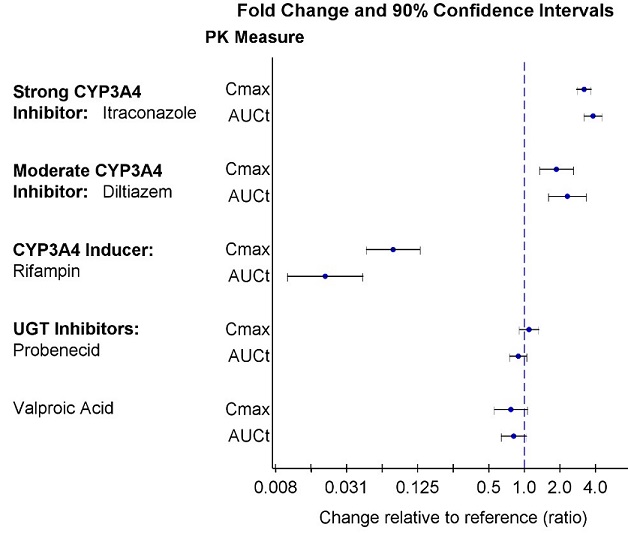
Patients with Hepatic Impairment: The pharmacokinetics of lumateperone were evaluated in patients with varying degrees of hepatic impairment (HI) and in those with normal hepatic function following administration of a single 14 mg dose of CAPLYTA. Table 7 displays the geometric mean ratios (90% confidence intervals) of lumateperone pharmacokinetic parameters in CAPLYTA-treated patients with varying degrees of HI to those with normal hepatic function [see Use in Specific Populations (8.6)].
Table 7: Geometric Mean Ratios (90% confidence interval) of Lumateperone Pharmacokinetic Parameters in CAPLYTA-treated Patients with Hepatic Impairment AUCt Cmax Mild HI (Child-Pugh Class A) 1.1 (0.8, 1.7) 1.4 (0.8, 2.3) Moderate HI (Child-Pugh Class B) 2.4 (1.6, 3.6) 2 (1.2, 3.2) Severe HI (Child-Pugh Class C) 1.8 (1.2, 2.8) 1.6 (0.9, 2.6) Patients with Renal Impairment: The pharmacokinetics of lumateperone were evaluated in patients with varying degrees of renal impairment (RI) and in those with normal renal function following administration of a single 14 mg dose of CAPLYTA. Table 8 displays the geometric mean ratios (90% confidence intervals) of lumateperone pharmacokinetic parameters in CAPLYTA-treated patients with varying degrees of RI to those with normal renal function (eGFR > 90 mL/minute/1.73 m2).
Table 8: Geometric Mean Ratios (90% confidence interval) of Lumateperone Pharmacokinetic Parameters in CAPLYTA-treated Patients with Renal Impairment AUCt Cmax Mild RI (eGFR of 60 - 89 mL/minute/1.73 m2) 0.54 (0.31, 0.92) 0.62 (0.35, 1.10) Moderate RI (eGFR of 30 - 59 mL/minute/1.73 m2) 1 (0.6, 1.8)* 1 (0.6, 1.9)* Severe RI (eGFR ≤ 29 mL/minute/1.73 m2) 1.5 (0.8, 2.6) 1.4 (0.8, 2.4) * The pharmacokinetics were unchanged.
Drug Interaction Studies
Clinical Studies:
Strong CYP3A4 Inhibitors: Itraconazole (strong CYP3A4 inhibitor) increased geometric mean (90% confidence interval) of lumateperone AUCt by 3.8-fold (3.2, 4.5) and lumateperone Cmax by 3.2-fold (2.8 – 3.7) [see Drug Interactions (7.1)]
Moderate CYP3A4 Inhibitors: Diltiazem (moderate CYP3A4 inhibitor) increased geometric mean (90% confidence interval) of lumateperone AUCt by 2.3-fold (1.6, 3.3) and lumateperone Cmax by 1.9-fold (1.3, 2.6) [see Drug Interactions (7.1)]
Strong CYP3A4 Inducers: Rifampin (strong CYP3A4 inducer) decreased geometric mean (90% confidence interval) of lumateperone AUCt by 98% (96%, 99%) and lumateperone Cmax by 92% (87%, 95%) [see Drug Interactions (7.1)]
UGT inhibitors: There were no clinically significant drug interactions with the UGT inhibitors probenecid and valproic acid.
CYP3A4 substrates: No clinically significant differences in the pharmacokinetics of midazolam (CYP3A4 substrate) or its metabolite 1-hydroxymidazolam were observed when used concomitantly with single or multiple doses of lumateperone in patients with schizophrenia.
In Vitro Studies:
Lumateperone showed little to no inhibition of CYP1A2, CYP2C9, CYP2C19, CYP2D6, or CYP3A4/5. It showed no induction of CYP1A2, CYP2B6, or CYP3A4.
Lumateperone did not appear to be a P-gp or BCRP substrate. It showed little to no inhibition of OCT2, OAT1, OAT3, OATP1B3, or OATP1B1.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Lifetime carcinogenicity studies were conducted in rats and mice, and results showed no carcinogenic potential in either species.
In Sprague-Dawley rats, males were administered lumateperone (free base) at oral doses of 3.5, 7 or 14 mg/kg/day and females were administered lumateperone at oral doses of 3.5, 10.5, or 21 mg/kg/day for the first 385 days, then doses were reduced for the two higher dose groups so that the females were administered 3.5, 7 or 14 mg/kg/day, respectively, for the duration of the study. In this study the no adverse effect level for neoplastic lesions was determined to be 14 mg/kg/day (84 mg/m2/day) for males and 10.5/7 mg/kg/day (42 mg/m2/day) for females, which are 1.6 times (females) to 3.2 times (males) the MRHD on a mg/m2 basis.
Male and female CD-1 mice were administered lumateperone at oral doses of 3.5, 10.5 or 21 mg/kg/day for the first 35 days, then doses were reduced to 1.4, 4.9, and 14 mg/kg/day, respectively, for the duration of the study. In this study, the no adverse effect level for neoplastic lesions was determined to be 10.5/4.9 mg/kg/day (15 mg/m2/day) for each sex which is 0.6 times the MRHD on a mg/m2 basis.
Mutagenesis
No evidence of mutagenic potential was found in the in vitro bacterial reverse mutation assay (Ames test) and the mouse lymphoma test without metabolic activation. Lumateperone was positive in the Ames test only in the presence of metabolic activation and only in the TA1537 strain and was positive in the mouse lymphoma test only in the presence of metabolic activation and only at high concentrations that inhibited cell growth; together these results were thought to be related to solubility limits and/or nonspecific effects on cellular function. Lumateperone was negative for clastogenic activity in the in vivo micronucleus assay in rats and was not genotoxic in the in vivo Comet assay in rats.
Impairment of Fertility
Female rats were treated with oral doses of 3.5, 10.5, 21 or 42 mg/kg/day lumateperone (free base) (0.8, 2.4, 4.9, and 9.7 times the MRHD on a mg/m2 basis) prior to mating and continuing through conception and implantation. Estrus cycle irregularities were observed at doses ≥10.5 mg/kg/day. Decreases in the median number of corpora lutea and implantation sites, and increases in the number of non-gravid uteruses, were recorded at 42 mg/kg/day. Decreased gestation body weight and body weight gain, and increases in time to mating, were observed at 21 and 42 mg/kg/day.
Male rats were treated with oral doses of 3.5, 10.5, 21 or 42 mg/kg/day lumateperone (0.8, 2.4, 4.9, and 9.7 times the MRHD on a mg/m2 basis) for 9 weeks prior to mating and throughout 14 days of mating. Decreased sperm motility, changes in sperm morphology, reduced epididymal counts, and adverse histopathology changes in testes and epididymides were observed at 21 and 42 mg/kg/day.
13.2 Animal Toxicology and/or Pharmacology
Oral administration of lumateperone caused systemic intracytoplasmic accumulation of pigmented material in dogs, rats, and mice at clinically relevant exposures (AUC). Intracytoplasmic pigmentation appeared to be localized in lysosomes. Accumulation of pigmented material persisted without reversal at the end of 1- to 2-month drug-free periods. Pigmented material was observed in the brain and spinal cord of all three species, and in the heart and eye of rats. Although the composition of the pigmented material was not established, the material is likely polymers or protein adducts formed from aniline metabolites of lumateperone.
In the dog, accumulation of pigmented material in the brain and spinal cord was associated with neuronal degeneration and necrosis, followed by axonal degeneration and histiocytic inflammation after oral administration of lumateperone for up to 9 months. In the rat, accumulation of pigmented material was associated with degenerative changes and signs of an inflammatory response in the spinal cord, peripheral nervous system, eye, and heart after oral administration of lumateperone for up to 2 years. Although overt degenerative changes were not observed in the rat brain, the presence of pigment-containing infiltrating macrophages is consistent with an inflammatory response.
The role of intracytoplasmic pigmented material in causing these lesions was not definitively established; however, the colocalization of pigmented material in tissues with degenerative changes and signs of inflammation is supportive. Alternatively, the aniline metabolites of lumateperone may undergo metabolic activation forming reactive metabolites that contribute to the observed toxicities. The role of intracellular accumulation of lumateperone or its non-aniline metabolites in these toxicities could not be ruled out.
The aniline metabolites thought to be responsible for these toxicities were detected in dogs and rats but were not present in humans at quantifiable levels. Based on all the available evidence, these toxicities do not appear to be relevant to humans.
-
14 CLINICAL STUDIES
14.1 Schizophrenia
CAPLYTA was evaluated for the treatment of schizophrenia in adults in two placebo-controlled trials (i.e., Studies 1 and 2).
Study 1 (Adults with Schizophrenia)
Study 1 Design: Study 1 (NCT01499563) was a four-week, randomized, double-blind, placebo-controlled, multi-center study in adult patients with a diagnosis of schizophrenia according to the DSM-IV-TR criteria. The primary efficacy measure was change from baseline in the Positive and Negative Syndrome Scale (PANSS) total score at Week 4. The PANSS is a 30-item scale used to measure symptoms of schizophrenia. Each item is rated by a clinician on a seven-point scale. A score of 1 indicates the absence of symptoms, and a score of 7 indicates extremely severe symptoms. The PANSS total score ranges from 30 to 210 with higher scores reflecting greater overall symptom severity.
A total of 335 patients were randomized to receive oral CAPLYTA 42 mg, CAPLYTA 84 mg (two times the recommended daily dose), an active comparator, or placebo once daily. The study was not designed to allow for efficacy comparison of CAPLYTA and the active comparator.
Study 1 Baseline Demographics: Demographic and baseline disease characteristics were similar for the CAPLYTA, active comparator, and placebo groups. Median age was 42 years (range 20 to 55 years). 17% were female, 19% were White, and 78% were Black.
Study 1: Summary of Primary Efficacy Results: Compared to the placebo group, patients randomized to CAPLYTA 42 mg once daily showed a statistically significant reduction from baseline to Day 28 in the PANSS total score. The treatment effect in the CAPLYTA 84 mg once daily treatment group (vs. placebo group) was not statistically significant. The results of Study 1 are shown in Table 9.
Study 2 (Adults with Schizophrenia)
Study 2 Design: Study 2 (NCT02282761) was a four-week, randomized, double-blind, placebo-controlled, multi-center study in adult patients with a diagnosis of schizophrenia according to the DSM-5 criteria. The primary efficacy measure was change from baseline in the PANSS total score at Week 4.
A total of 450 patients were randomized to receive oral CAPLYTA 28 mg (two-thirds the recommended daily dose), CAPLYTA 42 mg, or placebo once daily.
Study 2 Baseline Demographics: Demographic and baseline disease characteristics were similar for the CAPLYTA and placebo groups. Median age was 44 years (range 19 to 60 years); 23% were female, 26% were White and 66% were Black.
Study 2: Summary of Primary Efficacy Results: Compared to the placebo group, patients in the CAPLYTA 42 mg group showed a statistically significant reduction from baseline to Day 28 in the PANSS total score. The treatment effect in the CAPLYTA 28 mg group (vs. placebo group) was not statistically significant. The results of Study 2 are shown in Table 9.
Efficacy Results in Studies 1 and 2 (Adults with Schizophrenia)
Studies 1 and 2 did not include any patients aged 65 or older. Examination of subgroups by sex and race did not suggest differences in response in either study.
Table 9: Primary Efficacy Results for Change from Baseline in PANSS Total Score in Adult Patients with Schizophrenia (Studies 1 and 2) Primary Efficacy Endpoint: PANSS Total Score Study Number
Treatment Group N Mean Baseline Score (SD) LS Mean Change
from Baseline (SE)
Placebo-subtracted
Difference
(95% CI)1 CAPLYTA (42 mg once daily)* 84 88.1 (11.0) -13.2 (1.7) -5.8 (-10.5, -1.1)a Placebo 85 86.3 (13.1) -7.4 (1.7) -- 2 CAPLYTA (42 mg once daily)* 150 90.0 (9.6) -14.5 (1.3) -4.2 (-7.8, -0.6) Placebo 150 89.0 (10.3) -10.3 (1.3) -- The PANSS total score may range from 30 to 210; higher scores reflect greater symptom severity.
SD: standard deviation; SE: standard error; LS Mean: least squares mean; CI: unadjusted confidence interval.
aDifference (CAPLYTA minus placebo) in LS mean change from baseline not adjusted for sample size increase after unblinded interim analysis.
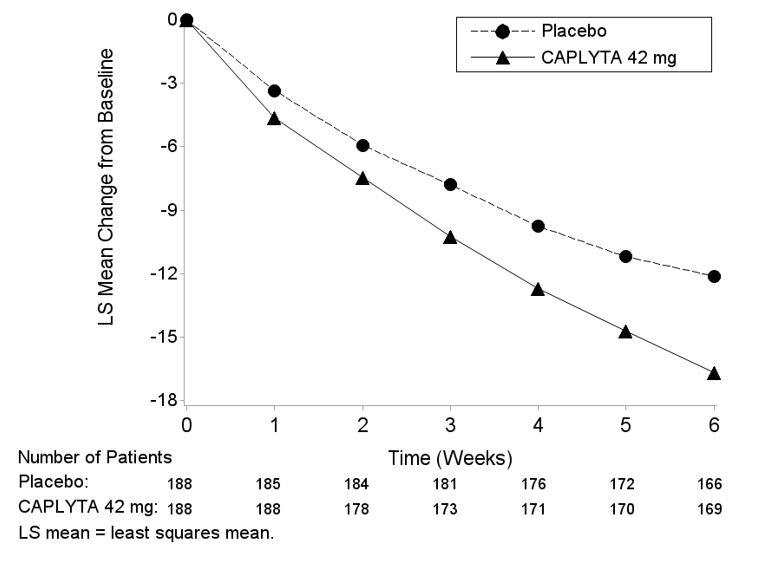
*CAPLYTA statistically significantly superior to placebo.Figure 1: Change from Baseline in PANSS Total Score by Time (Weeks) in Adult Patients with Schizophrenia in Study 2.
14.2 Depressive Episodes Associated with Bipolar I or II Disorder (Bipolar Depression)
Clinical Study of CAPLYTA Monotherapy for Bipolar I or II Disorder
Study 3 Design: The efficacy of CAPLYTA, as monotherapy, for the treatment of depressive episodes associated with bipolar I or II disorder (bipolar depression) in adults was established in a 6-week, randomized, double-blind, placebo-controlled, multi-center study in adult patients who met DSM-5 criteria for depressive episodes associated with bipolar I or bipolar II disorder (Study 3; NCT03249376). The primary efficacy measure was the change from baseline in Montgomery-Asberg Depression Rating Scale (MADRS) total score at Week 6. The MADRS is a 10-item clinician-rated scale with total scores ranging from 0 (no depressive features) to 60 (maximum score). The secondary endpoint was the change from baseline in Clinical Global Impression-Bipolar-Severity of Illness scale (CGI-BP-S) total score at Week 6. The CGI-BP-S total score is a clinician-rated scale that measures the patient’s current illness state on a 21-point scale that assesses depression, mania, and overall illness, where a higher score is associated with greater illness severity.
A total of 381 patients were randomized to receive oral CAPLYTA 42 mg or placebo once daily.
Study 3 Baseline Demographic Characteristics: Demographic and baseline characteristics were similar for the CAPLYTA and placebo groups. Median age was 45 (range 18 to 72). 58% were female, 91% were White, and 8% were Black.
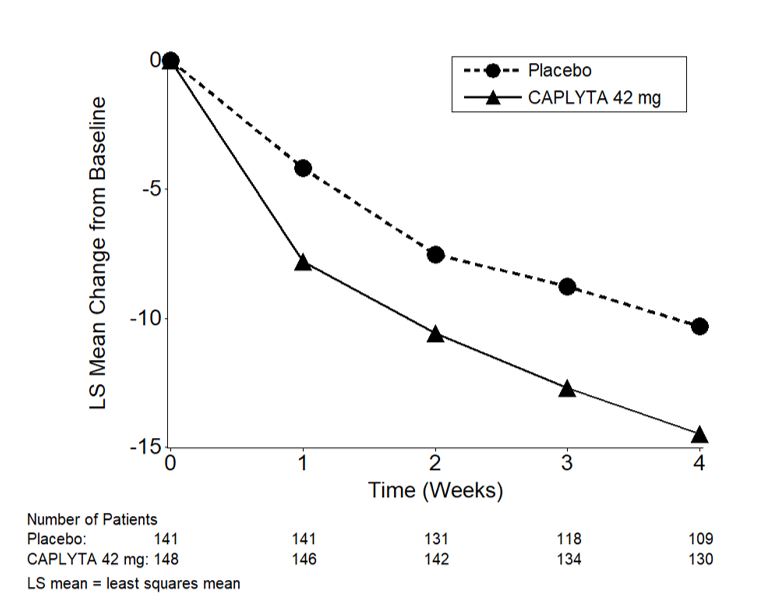
Study 3 Efficacy Results: Compared to the placebo group, patients in the CAPLYTA 42 mg group showed a statistically significant improvement from baseline to Day 43 in the MADRS total score and CGI-BP-S total score. The results of Study 3 are shown in Table 10.
Examination of subgroups by age, sex, and race did not suggest differences in response in the study.
Clinical Study of CAPLYTA Adjunctive Therapy with Lithium or Valproate for Bipolar I or II Disorder
Study 4 Design: The efficacy of CAPLYTA, as adjunctive therapy with lithium or valproate, for the treatment of depressive episodes associated with bipolar I or II disorder in adults was established in a 6-week, randomized, double-blind, placebo-controlled, multi-center study in adult patients who met DSM-5 criteria for depressive episodes associated with bipolar I or bipolar II disorder (Study 4; NCT02600507). The primary efficacy measure was the change from baseline in MADRS total score at Week 6. The secondary endpoint was the change from baseline in CGI-BP-S depression score at Week 6. The CGI-BP-S depression score is a clinician-rated scale that measures the patient’s current illness state on a 7-point scale, where a higher score is associated with greater illness severity.
A total of 529 patients were randomized to receive oral CAPLYTA 28 mg (two-thirds the recommended daily dosage), CAPLYTA 42 mg, or placebo once daily. Patients in all treatment groups continued treatment with lithium or valproate.
Study 4 Baseline Demographic Characteristics: Demographic and baseline characteristics were similar for the CAPLYTA and placebo groups. Median age was 46 (range 18 to 74). 58% were female, 88% were White, and 11% were Black.
Study 4 Efficacy Results: Compared to the placebo + lithium or valproate group, patients in the CAPLYTA 42 mg + lithium or valproate group showed a statistically significant improvement from baseline to Day 43 in the MADRS total score and CGI-BP-S depression score. The treatment effect in the CAPLYTA 28 mg + lithium or valproate group (vs. placebo + lithium or valproate group) was not statistically significant. The results of Study 4 are shown in Table 10.
Examination of subgroups by age, sex, and race did not suggest differences in response in the study.
Table 10: Primary Efficacy Results from Bipolar Depression Trials (Studies 3 and 4) Primary Efficacy Endpoint: MADRS Total Score Study
NumberTreatment Group N Mean
Baseline
Score (SD)LS Mean
Change from
Baseline (SE)Placebo-subtracted
Differencea
(95% CI)Monotherapy 3 CAPLYTA (42 mg) once daily* 188 30.8 (4.9) -16.7 (0.7) -4.6 (-6.3, -2.8) Placebo 188 30.3 (4.6) -12.1 (0.7) Adjunctive Therapy 4 CAPLYTA (42 mg) once daily* + lithium or valproate 174 32.2 (5.0) -16.9 (0.8) -2.4 (-4.4, -0.4) Placebo + lithium or valproate 174 32.1 (5.2) -14.5 (0.8)
The MADRS total score ranges from 0 to 60; higher scores reflect greater symptom severity
SD: standard deviation; SE: standard error; LS Mean: least squares mean; CI: confidence interval
a Difference (CAPLYTA minus placebo) in LS mean change from baseline
* CAPLYTA statistically significantly superior to placebo.Figure 2. Change from Baseline in MADRS Total Score by Visits (Study 3) in Adult Patients with Depressive Episodes Associated with Bipolar I or II Disorder (Monotherapy)
14.3 Adjunctive Treatment of Major Depressive Disorder in Adults
Studies 5 and 6 Designs: The effectiveness of CAPLYTA, as adjunctive therapy with antidepressants, for the treatment of major depressive disorder (MDD) in adults was established in two 6-week, randomized, double-blind, placebo-controlled, multi-center trials in adult patients who met DSM-5 criteria for MDD, with or without symptoms of anxiety who had inadequate response to one to two courses of prior antidepressant therapy (ADT) (Study 5 NCT04985942 and Study 6 NCT05061706). Inadequate response to ADT was defined as having less than 50% improvement after at least six weeks of treatment with selective serotonin or serotonin norepinephrine reuptake inhibitors or bupropion at the minimum effective ADT dosage or greater. Patients were randomized to receive oral CAPLYTA 42 mg or placebo once daily and all patients continued their background ADT.
Studies 5 and 6 Endpoints: In Study 5 and 6, the primary endpoint was change from baseline to Week 6 in the Montgomery-Asberg Depression Rating Scale (MADRS) total score [see Clinical Studies (14.2)] in the CAPLYTA + ADT group compared to the placebo + ADT group. The key secondary endpoint was the change from baseline to Week 6 in the Clinical Global Impression Scale-Severity (CGI-S) score. The CGI-S is a validated clinician-rated scale that measures the patient’s current illness state on a 1 (normal, not at all ill) to 7-point (extremely ill) scale.
Studies 5 and 6 Baseline Demographics:
- In Study 5, a total of 485 patients were randomized to receive CAPLYTA 42 mg + ADT or placebo + ADT. Demographic and baseline characteristics were similar for the CAPLYTA and placebo groups. Median age was 46 (range 18 to 65). 66% were female, 77% were White, 15% were Asian, and 7% were Black.
- In Study 6, a total of 480 patients were randomized to receive CAPLYTA 42 mg + ADT or placebo + ADT. Demographic and baseline characteristics were similar for the CAPLYTA and placebo groups. Median age was 48 (range 18 to 65). 70% were female, 95% were White.
Studies 5 and 6 Efficacy Results: In Studies 5 and 6, patients randomized to CAPLYTA 42 mg + ADT showed a statistically significant improvement from baseline to day 43 in the MADRS total score and CGI-S score compared to the placebo + ADT group. The results of Study 5 and Study 6 are shown in Table 11.
Examination of subgroups by age, sex, race, and ADT class did not suggest differences in response in these studies.
Table 11: Primary Efficacy Results from Adjunctive MDD Trials (Studies 5 and 6) Primary Efficacy Endpoint: MADRS Total Score Study
NumberTreatment Group N Mean Baseline
Score (SD)LS Mean
Change from
Baseline (SE)Placebo-subtracted
Differencea
(95% CI)5 CAPLYTA (42 mg) + ADT* 239 30.4 (3.75) -14.7 (0.54) -4.9 (-6.38, -3.44) Placebo + ADT 242 30.1 (3.50) -9.8 (0.53) 6 CAPLYTA (42 mg) + ADT* 232 30.8 (3.88) -14.7 (0.56) -4.5 (-6.03, -3.02) Placebo + ADT 237 31.5 (3.97) -10.2 (0.54) The MADRS total score ranges from 0 to 60; higher scores reflect greater symptom severity
SD: standard deviation; SE: standard error; LS Mean: least squares mean; CI: confidence interval
a Difference (CAPLYTA + ADT minus placebo + ADT) in LS mean change from baseline
* CAPLYTA + ADT statistically significantly superior to placebo + ADT.The change from baseline in MADRS total score over time in adult patients who received adjunctive treatment for MDD in Study 5 is displayed in Figure 3.
Figure 3. Change from Baseline in MADRS Total Score by Time (Weeks) in Patients with MDD (Adjunctive Treatment) in Study 5
-
16 HOW SUPPLIED/ STORAGE AND HANDLING
CAPLYTA (lumateperone) capsules are supplied as follows:
Capsule Strength Capsule Color Imprint Codes Package Configuration NDC Code 42 mg Blue cap and opaque white body ITI-007 42 mg Bottle of 30 72060-142-40 21 mg Opaque white cap and body ITI-007 21 mg Bottle of 30 72060-121-40 10.5 mg Opaque light pink cap and body ITI-007 10.5 mg Bottle of 30 72060-110-40 Store at controlled room temperature 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
-
17 PATIENT COUNSELING INFORMATION
Advise the patient or caregiver to read the FDA-approved patient labeling (Medication Guide).
Suicidal Thoughts and Behavior
Advise patients and caregivers to look for the emergence of suicidal thoughts and behaviors, especially early during CAPLYTA treatment and instruct them to report such symptoms to their healthcare provider [see Boxed Warning, Warnings and Precautions (5.2)].
Neuroleptic Malignant Syndrome
Counsel patients about a potentially fatal neuroleptic malignant syndrome (NMS), that has been reported with administration of antipsychotic drugs. Advise patients, family members, or caregivers to contact the healthcare provider or to report to the emergency room if they experience signs and symptoms of NMS [see Warnings and Precautions (5.4)].
Tardive Dyskinesia
Counsel patients on the signs and symptoms of tardive dyskinesia and to contact their healthcare provider if these abnormal movements occur [see Warnings and Precautions (5.5)].
Metabolic Changes
Educate patients about the risk of metabolic changes, how to recognize symptoms of hyperglycemia and diabetes mellitus, and the need for specific monitoring, including blood glucose, lipids, and weight [see Warnings and Precautions (5.6)].
Leukopenia/Neutropenia
Advise patients with a pre-existing low WBC or a history of drug induced leukopenia/ neutropenia that they should have their CBC monitored while taking CAPLYTA [see Warnings and Precautions (5.7)].
Orthostatic Hypotension and Syncope
Educate patients about the risk of orthostatic hypotension and syncope, especially early in treatment, and also at times of re-initiating treatment [see Warnings and Precautions (5.8)].
Effects on Driving and Operating Heavy Machinery
Caution patients about performing activities requiring mental alertness, such as operating hazardous machinery or operating a motor vehicle, until they are reasonably certain that CAPLYTA therapy does not affect them adversely [see Warnings and Precautions (5.11)].
Heat Exposure and Dehydration
Educate patients regarding appropriate care in avoiding overheating and dehydration [see Warnings and Precautions (5.12)].
Concomitant Drugs
Advise patients to inform their health care providers of any changes to their current prescription or over-the-counter drugs because there is a potential for clinically significant interactions [see Drug Interactions (7.1)].
Pregnancy
Advise patients to notify their healthcare provider if they become pregnant or intend to become pregnant during treatment with CAPLYTA. Advise patients that CAPLYTA used during the third trimester may cause extrapyramidal and/or withdrawal symptoms (agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress, and feeding disorder) in the neonate. Advise patients that there is a pregnancy registry that monitors pregnancy outcomes in women exposed to CAPLYTA during pregnancy [see Use in Specific Populations (8.1)].
Infertility
Advise males and females of reproductive potential that CAPLYTA may impair fertility [see Use in Specific Populations (8.3)].
Distributed by Intra-Cellular Therapies, Inc.
Bedminster, NJ 07921
CAPLYTA is a registered trademark of Intra-Cellular Therapies, Inc.
© 2025 Intra-Cellular Therapies, Inc. All rights reserved.
-
MEDICATION GUIDE
MEDICATION GUIDE
CAPLYTA (kap-LITE-ah)
(lumateperone) capsules, for oral useWhat is the most important information I should know about CAPLYTA?
CAPLYTA may cause serious side effects, including:
- Increased risk of death in elderly people with dementia related psychosis. Medicines like CAPLYTA can raise the risk of death in elderly people who have lost touch with reality (psychosis) due to confusion and memory loss (dementia). CAPLYTA is not approved for the treatment of people with dementia-related psychosis.
-
Increased risk of suicidal thoughts and actions. CAPLYTA and antidepressant medicines increase the risk of suicidal thoughts and actions in people 24 years of age and younger, especially within the first few months of treatment or when the dose is changed.
- Depression and other mental illnesses are the most important causes of suicidal thoughts and actions.
How can I watch for and try to prevent suicidal thoughts and actions in myself or a family member?
- Pay close attention to any changes, especially sudden changes in mood, behaviors, thoughts, or feelings. This is very important when CAPLYTA or the antidepressant medicine is started or when the dose is changed.
- Call your healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings, or if you develop suicidal thoughts or actions.
- Keep all follow-up visits with your healthcare provider as scheduled. Call your healthcare provider between visits as needed, especially if you have concerns about symptoms.
Call a healthcare provider right away if you or your family member have any of the following symptoms, especially if they are new, worse, or worry you:
• thoughts about suicide or dying
• suicide attempts
• new or worse depression
• new or worse anxiety
• feeling very agitated or restless
• panic attacks
• trouble sleeping
• new or worse irritability
• acting aggressive, being angry, or violent
• acting on dangerous impulses
• an extreme increase in activity and talking (mania)
• other unusual changes in behavior or mood
See “What are the possible side effects of CAPLYTA?” for more information about side effects. What is CAPLYTA? CAPLYTA is a prescription medicine used in adults:
- to treat schizophrenia
- alone or along with the medicine lithium or valproate to treat depressive episodes that happen with bipolar I or bipolar II disorder (bipolar depression)
- along with antidepressant medicines to treat major depressive disorder (MDD)
It is not known if CAPLYTA is safe and effective in children.
Who should not take CAPLYTA?
Do not take CAPLYTA if you:
- are allergic to lumateperone or any of the ingredients in CAPLYTA. See the end of this Medication Guide for a complete list of ingredients in CAPLYTA.
Before taking CAPLYTA, tell your healthcare provider about all of your medical conditions, including if you: - have or have had heart problems or a stroke
- have or have had low or high blood pressure
- have or have had diabetes or high blood sugar, or a family history of diabetes or high blood sugar.
- have or have had high levels of total cholesterol, LDL cholesterol, or triglycerides or low levels of HDL cholesterol
- have or have had a low white blood cell count
- have or have had seizures (convulsions)
- have or have had kidney or liver problems
- are pregnant or plan to become pregnant. CAPLYTA may harm your unborn baby. Taking CAPLYTA during your third trimester of pregnancy may cause your baby to have abnormal muscle movements or withdrawal symptoms after birth. Talk to your healthcare provider about the risk to your unborn baby if you take CAPLYTA during pregnancy.
- Tell your healthcare provider if you become pregnant or think you are pregnant during treatment with CAPLYTA.
- There is a pregnancy registry for females who are exposed to CAPLYTA during pregnancy. If you become pregnant during treatment with CAPLYTA, talk to your healthcare provider about registering with the National Pregnancy Registry for Atypical Antipsychotics. You can register by calling 1-866-961-2388 or go to https://womensmentalhealth.org/research/pregnancyregistry/.
- are breastfeeding or plan to breastfeed. CAPLYTA passes into your breast milk. Talk to your healthcare provider about the risks and benefits of breastfeeding and the best way to feed your baby during treatment with CAPLYTA.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
CAPLYTA and other medicines may affect each other causing possible serious side effects. CAPLYTA may affect the way other medicines work, and other medicines may affect how CAPLYTA works.
Your healthcare provider can tell you if it is safe to take CAPLYTA with your other medicines. Do not start or stop any medicines during treatment with CAPLYTA without first talking to your healthcare provider.
Know the medicines you take. Keep a list of your medicines to show your healthcare provider and pharmacist when you get a new medicine.
How should I take CAPLYTA? - Take CAPLYTA exactly as your healthcare provider tells you to take it. Do not change the dose or stop taking CAPLYTA without first talking to your healthcare provider.
- Take CAPLYTA 1 time each day with or without food.
- If you take too much CAPLYTA, call your healthcare provider or Poison Help Line at 1-800-222-1222 or go to the nearest hospital emergency room right away.
What should I avoid while taking CAPLYTA? - Do not drive, operate machinery, or do other dangerous activities until you know how CAPLYTA affects you. CAPLYTA may make you drowsy.
- Do not become too hot or dehydrated during treatment with CAPLYTA.
- Do not exercise too much.
- In hot weather, stay inside in a cool place if possible.
- Stay out of the sun.
- Do not wear too much clothing or heavy clothing.
- Drink plenty of water.
What are the possible side effects of CAPLYTA?
CAPLYTA may cause serious side effects, including:
- See “What is the most important information I should know about CAPLYTA?”
- Stroke (cerebrovascular problems) in elderly people with dementia-related psychosis that can lead to death.
- Neuroleptic malignant syndrome (NMS) is a serious condition that can lead to death. Call your healthcare provider or go to the nearest hospital emergency room right away if you have some or all of the following signs and symptoms of NMS:
◦ high fever
◦ confusion
◦ changes in your breathing, heart rate, and blood pressure
◦ stiff muscles
◦ increased sweating
- Uncontrolled body movements (tardive dyskinesia). CAPLYTA may cause movements that you cannot control in your face, tongue, or other body parts. Tardive dyskinesia may not go away, even if you stop taking CAPLYTA. Tardive dyskinesia may also start after you stop taking CAPLYTA.
-
Problems with your metabolism such as:
- high blood sugar (hyperglycemia) and diabetes. Increases in blood sugar can happen in some people who take CAPLYTA. Extremely high blood sugar can lead to coma or death. Your healthcare provider should check your blood sugar before you start, or soon after you start CAPLYTA, and then regularly during long term treatment with CAPLYTA.
Call your healthcare provider if you have any of these symptoms of high blood sugar during treatment with CAPLYTA:
• feel very thirsty
• need to urinate more than usual
• feel very hungry
• feel weak or tired
• feel sick to your stomach
• feel confused, or your breath smells fruity
- increased fat levels (cholesterol and triglycerides) in your blood. Your healthcare provider should check the fat levels in your blood before you start, or soon after you start CAPLYTA, and then periodically during treatment with CAPLYTA.
- weight gain. You and your healthcare provider should check your weight before you start and often during treatment with CAPLYTA.
- Low white blood cell count. Your healthcare provider may do blood tests during the first few months of treatment with CAPLYTA.
- Decreased blood pressure (orthostatic hypotension). You may feel lightheaded or faint when you rise too quickly from a sitting or lying position.
- Falls. CAPLYTA may make you sleepy or dizzy, may cause a decrease in your blood pressure when changing position (orthostatic hypotension), and can slow your thinking and motor skills which may lead to falls that can cause fractures or other injuries.
- Seizures (convulsions).
- Sleepiness, drowsiness, feeling tired, difficulty thinking and doing normal activities. See "What should I avoid while taking CAPLYTA?"
- Problems controlling your body temperature so that you feel too warm. See “What should I avoid while taking CAPLYTA?”
- Difficulty swallowing that can cause food or liquid to get into your lungs.
The most common side effects of CAPLYTA include sleepiness, dizziness, nausea, dry mouth, feeling tired, and diarrhea.
CAPLYTA may cause fertility problems in females and males. Talk to your healthcare provider if this is a concern for you.
These are not all the possible side effects of CAPLYTA.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store CAPLYTA? • Store CAPLYTA at room temperature between 68°F to 77°F (20°C to 25°C).
Keep CAPLYTA and all medicines out of the reach of children.General information about the safe and effective use of CAPLYTA.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CAPLYTA for a condition for which it was not prescribed. Do not give CAPLYTA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about CAPLYTA that is written for health professionals.
What are the ingredients in CAPLYTA? Active ingredient: lumateperone
Inactive ingredients: croscarmellose sodium, gelatin, magnesium stearate, mannitol, and talc. Colorants include FD&C blue #1 and red #3 (42 mg), FDA/E172 black iron oxide, FDA/E172 red iron oxide and FD&C red #3 (10.5 mg), and titanium dioxide (42 mg, 21 mg and 10.5 mg).
Distributed by: Intra-Cellular Therapies, Inc., Bedminster, NJ 07921
©2025 Intra-Cellular Therapies, Inc. All rights reserved.
For more information, go to www.CAPLYTA.com or call (888) 252-4824
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised:11/2025
-
PRINCIPAL DISPLAY PANEL
42 mg 30 Count Bottle Label
Rx Only
30 capsules
NDC 72060-142-40
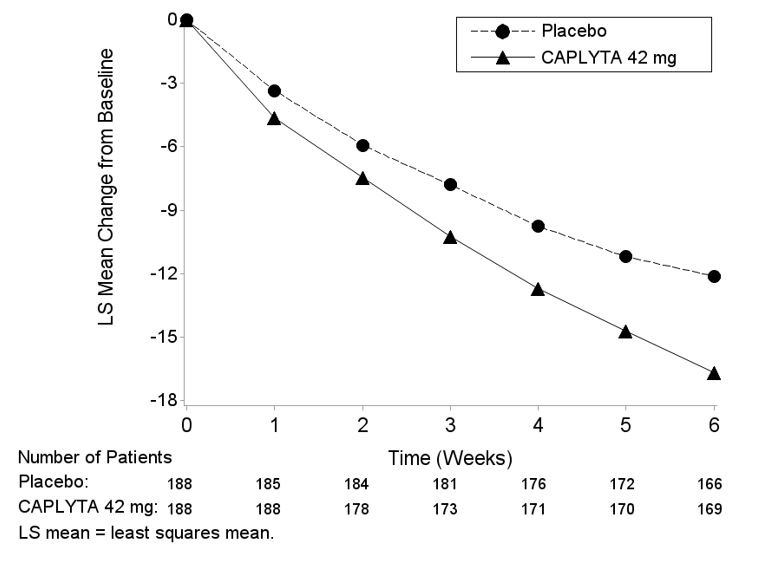
21 mg 30 Count Bottle Label
Rx Only
30 capsules
NDC 72060-121-40
10.5 mg 30 Count Bottle Label
Rx Only
30 capsules
NDC 72060-110-40

42 mg 7 Count Sample Carton
Professional Sample.
Not for Sale.
NDC 72060-142-07

21 mg 7 Count Sample Carton
Professional Sample.
Not for Sale.
NDC 72060-121-07

10.5 mg 7 Count Sample Carton
Professional Sample.
Not for Sale.
NDC 72060-110-07

-
INGREDIENTS AND APPEARANCE
CAPLYTA
lumateperone capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:72060-142 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LUMATEPERONE (UNII: 70BSQ12069) (LUMATEPERONE - UNII:70BSQ12069) LUMATEPERONE 42 mg Inactive Ingredients Ingredient Name Strength MANNITOL (UNII: 3OWL53L36A) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) TALC (UNII: 7SEV7J4R1U) MAGNESIUM STEARATE (UNII: 70097M6I30) GELATIN (UNII: 2G86QN327L) Product Characteristics Color blue (Blue cap) , white (opaque white body) Score no score Shape CAPSULE Size 22mm Flavor Imprint Code ITI;007;42;mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72060-142-40 30 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2022 2 NDC:72060-142-07 7 in 1 BOTTLE; Type 0: Not a Combination Product 11/01/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209500 02/01/2020 CAPLYTA
lumateperone capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:72060-121 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LUMATEPERONE (UNII: 70BSQ12069) (LUMATEPERONE - UNII:70BSQ12069) LUMATEPERONE 21 mg Product Characteristics Color white (Opaque white cap and body) Score no score Shape CAPSULE Size 19mm Flavor Imprint Code ITI;007;21;mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72060-121-40 30 in 1 BOTTLE; Type 0: Not a Combination Product 08/09/2022 2 NDC:72060-121-07 7 in 1 BOTTLE; Type 0: Not a Combination Product 06/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209500 08/09/2022 CAPLYTA
lumateperone capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:72060-110 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LUMATEPERONE (UNII: 70BSQ12069) (LUMATEPERONE - UNII:70BSQ12069) LUMATEPERONE 10.5 mg Product Characteristics Color pink (Opaque light pink cap and body) Score no score Shape CAPSULE Size 18mm Flavor Imprint Code ITI;007;10;5;mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72060-110-40 30 in 1 BOTTLE; Type 0: Not a Combination Product 08/09/2022 2 NDC:72060-110-07 7 in 1 BOTTLE; Type 0: Not a Combination Product 06/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209500 08/09/2022 Labeler - Intra-Cellular Therapies, Inc (112765909)