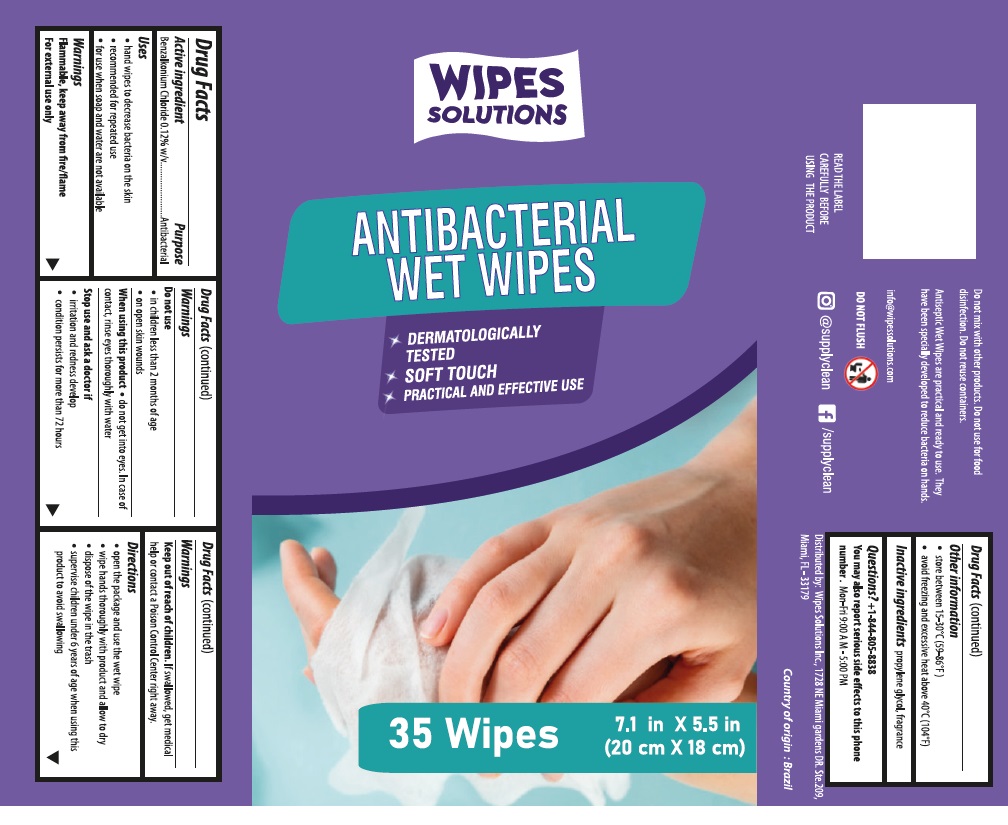
Label: WIPES SOLUTIONS ANTIBACTERIAL WET WIPES- benzalkonium chloride cloth
-
Contains inactivated NDC Code(s)
NDC Code(s): 80310-036-18, 80310-036-36 - Packager: Supplyclean Industria de Produtos de Higiene S/A
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 27, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Purpose
- Uses
-
Warnings
Flammable, keep away from fire/flame
For external use only
Do not use
• in children less than 2 months of age
• on open skin wounds
When using this product • do not get into eyes. In case of contact, rinse eyes thoroughly with water
• do not ingest
Stop use and ask a doctor if
• irritation and redness develop
• condition persists for more than 72 hours
-
Directions
• remove the plastic cover and pull the tip of the first wipe in the center of the roll
• insert the tip of the scarf into the center opening of the cover from the bottom up
• replace the cap, pull the first wipe and the rest will come off automatically
• wipe hands thoroughly with product and allow to dry without wiping
• dispose of the wipe in the trash
• supervise children under 6 years of age when using this product to avoid swallowing
- Other information
- INACTIVE INGREDIENT
- QUESTIONS
-
SPL UNCLASSIFIED SECTION
DERMATOLOGICALLY TESTED
SOFT TOUCH
PRACTICAL AND EFFECTIVE USE
READ THE LABEL CAREFULLY BEFORE USING THE PRODUCT
Do not mix with other products. Do not use for food disinfection. Do not reuse containers.
Antiseptic Wet Wipes are practical and ready to use. They have been specially developed to reduce bacteria on hands
Distributed by: Wipes Solutions Inc.
1728 NE Miami Gardens DR. Ste.209,
Miami, FL 33179
Made in Brazil
info@wipessolutions.com
Directions for use: Open the package and use the wet wipe After use, dispose of the wipe in the trash
DO NOT FLUSH
Shelf life 24 months
/supplyclean
@supplyclean
- Packaging
-
INGREDIENTS AND APPEARANCE
WIPES SOLUTIONS ANTIBACTERIAL WET WIPES
benzalkonium chloride clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80310-036 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.12 g in 100 mL Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80310-036-36 35 in 1 TUBE 08/28/2020 1 0.00685 mL in 1 PACKAGE; Type 0: Not a Combination Product 2 NDC:80310-036-18 150 in 1 PAIL 08/28/2020 2 0.0044 mL in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 08/27/2020 Labeler - Supplyclean Industria de Produtos de Higiene S/A (944745857) Establishment Name Address ID/FEI Business Operations Supplyclean Industria de Produtos de Higiene S/A 944745857 manufacture(80310-036)