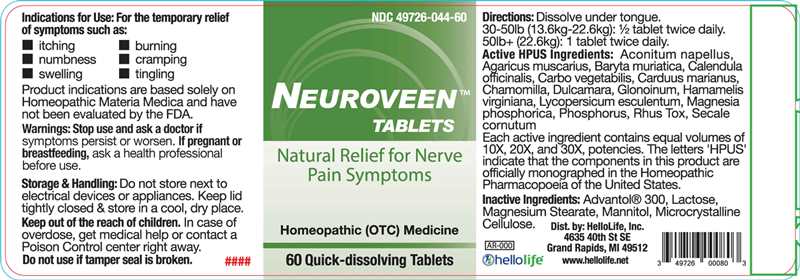
Label: NEUROVEEN- aconitum napellus, agaricus muscarius, baryta muriatica, calendula officinalis, carbo vegetabilis, carduus marianus, chamomilla, dulcamara, glonoinum, hamamelis virginiana, lycopersicum esculentum, magnesia phosphorica, phosphorus, rhus tox, secale cornutum tablet
- NDC Code(s): 49726-044-60
- Packager: HelloLife, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated October 20, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE *HPUS INGREDIENTS:
Aconitum Napellus 10X, 20X, 30X, Agaricus Muscarius 10X, 20X, 30X, Baryta Muriatica 10X, 20X, 30X, Calendula Officinalis 10X, 20X, 30X, Carbo Vegetabilis 10X, 20X, 30X, Carduus Marianus 10X, 20X, 30X, Chamomilla 10X, 20X, 30X, Dulcamara 10X, 20X, 30X, Glonoinum 10X, 20X, 30X, Hamamelis Virginiana 10X, 20X, 30X, Lycopersicum Esculentum 10X, 20X, 30X, Magnesia Phosphorica 10X, 20X, 30X, Phosphorus 10X, 20X, 30X, Rhus Tox 10X, 20X, 30X, Secale Cornutum 10X, 20X, 30X.
Each active ingredient contains equal volumes of 10X, 20X, and 30X, potencies. The letters "HPUS" indicate that the components in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
- INDICATIONS FOR USE:
- WARNINGS:
- KEEP OUT OF REACH OF CHILDREN:
- STORAGE & HANDLING:
- DIRECTIONS:
- INDICATIONS FOR USE:
- INACTIVE INGREDIENTS:
- QUESTIONS:
- PACKAGE LABEL DISPLAY:
-
INGREDIENTS AND APPEARANCE
NEUROVEEN
aconitum napellus, agaricus muscarius, baryta muriatica, calendula officinalis, carbo vegetabilis, carduus marianus, chamomilla, dulcamara, glonoinum, hamamelis virginiana, lycopersicum esculentum, magnesia phosphorica, phosphorus, rhus tox, secale cornutum tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49726-044 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACONITUM NAPELLUS (UNII: U0NQ8555JD) (ACONITUM NAPELLUS - UNII:U0NQ8555JD) ACONITUM NAPELLUS 10 [hp_X] AMANITA MUSCARIA FRUITING BODY (UNII: DIF093I037) (AMANITA MUSCARIA FRUITING BODY - UNII:DIF093I037) AMANITA MUSCARIA FRUITING BODY 10 [hp_X] BARIUM CHLORIDE DIHYDRATE (UNII: EL5GJ3U77E) (BARIUM CATION - UNII:V645272HLN) BARIUM CHLORIDE DIHYDRATE 10 [hp_X] CALENDULA OFFICINALIS FLOWERING TOP (UNII: 18E7415PXQ) (CALENDULA OFFICINALIS FLOWERING TOP - UNII:18E7415PXQ) CALENDULA OFFICINALIS FLOWERING TOP 10 [hp_X] ACTIVATED CHARCOAL (UNII: 2P3VWU3H10) (ACTIVATED CHARCOAL - UNII:2P3VWU3H10) ACTIVATED CHARCOAL 10 [hp_X] SILYBUM MARIANUM SEED (UNII: U946SH95EE) (MILK THISTLE - UNII:U946SH95EE) SILYBUM MARIANUM SEED 10 [hp_X] MATRICARIA RECUTITA (UNII: G0R4UBI2ZZ) (MATRICARIA CHAMOMILLA - UNII:G0R4UBI2ZZ) MATRICARIA RECUTITA 10 [hp_X] SOLANUM DULCAMARA TOP (UNII: KPS1B1162N) (SOLANUM DULCAMARA TOP - UNII:KPS1B1162N) SOLANUM DULCAMARA TOP 10 [hp_X] NITROGLYCERIN (UNII: G59M7S0WS3) (NITROGLYCERIN - UNII:G59M7S0WS3) NITROGLYCERIN 10 [hp_X] HAMAMELIS VIRGINIANA ROOT BARK/STEM BARK (UNII: T7S323PKJS) (HAMAMELIS VIRGINIANA ROOT BARK/STEM BARK - UNII:T7S323PKJS) HAMAMELIS VIRGINIANA ROOT BARK/STEM BARK 10 [hp_X] TOMATO (UNII: Z4KHF2C175) (TOMATO - UNII:Z4KHF2C175) TOMATO 10 [hp_X] MAGNESIUM PHOSPHATE, DIBASIC TRIHYDRATE (UNII: HF539G9L3Q) (MAGNESIUM CATION - UNII:T6V3LHY838) MAGNESIUM PHOSPHATE, DIBASIC TRIHYDRATE 10 [hp_X] PHOSPHORUS (UNII: 27YLU75U4W) (PHOSPHORUS - UNII:27YLU75U4W) PHOSPHORUS 10 [hp_X] TOXICODENDRON PUBESCENS LEAF (UNII: 6IO182RP7A) (TOXICODENDRON PUBESCENS LEAF - UNII:6IO182RP7A) TOXICODENDRON PUBESCENS LEAF 10 [hp_X] CLAVICEPS PURPUREA SCLEROTIUM (UNII: 01G9XEA93N) (CLAVICEPS PURPUREA SCLEROTIUM - UNII:01G9XEA93N) CLAVICEPS PURPUREA SCLEROTIUM 10 [hp_X] Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSPOVIDONE (UNII: 2S7830E561) COPOVIDONE (UNII: D9C330MD8B) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) Product Characteristics Color white Score no score Shape ROUND Size 10mm Flavor Imprint Code H Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49726-044-60 60 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 10/15/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 10/15/2018 Labeler - HelloLife, Inc. (065619378) Registrant - Apotheca Company (844330915) Establishment Name Address ID/FEI Business Operations Apotheca Company 844330915 manufacture(49726-044) , api manufacture(49726-044) , label(49726-044) , pack(49726-044)