Label: CEFAZOLIN- cefazolin sodium injection, solution
- NDC Code(s): 0338-3503-41, 0338-3508-41
- Packager: Baxter Healthcare Corporation
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated February 1, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use CEFAZOLIN IN DEXTROSE INJECTION safely and effectively. See full prescribing information for CEFAZOLIN IN DEXTROSE INJECTION.
CEFAZOLIN in Dextrose injection, for intravenous use
Initial U.S. Approval: 1973RECENT MAJOR CHANGES
INDICATIONS AND USAGE
Cefazolin in Dextrose Injection is a cephalosporin antibacterial indicated for:
- •
- Treatment of the following infections caused by susceptible isolates of the designated microorganisms in adult and pediatric patients for whom appropriate dosing with this formulation can be achieved: (1)
- •
- Perioperative prophylaxis in adults and pediatric patients aged 10 to 17 years old for whom appropriate dosing with this formulation can be achieved (1.9)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Cefazolin in Dextrose Injection and other antibacterial drugs, Cefazolin in Dextrose Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.10).
DOSAGE AND ADMINISTRATION
If a dose of Cefazolin in Dextrose Injection is required that does not equal 1 gram or 2 grams, this product is not recommended for use and as an alternative formulation of cefazolin should be considered. (2.1)
For intravenous use only over approximately 30 minutes. (2)- *
- In rare instances, doses of up to 12 grams of cefazolin per day have been used
Recommended Dosing Schedule in Adult Patients with CLcr Greater Than or Equal To 55 m L/min. (2.1)
Site and Type of Infection
Dose
Frequency
Moderate to severe infections
500 mg to 1 gram
every 6 to 8 hours
Mild infections caused by susceptible gram-positive cocci
250 mg to 500 mg
every 8 hours
Acute, uncomplicated urinary tract infections
1 gram
every 12 hours
Pneumococcal pneumonia
500 mg
every 12 hours
Severe, life-threatening infections (e.g., endocarditis, septicemia)*
1 gram to 1.5 grams
every 6 hours
Perioperative prophylaxis1 gram to 2 grams
½ to 1 hour prior to start of surgery
500 mg to 1 gram
during surgery for lengthy procedures
500 mg to 1 gram
every 6 to 8 hours for 24 hours postoperatively
- Site and Type of Infection
- Dose
- Frequency
- Moderate to severe infections*
- 25 to 50 mg per kg
- divided into 3 or 4 equal doses
- Severe infections*
- May increase to 100 mg
per kg
- divided into 3 or 4 equal doses
- Perioperative prophylaxis (10 to 17 years old)
- < 50 kg: 1 gram
- ½ to 1 hour prior to start of surgery
- ≥ 50 kg: 2 grams
- 500 mg to 1 g
- during surgery for lengthy procedures
- 500 mg to 1 g
- every 6 to 8 hours for 24 hours postoperatively
DOSAGE FORMS AND STRENGTHS
- Injection: 1 gram in 50 mL and 2 grams in 100 mL (3)
CONTRAINDICATIONS
- Hypersensitivity to cefazolin or other cephalosporin class antibacterial drugs, penicillins, or other beta-lactams (4.1)
WARNINGS AND PRECAUTIONS
- •
- Hypersensitivity reactions: Cross-hypersensitivity may occur in up to 10% of patients with a history of penicillin allergy. If an allergic reaction occurs, discontinue the drug. (5.1)
- •
- Clostridioides difficile-associated diarrhea (CDAD): May range from mild diarrhea to fatal colitis. Evaluate if diarrhea occurs. (5.3)
- •
- Prothrombin Activity: May be associated with fall in prothrombin activity. Prothrombin time should be monitored in patients at risk and exogenous vitamin K administered as indicated (5.5)
ADVERSE REACTIONS
- •
- Adult and Pediatric Patients: Most common adverse reactions: gastrointestinal (nausea, vomiting, diarrhea), and allergic reactions (anaphylaxis, urticaria, skin rash). (6)
- •
- Pediatric Patients with Perioperative Prophylaxis: The most frequently reported adverse reactions (incidence ≥ 5%) were nausea, infusion site pain, and headache. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Baxter Healthcare at 1-866-888-2472 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Probenecid: The renal excretion of cefazolin is inhibited by probenecid. Co-administration of probenecid with Cefazolin in Dextrose Injection is not recommended. (7)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 2/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Respiratory Tract Infections
1.2 Urinary Tract Infections
1.3 Skin and Skin Structure Infections
1.4 Biliary Tract Infections
1.5 Bone and Joint Infections
1.6 Genital Infections
1.7 Septicemia
1.8 Endocarditis
1.9 Perioperative Prophylaxis
1.10 Usage
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
2.2 Dosage for the Treatment of Infections
2.3 Dosage for Perioperative Prophylaxis
2.4 Dosage Recommendations in Adult and Pediatric Patients with Renal Impairment
2.5 Preparation for Use of Cefazolin in Dextrose Injection
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 Hypersensitivity to Cefazolin or the Cephalosporin Class of Antibacterial Drugs, Penicillins, or Other Beta-lactams
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions to Cefazolin, Cephalosporins, Penicillins, or Other Beta-lactams
5.2 Seizures in Patients with Renal Impairment
5.3 Clostridioides difficile-associated Diarrhea
5.4 Hypersensitivity to Dextrose-containing Products
5.5 Prothrombin Activity
5.6 Risk of Development of Drug-resistant Bacteria
5.7 Drug/Laboratory Test Interactions
5.8 Patients with Overt or Known Subclinical Diabetes Mellitus or Carbohydrate Intolerance
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
6.3 Cephalosporin-class Adverse Reactions
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Patients with Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Respiratory Tract Infections
Cefazolin in Dextrose Injection is indicated for the treatment of respiratory tract infections due to Streptococcus pneumoniae, Staphylococcus aureus and Streptococcus pyogenes in adults and pediatric patients for whom appropriate dosing with this formulation can be achieved [see Dosage and Administration (2.1, 2.2, 2.4 and 2.5) and Use in Specific Populations (8.4)].
Limitations of Use
Injectable benzathine penicillin is considered the drug of choice in treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever.
Cefazolin in Dextrose Injection is effective in the eradication of streptococci from the nasopharynx; however, data establishing the efficacy of cefazolin in the subsequent prevention of rheumatic fever are not available.
1.2 Urinary Tract Infections
Cefazolin in Dextrose Injection is indicated for the treatment of urinary tract infections due to Escherichia coli, and Proteus mirabilis in adults and pediatric patients for whom appropriate dosing with this formulation can be achieved [see Dosage and Administration (2.1, 2.2, 2.4 and 2.5) and Use in Specific Populations (8.4)].
1.3 Skin and Skin Structure Infections
Cefazolin in Dextrose Injection is indicated for the treatment of skin and skin structure infections due to S. aureus, S. pyogenes, and Streptococcus agalactiae in adults and pediatric patients for whom appropriate dosing with this formulation can be achieved [see Dosage and Administration (2.1, 2.2, 2.4 and 2.5) and Use in Specific Populations (8.4)].
1.4 Biliary Tract Infections
Cefazolin in Dextrose Injection is indicated for the treatment of biliary infections due to E. coli, various isolates of streptococci, P. mirabilis, and S. aureus in adults and pediatric patients for whom appropriate dosing with this formulation can be achieved [see Dosage and Administration (2.1, 2.2, 2.4 and 2.5) and Use in Specific Populations (8.4)].
1.5 Bone and Joint Infections
Cefazolin in Dextrose Injection is indicated for the treatment of bone and joint infections due to S. aureus in adults and pediatric patients for whom appropriate dosing with this formulation can be achieved [see Dosage and Administration (2.1, 2.2, 2.4 and 2.5) and Use in Specific Populations (8.4)].
1.6 Genital Infections
Cefazolin in Dextrose Injection is indicated for the treatment of genital infections due to E. coli, and P. mirabilis in adults and pediatric patients for whom appropriate dosing with this formulation can be achieved [see Dosage and Administration (2.1, 2.2, 2.4 and 2.5) and Use in Specific Populations (8.4)].
1.7 Septicemia
Cefazolin in Dextrose Injection is indicated for the treatment of septicemia due to S. pneumoniae, S. aureus, P. mirabilis, and E. coli in adults and pediatric patients for whom appropriate dosing with this formulation can be achieved [see Dosage and Administration (2.1, 2.2, 2.4 and 2.5) and Use in Specific Populations (8.4)].
1.8 Endocarditis
Cefazolin in Dextrose Injection is indicated for the treatment of endocarditis due to S. aureus and S. pyogenes in adults and pediatric patients for whom appropriate dosing with this formulation can be achieved [see Dosage and Administration (2.1, 2.2, 2.4 and 2.5) and Use in Specific Populations (8.4)].
1.9 Perioperative Prophylaxis
Cefazolin in Dextrose Injection is indicated is indicated for perioperative prophylaxis in adults and pediatric patients aged 10 to 17 years old for whom appropriate dosing with this formulation can be achieved [see Dosage and Administration (2.1, 2.3, 2.4, 2.5) and Use in Specific Populations (8.4)].
The perioperative use of Cefazolin in Dextrose Injection is indicated in adult and pediatric patients (aged 10 to 17 years old) surgical patients in whom infection at the operative site would present a serious risk (e.g., during open-heart surgery and prosthetic arthroplasty).
The prophylactic administration of Cefazolin in Dextrose Injection preoperatively, intraoperatively, and postoperatively may reduce the incidence of certain postoperative infections in patients undergoing surgical procedures which are classified as contaminated or potentially contaminated (e.g., vaginal hysterectomy, and cholecystectomy in high-risk patients such as those older than 70 years, with acute cholecystitis, obstructive jaundice, or common duct bile stones).
1.10 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Cefazolin in Dextrose Injection and other antibacterial drugs, Cefazolin in Dextrose Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
If a dose of Cefazolin in Dextrose Injection is required that does not equal 1 gram or 2 grams, this product is not recommended for use and as an alternative formulation of cefazolin should be considered.
Administer Cefazolin in Dextrose Injection intravenously over approximately 30 minutes.
2.2 Dosage for the Treatment of Infections
Dosage for the Treatment of Infections in Adults with Creatinine Clearance (CLcr) Equal to 55 mL/min or Greater
The recommended adult dosages for the treatment of infections [see Indications and Usage (1.1 to 1.8)] are outlined in Table 1 below. Administer Cefazolin in Dextrose Injection intravenously over approximately 30 minutes.
Table 1: Recommended Dosing Schedule in Adult Patients with CLcr Greater Than or Equal To 55 mL/min*. Site and Type of Infection
Dose
Frequency
Moderate to severe infections
500 mg to 1 gram
every 6 to 8 hours
Mild infections caused by susceptible gram-positive cocci
250 mg to 500 mg
every 8 hours
Acute, uncomplicated urinary tract infections
1 gram
every 12 hours
Pneumococcal pneumonia
500 mg
every 12 hours
Severe, life-threatening infections (e.g., endocarditis, septicemia)†
1 gram to 1.5 grams
every 6 hours
Dosage for the Treatment of Infections in Pediatric Patients with CLcr Equal to 70 mL/min or Greater
The recommended pediatric dosages for the treatment of infections [see Indications and Usage (1.1 to 1.8)] are outlined in Table 2 below. Administer Cefazolin in Dextrose Injection intravenously over approximately 30 minutes.
If a dose of Cefazolin in Dextrose Injection is required that does not equal 1 gram or 2 grams, this product is not recommended for use and an alternative formulation of cefazolin should be considered [see Use in Specific Populations (8.4)].
Table 2: Recommended Dosage in Pediatric Patients with CLcr 70 mL/min or greater for Treatment of Infections [see Indications and Usage (1.1 to 1.8)] Type of Severity
Recommended Total Daily Dosage
Mild to moderate infections
25 mg/kg to 50 mg/kg, divided into 3 or 4 equal doses
Severe infections
May increase to 100 mg/kg, divided into 3 or 4 equal doses
2.3 Dosage for Perioperative Prophylaxis
Dosage for Perioperative Prophylaxis in Adults with CLcr Equal to 55 mL/min or Greater
To prevent postoperative infection in contaminated or potentially contaminated surgery, recommended dosages are described in Table 3 below.
Table 3: Recommended Dosage for Perioperative Prophylaxis in Adults with Clcr of 55 mL/min or Greater - Dose administered ½ hour to 1 hour prior to the start of surgery
- Additional dose during lengthy operative procedures (e.g., 2 hours or more)
- Dose for 24 hours postoperatively
- 1 g
2 g
- 500 mg to 1 g
- 500 mg to 1 g every
6 hours to 8 hours
If a dose of Cefazolin in Dextrose Injection is required that does not equal 1 gram or 2 grams, this product is not recommended, and an alternative formulation of cefazolin should be considered.
It is important that (i) the preoperative dose be given just prior (1/2 hour to 1 hour) to the start of surgery so that adequate antibacterial concentrations are present in the serum and tissues at the time of initial surgical incision; and (ii) cefazolin be administered, if necessary, at appropriate intervals during surgery to provide sufficient concentrations of the antibacterial drug at the anticipated moments of greatest exposure to infective organisms.
The perioperative prophylactic administration of cefazolin should usually be discontinued within a 24-hour period after the surgical procedure. In surgery where the occurrence of infection may be particularly devastating (e.g., open-heart surgery and prosthetic arthroplasty), the prophylactic administration of cefazolin may be continued for 3 to 5 days following the completion of surgery.
Dosage for Perioperative Prophylaxis in Pediatric Patients Aged 10 to 17 Years Old with CLcr 70 mL/min or Greater
To prevent postoperative infection in contaminated or potentially contaminated surgery, recommended doses are described in Table 4 below.
Table 4: Recommended Dosage for Perioperative Prophylaxis in Pediatric with CLcr 70 mL/min or greater Aged 10 to 17 years Old* - *
- If a dose of Cefazolin in Dextrose Injection is required that does not equal 1 gram or 2 grams, this product is not recommended for use and an alternative formulation of cefazolin should be considered
Body weight (kg)
Dose administered ½ hour to 1 hour prior to start of surgery
Additional dose during lengthy operative procedures (e.g., 2 hours or more)
Dose for 24 hours postoperatively
Less than 50 kg
1 g
500 mg to 1 g
500 mg to 1 g every 6 hours to 8 hours
Greater than or equal to 50 kg
2 g
It is important that (i) the preoperative dose be given just prior (1/2 hour to 1 hour) to the start of surgery so that adequate antibacterial concentrations are present in the serum and tissues at the time of initial surgical incision; and (ii) cefazolin be administered, if necessary, at appropriate intervals during surgery to provide sufficient concentrations of the antibacterial drug at the anticipated moments of greatest exposure to infective organisms.
The administration of Cefazolin in Dextrose Injection for perioperative prophylaxis should usually be discontinued within a 24-hour period after the surgical procedure. In surgery where the occurrence of infection may be particularly devastating the administration of Cefazolin in Dextrose Injection for perioperative prophylaxis may be continued for 3 days to 5 days following the completion of surgery.
2.4 Dosage Recommendations in Adult and Pediatric Patients with Renal Impairment
Dosage Recommendations in Adult Patients with CLcr less than 55 mL/min
The dosage recommendation for Cefazolin in Dextrose Injection in adult patients with renal impairment (CLcr less than 55 mL/min) is outlined in Table 5 below.
Table 5: Dosage Recommendation for Adult Patients with CLcr less than 55 mL/min - *
- If the recommended dose in adult patients with creatinine clearance equal to 35 mL/min or greater is 1 gram, then this product is not recommended for use in patients with creatinine clearance less than 35 mL/min and an alternative formulation of cefazolin should be considered.
- Creatinine Clearance
- Dose
- Frequency
- 35 to 54 mL/min
- Recommended dose
- every 8 hours or longer
- 11 to 34 mL/min
- Half of recommended dose*
- every 12 hours
- 10 mL/min or less
- Half of recommended dose*
- every 18 to 24 hours
Dosage Recommendations in Pediatric Patients with CLcr less than 70 mL/min
The dosage recommendation for Cefazolin in Dextrose Injection in pediatric patients with renal impairment (CLcr less than 70 mL/min) is outlined in Table 6 below.
Table 6: Recommended Dosage in Pediatric Patients with CLcr less than 70 mL/min* - *
- If a dose of Cefazolin in Dextrose Injection is required that does not equal 1 gram or 2 grams, this product is not recommended for use and an alternative formulation of cefazolin should be considered.
- Creatinine Clearance
- Recommended Dosage
- 40 to 70 mL/min
- 60% of the normal daily dose given in equally divided doses every 12 hours
- 20 to 40 mL/min
- 25% of the normal daily dose given in equally divided doses every 12 hours
- 5 to 20 mL/min
- 10% of the normal daily dose every 24 hours
- 1.
- If a dose of Cefazolin in Dextrose Injection is required that does not equal 1 gram or 2 grams, this product is not recommended for use and an alternative formulation of cefazolin should be considered.
2.5 Preparation for Use of Cefazolin in Dextrose Injection
Thawing of Plastic Container
- •
- Thaw frozen container at room temperature 20-25°C (68 - 77°F) or under refrigeration 2-8°C (36 - 46°F). Product should not be thawed by immersion in water baths or by microwave irradiation. Do not force thaw.
- •
- No further dilution is necessary.
- •
- Check for minute leaks by squeezing container firmly. If leaks are detected, discard solution as sterility may be impaired.
- •
- Do not add supplementary medication.
- •
- The container should be visually inspected. If the outlet port protector is damaged, detached, or not present, discard container as solution path sterility may be impaired. Components of the solution may precipitate in the frozen state and will dissolve upon reaching room temperature with little or no agitation. Potency is not affected. Agitate after solution has reached room temperature. If after visual inspection the solution remains cloudy or if an insoluble precipitate is noted or if any seals are not intact, the container should be discarded.
- •
- The thawed solution is stable for 30 days under refrigeration (5°C/41°F) or 48 hours at 25°C/77°F. Do not refreeze thawed antibacterial drugs.
Preparation for Administration
- •
- Suspend container from support.
- •
- Remove protector from outlet port at bottom of container.
- •
- Attach Intravenous administration set to outlet port. Refer to the manufacturer’s instructions accompanying the administration set for complete directions.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
4.1 Hypersensitivity to Cefazolin or the Cephalosporin Class of Antibacterial Drugs, Penicillins, or Other Beta-lactams
Cefazolin in Dextrose Injection is contraindicated in patients who have a history of immediate hypersensitivity reactions (e.g., anaphylaxis, serious skin reactions) to cefazolin or the cephalosporin class of antibacterial drugs, penicillins, or other beta-lactams [see Warnings and Precautions (5.1)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions to Cefazolin, Cephalosporins, Penicillins, or Other Beta-lactams
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving beta-lactam antibacterial drugs. Before therapy with Cefazolin in Dextrose Injection is instituted, careful inquiry should be made to determine whether the patient has had previous immediate hypersensitivity reactions to cefazolin, cephalosporins, penicillins, or carbapenems. Exercise caution if this product is to be given to penicillin-sensitive patients because cross-hypersensitivity among beta-lactam antibacterial drugs has been clearly documented and may occur in up to 10% of patients with a history of penicillin allergy. If an allergic reaction to Cefazolin in Dextrose Injection occurs, discontinue the drug.
5.2 Seizures in Patients with Renal Impairment
Seizures may occur with the administration of Cefazolin in Dextrose Injection, particularly in patients with renal impairment when the dosage is not reduced appropriately. Discontinue Cefazolin in Dextrose Injection if seizures occur or make appropriate dosage adjustments in patients with renal impairment [see Dosage and Administration (2.4)]. Anticonvulsant therapy should be continued in patients with known seizure disorders.
5.3 Clostridioides difficile-associated Diarrhea
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including cefazolin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B, which contribute to the development of CDAD. Hypertoxin-producing isolates of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial drug treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5.4 Hypersensitivity to Dextrose-containing Products
Hypersensitivity reactions, including anaphylaxis, have been reported with administration of dextrose- containing products. These reactions have been reported in patients receiving high concentrations of dextrose (i.e. 50% dextrose)1. The reactions have also been reported when corn-derived dextrose solutions were administered to patients with or without a history of hypersensitivity to corn products.2
5.5 Prothrombin Activity
Cefazolin in Dextrose Injection may be associated with a fall in prothrombin activity. Those at risk include patients with renal or hepatic impairment or poor nutritional state, as well as patients receiving a protracted course of antimicrobial therapy, and patients previously stabilized on anticoagulant therapy. Prothrombin time should be monitored in patients at risk and exogenous vitamin K administered as indicated.
5.6 Risk of Development of Drug-resistant Bacteria
Prescribing Cefazolin in Dextrose Injection in the absence of proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
As with other antimicrobials, prolonged use of Cefazolin in Dextrose Injection may result in overgrowth of nonsusceptible microorganisms. Repeated evaluation of the patient's condition is essential. Should superinfection occur during therapy, appropriate measures should be taken.
5.7 Drug/Laboratory Test Interactions
Urinary Glucose
The administration of cefazolin may result in a false-positive reaction with glucose in the urine when using glucose tests based on Benedict’s copper reduction reaction that determine the amount of reducing substances like glucose in the urine. It is recommended that glucose tests based on enzymatic glucose oxidase be used.
Coombs’ Test
Positive direct Coombs' tests have been reported during treatment with cefazolin. In hematologic studies or in transfusion cross-matching procedures when antiglobulin tests are performed on the minor side or in Coombs' testing of newborns whose mothers have received cephalosporin antibacterial drugs before parturition, it should be recognized that a positive Coombs' test may be due to the drug.
5.8 Patients with Overt or Known Subclinical Diabetes Mellitus or Carbohydrate Intolerance
As with other dextrose-containing solutions, Cefazolin in Dextrose Injection should be prescribed with caution in patients with overt or known subclinical diabetes mellitus or carbohydrate intolerance for any reason.
-
6 ADVERSE REACTIONS
The following serious adverse reactions to Cefazolin in Dextrose Injection are described below and elsewhere in the labeling:
- •
- Hypersensitivity reactions [see Warnings and Precautions (5.1)]
- •
- Seizures in Patients with Renal Impairment [see Warnings and Precautions (5.2)]
- •
- Clostridioides difficile-associated diarrhea [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The following adverse reactions were reported from clinical trials:
Gastrointestinal: Diarrhea, oral candidiasis (oral thrush), mouth ulcers, vomiting, nausea, stomach cramps, epigastric pain, heartburn, flatus, anorexia and pseudomembranous colitis. Onset of pseudomembranous colitis symptoms may occur during or after antibacterial treatment [see Warnings and Precautions (5.3)].
Allergic: Anaphylaxis, eosinophilia, urticaria, itching, drug fever, skin rash, Stevens-Johnson syndrome.
Hematologic: Neutropenia, leukopenia, thrombocytopenia, thrombocythemia.
Hepatic: Transient rise in SGOT, SGPT, and alkaline phosphatase levels has been observed. Reports of hepatitis have been received.
Renal: Reports of increased BUN and creatinine levels, as well as renal failure, have been received.
Local Reactions: Instances of phlebitis have been reported at site of injection. Some induration has occurred.
Other Reactions: Pruritus (including genital, vulvar and anal pruritus, genital moniliasis, and vaginitis). Dizziness, fainting, lightheadedness, confusion, weakness, tiredness, hypotension, somnolence and headache.
Adverse Reactions in Pediatric Patients for Perioperative Prophylaxis
Two studies (Study 1: NCT 3231228 and Study 2: NCT 01904357) were conducted to assess the safety and pharmacokinetics of a single 30-minute infusion of either 1 gram or 2 grams (based on weight) of cefazolin for injection for perioperative prophylaxis in pediatric patients.
Study 1 was a multicenter, open-label, non-comparative, parallel group study to evaluate the safety and pharmacokinetics of a single 30-minute infusion of either 1 gram or 2 grams (based on weight) of cefazolin for injection for perioperative prophylaxis in 61 pediatric patients 10 to 17 years of age. Thirty-three subjects with a weight of at least 25 kg but less than 60 kg received a single dose of 1 gram of cefazolin and 28 subjects with a weight of at least 60 kg received a single dose of 2 grams of cefazolin. The mean age of the safety population was 14 years and ranged from 10 to 17 years. There were no adverse reactions leading to study discontinuation or deaths reported during the study. The most frequently reported adverse reactions were nausea (14.8%), infusion site pain (6.6%), and headache (4.9%).
Study 2 was a multicenter, non-comparative study that evaluated the safety and pharmacokinetics of a single 30-minute infusion of either 1 gram or 2 grams (based on weight) of cefazolin for injection for perioperative prophylaxis in 12 pediatric patients 10 to 12 years of age. Subjects weighing at least 25 kg to less than 50 kg received a single dose of 1 gram of cefazolin for injection and subjects weighing at least 50 kg to less than 85 kg received a single dose of 2 grams of cefazolin for injection. The safety findings in Study 2 in pediatric patients aged 10 to 12 years old were similar to those observed in adult patients and the pediatric patients aged 10 to 17 years old in Study 1.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of cefazolin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: Serum sickness-like reaction
Renal and urinary disorders: Acute tubulointerstitial nephritis (ATIN)
Skin and subcutaneous tissue disorders: Acute generalized exanthematous pustulosis (AGEP)
6.3 Cephalosporin-class Adverse Reactions
In addition to the adverse reactions listed above that have been observed in patients treated with cefazolin, the following adverse reactions and altered laboratory tests have been reported for cephalosporin-class antibacterials:
Stevens-Johnson syndrome, erythema multiforme, toxic epidermal necrolysis, renal impairment, toxic nephropathy, aplastic anemia, hemolytic anemia, hemorrhage, a fall in prothrombin activity, hepatic impairment including cholestasis, and pancytopenia.
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from published prospective cohort studies, case series and case reports over several decades with cephalosporin use, including cefazolin, in pregnant women have not established a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Cefazolin crosses the placenta.Animal reproduction studies with rats, mice and rabbits administered cefazolin during organogenesis at doses 1 to 3 times the maximum recommended human dose (MRHD) did not demonstrate adverse developmental outcomes. In rats subcutaneously administered cefazolin prior to delivery and throughout lactation, there were no adverse effects on offspring at a dose approximately 2 times the MRHD (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
While available studies cannot definitively establish the absence of risk, published data from case-control studies and case reports over several decades have not identified an association with cephalosporin use during pregnancy and major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Available studies have methodologic limitations, including small sample size, retrospective data collection, and inconsistent comparator groups.
Animal Data
Reproduction studies have been performed in rats, mice and rabbits administered cefazolin during organogenesis at doses of 2000, 4000 and 240 mg/kg/day (approximately 1 to 3 times the maximum recommended human dose on a body surface area comparison). There was no evidence of any adverse effects on embryofetal development due to cefazolin. In a peri‑postnatal study in rats, cefazolin administered subcutaneously up to 1200 mg/kg/day (approximately 2 times the MRHD based on body surface area comparison) to pregnant dams prior to delivery and through lactation caused no adverse effects on offspring.
8.2 Lactation
Risk Summary
Data from published literature report that cefazolin is present in human milk, but is not expected to accumulate in a breastfed infant. There are no data on the effects of cefazolin on the breastfed child or on milk production.The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Cefazolin in Dextrose Injection and any potential adverse effects on the breastfed child from Cefazolin in Dextrose Injection or from the mother’s underlying condition.
8.4 Pediatric Use
Cefazolin in Dextrose Injection is indicated for the treatment of respiratory tract infections, urinary tract infections, skin and skin structure infections, biliary tract infections, bone and joint infections, genital infections, septicemia, and endocarditis in pediatric patients for whom appropriate dosing with this formulation can be achieved and for perioperative prophylaxis in pediatric patients aged 10 to 17 years old [see Indications and Usage (1.1 to 1.8)].
Safety and effectiveness of Cefazolin in Dextrose Injection in premature infants and neonates have not been established and is not recommended for use in this age group of pediatric patients. Dosing for cefazolin in pediatric patients younger than one month old has not been established.
Because of the limitations of the available strengths and administration requirements (i.e., administration of fractional doses is not recommended) of Cefazolin in Dextrose Injection, and to avoid unintentional overdose, this product is not recommended for use if a dose of Cefazolin in Dextrose Injection that does not equal 1 gram or 2 grams is required and an alternative formulation of cefazolin should be considered [see Dosage and Administration (2.2, 2,3, 2.4 and 2.5)].
The safety and effectiveness of Cefazolin in Dextrose Injection for perioperative prophylaxis have not been established in pediatric patients younger than 10 years old.
The safety and effectiveness of Cefazolin in Dextrose Injection for perioperative prophylaxis have been established in pediatric patients aged 10 to 17 years old. Use of Cefazolin in Dextrose Injection in these age groups is supported by evidence from adults with additional safety and pharmacokinetic data in pediatric patients aged 10 to 17 years old. Safety and pharmacokinetics were evaluated in two multicenter, non-comparative studies (Study 1 and Study 2). These studies were conducted to assess the safety and pharmacokinetics of a single 30-minute infusion of either 1 gram or 2 grams (based on weight) of Cefazolin in Dextrose Injection for perioperative prophylaxis in pediatric patients. Study 1 evaluated the safety and pharmacokinetics of 1 g of cefazolin for injection in pediatric patients aged 10 to 17 years old scheduled for surgery with a weight of at least 25 kg but less than 60 kg and, 2 g in pediatric patients with a weight of at least 60 kg. Study 2 evaluated 1 g of cefazolin for injection in pediatric patients aged 10 to 12 years old scheduled for surgery with a weight of at least 25 kg but less than 50 kg and, 2 g in pediatric patients with a weight of at least 50 kg to less than 85 kg [see Dosage and Administration (2.3), Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
The safety and effectiveness of Cefazolin in Dextrose Injection for perioperative prophylaxis have not been established in pediatric patients younger than 10 years old.
8.5 Geriatric Use
Of the 920 subjects who received cefazolin in clinical studies, 313 (34%) were 65 years and over, while 138 (15%) were 75 years and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.3) and Warnings and Precautions (5.2)].
8.6 Patients with Renal Impairment
When Cefazolin in Dextrose Injection is administered to adult and pediatric patients with low urinary output because of impaired renal function (creatinine clearance less than 55 mL/min and 70 mL/min for adults and pediatric patients, respectively), lower daily dosage is required [see Dosage and Administration (2.4) and Warnings and Precautions (5.2)].
-
10 OVERDOSAGE
Accidental overdosage resulting in seizures may occur in patients with renal impairment who receive doses greater than the recommended dosage of Cefazolin in Dextrose Injection [see Warnings and Precautions (5.2)]. If seizures associated with accidental overdosage occur, discontinue Cefazolin in Dextrose Injection and give supportive treatment.
-
11 DESCRIPTION
Cefazolin in Dextrose Injection is a frozen, premixed, iso-osmotic, sterile, nonpyrogenic, single-dose solution containing either 1g Cefazolin, USP, equivalent to 1.05 g Cefazolin Sodium, USP per 50 mL or 2 g Cefazolin, USP, equivalent to 2.1 g Cefazolin Sodium, USP per 100 mL GALAXY container (PL 2040 Plastic). Dextrose, USP has been added to adjust osmolality (4 % as dextrose hydrous). The approximate osmolality for Cefazolin in Dextrose Injection is 290 mOsmol/kg.
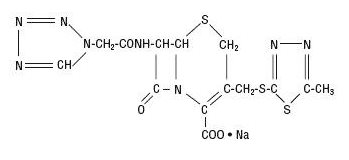
Cefazolin Sodium is a semi-synthetic cephalosporin antibacterial for parenteral administration and has the following IUPAC nomenclature: Sodium (6R,7R)-3-[[(5-methyl-1,3,4- thiadiazol-2-yl)thio]methyl]-8-oxo-7-[2-(1H-tetrazol-1-yl)acetamido]-5-thia-1-azabicyclo[4.2.0]oct-2- ene-2-carboxylate. Its molecular formula is C14H13N8O4S3·Na and its molecular weight is 454.51 (free acid).
Cefazolin Sodium USP has the following structural formula:
The sodium content is 48 mg/g of cefazolin sodium.
The pH of Cefazolin in Dextrose Injection may have been adjusted with sodium bicarbonate, or sodium hydroxide/hydrochloric acid during manufacture. Water for injection, USP is added as drug vehicle. Contains no preservative. The solution is intended for intravenous use after thawing to room temperature.
This GALAXY container (PL 2040 Plastic) is fabricated from a specially designed multilayer plastic (PL 2040). Solutions are in contact with the polyethylene layer of this container and can leach out certain chemical components of the plastic in very small amounts within the expiration period. However, the suitability of the plastic has been confirmed in tests in animals according to the USP biological tests for plastic containers, as well as by tissue culture toxicity studies.
-
12 CLINICAL PHARMACOLOGY
12.2 Pharmacodynamics
The pharmacokinetic/pharmacodynamic relationship for cefazolin has not been evaluated in patients.
12.3 Pharmacokinetics
Studies have shown that following intravenous administration of cefazolin to normal volunteers, mean serum concentrations peaked at approximately 185 mcg/mL and were approximately 4 mcg/mL at 8 hours for a 1 gram dose.
The serum half-life for cefazolin is approximately 1.8 hours following IV administration.
In a study of constant intravenous infusion with dosages of 3.5 mg/kg for 1 hour (approximately 250 mg) and 1.5 mg/kg the next 2 hours (approximately 100 mg), cefazolin serum concentrations at the third hour of approximately 28 mcg/mL.
Plasma pharmacokinetic parameters of cefazolin in healthy volunteers (N=12) following a single 15- minute IV infusion of 2 grams of Cefazolin in Dextrose Injection are summarized in Table 7.
Table 7: Mean (Standard Deviation) Plasma Pharmacokinetic Parameters of Cefazolin in Healthy Volunteers - *
- Tmax reported as median (range)
N
Cmax
(mcg/mL)
Tmax*
(h)
AUC0-inf
(mcg*h/mL)
T1/2
(h)
CL
(L/h)
Vz
(L)
Single 2 grams Dose as a 15- Minute IV Infusion
12
280.9
(45.9)
0.25
(0.25-0.33)
509.9
(89.3)
2.01
(0.28)
4.03
(0.68)
11.50
(1.53)
N= number of subjects observed; Cmax = maximum plasma concentration; Tmax = time to maximum plasma concentration; AUC0-inf = area under the plasma concentration-time curve extrapolated to infinity; t1/2 = apparent plasma terminal elimination half-life; CL = total clearance; Vz = volume of distribution
Studies in patients hospitalized with infections indicate that cefazolin produces mean peak serum concentrations approximately equivalent to those seen in healthy volunteers.
Bile concentrations in patients without obstructive biliary disease can reach or exceed serum concentrations by up to five times; however, in patients with obstructive biliary disease, bile concentrations of cefazolin are considerably lower than serum concentrations (less than 1.0 mcg/mL).
In synovial fluid, the cefazolin concentration becomes comparable to that reached in serum at about 4 hours after drug administration.
Studies of cord blood show prompt transfer of cefazolin across the placenta. Cefazolin is present in very low concentrations in the milk of nursing mothers.
Cefazolin is excreted unchanged in the urine. In the first 6 hours approximately 60% of the drug is excreted in the urine and this increases to 70% to 80% within 24 hours.
Specific Populations
Pediatric Patients for Perioperative Prophylaxis
A simulation based on pharmacokinetic data from healthy adults (n=24), pediatric patients aged 10 to 17 years (n=26: Study 1 [see Adverse Reactions (6.1)]), and pediatric patients aged 10 to 12 years (n=12: Study 2 [see Adverse Reactions (6.1)] indicate that the administration of a 1 gram cefazolin dose for pediatric patients weighing less than 50 kg and a 2 grams cefazolin dose for those weighing 50 kg or greater will provide comparable exposures between pediatric patients aged 10 to 17 years and healthy adults receiving 2 grams Cefazolin in Dextrose Injection [see Dosage and Administration (2.2 and 2.3) and Use in Specific Populations (8.4)].
12.4 Microbiology
Mechanism of Action
Cefazolin is a bactericidal agent that acts by inhibition of bacterial cell wall synthesis.
Resistance
Predominant mechanisms of bacterial resistance to cephalosporins include the presence of extended- spectrum beta-lactamases and enzymatic hydrolysis.
Antimicrobial Activity
Cefazolin has been shown to be active against most isolates of the following microorganisms, both in vitro and in clinical infections [see Indications and Usage (1)].
- Gram-Positive Bacteria
- Staphylococcus aureus
- Staphylococcus epidermidis
- Streptococcus agalactiae
- Streptococcus pneumoniae
- Streptococcus pyogenes
Methicillin-resistant staphylococci are uniformly resistant to cefazolin.
- Gram-Negative Bacteria
- Escherichia coli
- Proteus mirabilis
Most isolates of indole positive Proteus (Proteus vulgaris), Enterobacter spp., Morganella morganii, Providencia rettgeri, Serratia spp., and Pseudomonas spp. are resistant to cefazolin.
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria, and associated test methods and quality control standards recognized by FDA for this drug, please see: http://www.fda.gov/STIC.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity and Mutagenesis
Mutagenicity studies and long-term studies in animals to determine the carcinogenic potential of Cefazolin in Dextrose Injection have not been performed.
Impairment of Fertility
Fertility studies conducted in rats subcutaneously administered cefazolin at doses of
2000 mg/kg/day (approximately 3 times the maximum recommended human dose based on body surface area comparison) showed no impairment of mating and fertility.
- 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
- Cefazolin in Dextrose Injection is supplied as a premixed frozen iso-osmotic solution in 50 mL or 100 mL single-dose GALAXY plastic containers as follows:
- Code
- Product Description
- NDC Number
- 2G3503
- 1 gram cefazolin in 50 mL
- Supplied as 24 bags per carton
- NDC 0338-3503-41
- 2G3508
- 2 grams cefazolin in 100 mL
- Supplied as 12 bags per carton
- NDC 0338-3508-41
Store in a freezer capable of maintaining a temperature of -20°C (-4°F) or below [see Dosage and Administration (2.3)].
Handle frozen product containers with care. Product containers may be fragile in the frozen state.
-
17 PATIENT COUNSELING INFORMATION
Serious Allergic Reactions
Advise patients that allergic reactions, including serious allergic reactions could occur and that serious reactions require immediate treatment and discontinuation of Cefazolin in Dextrose Injection. Patients should report to their health care provider any previous allergic reactions to cefazolin, cephalosporins, penicillins, or other similar antibacterials.
Seizures
Advise patients that seizures could occur with Cefazolin in Dextrose Injection. Instruct patients to inform a healthcare provider at once of any signs and symptoms of seizures, for immediate treatment, dosage adjustment, or discontinuation of Cefazolin in Dextrose Injection.
Diarrhea
Advise patients that diarrhea is a common problem caused by antibacterials, which usually ends when the antibacterial is discontinued. Sometimes after starting treatment with antibacterials, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibacterials. If this occurs, patients should contact a physician as soon as possible.
Antibacterial Resistance
Patients should be counseled that antibacterial drugs, including Cefazolin in Dextrose Injection should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Cefazolin in Dextrose Injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Cefazolin in Dextrose Injection or other antibacterial drugs in the future.
- SPL UNCLASSIFIED SECTION
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
Cefazolin in Dextrose
Injection, USP
1 g / 50 mL
(20 mg / mL)GALAXY
Single Dose Container
Discard unused portionNDC 0338-3503-41
Code 2G3503
Sterile Nonpyrogenic
Iso-osmoticEach 50 mL contains: 1 g Cefazolin, USP, equivalent to 1.05 g Cefazolin
Sodium, USP with approx. 2 g Dextrose Hydrous, USP added to adjust
osmolality, and Water for Injection, USP. pH may have been adjusted with
sodium bicarbonate or sodium hydroxide/hydrochloric acid.Dosage: For Intravenous Infusion Only. See prescribing information.
Cautions: Do not add supplementary medication or additives.Rx only
Store at or below -20°C (-4°F). Thaw at room temperature 20°C to 25°C (68°F to
77°F) or under refrigeration 2°C to 8°C (36°F to 46°F). DO NOT FORCE
THAW BY IMMERSION IN WATER BATHS OR BY MICROWAVE IRRADIATION.
Thawed solution is stable for 30 days under refrigeration or 48 hours at room
temperature. Do not refreeze.Baxter Logo
Baxter and Galaxy are registered trademarks of
Baxter International Inc.
Manufactured by Baxter Healthcare Corporation
Deerfield, IL 60015 USA
Made in USPL2040 Plastic
07-34-00-1642
* BAR CODE POSITION
ONLY303383503411
Thaw at room temperature 20°C to 25°C (68°F to 77°F) or under refrigeration 2°C to 8°C (36°F to 41°F).
DO NOT FORCE THAW BY IMMERSION IN WATER BATHS OR BY MICROWAVE IRRADIATION. Thawed solution is stable for 30 days under refrigeration or 48 hours at room temperature.
Do not refreeze.
Handle frozen product containers with care. Product containers may be fragile in the
frozen state.
Baxter and Galaxy are registered trademarks of Baxter International Inc.
PL 2040 Plastic07-04-00-0801
Cefazolin in Dextrose Injection, USP
1 g / 50 mL (20 mg / mL)
Contains 12 units of Single Dose bags. Each bag contains 50 mL Iso-osmotic
Store at or below -20°C (-4°F). Do not refreeze.Rx Only
NDC 0338-3503-41
Code 2G3503*FOR BAR CODE POSITION ONLY
(01) 20303383503415GALAXY Container
Sterile Nonpyrogenic
Each 50 mL contains: 1g Cefazolin, USP, equivalent to 1.05 g Cefazolin Sodium, USP with approx. 2 g
Dextrose Hydrous, USP added to adjust osmolality, and Water for Injection, USP. pH may have been adjusted
with sodium bicarbonate or sodium hydroxide/hydrochloric acid.Dosage: For Intravenous Infusion Only. See prescribing information.
Cautions: Do not add supplementary medications or additives.
Baxter Logo
Baxter and Galaxy are registered trademarks of Baxter International, Inc.
Baxter Healthcare Corporation Deerfield, IL 60015 USACefazolin in Dextrose
Injection, USP
2 g / 100 mL
(20 mg / mL)GALAXY
Single Dose Container
Discard unused portionNDC 0338-3508-41
Code 2G3508
Sterile Nonpyrogenic
Iso-osmoticEach 100 mL contains: 2 g Cefazolin, USP, equivalent to 2.1 g Cefazolin
Sodium, USP with approx. 4 g Dextrose Hydrous, USP added to adjust
osmolality, and Water for Injection, USP. pH may have been adjusted with
sodium bicarbonate or sodium hydroxide/hydrochloric acid.Dosage: For Intravenous Infusion Only. See prescribing information.
Cautions: Do not add supplementary medication or additives.Rx only
Store at or below -20°C (-4°F). Thaw at room temperature 20°C to 25°C (68°F to
77°F) or under refrigeration 2°C to 8°C (36°F to 46°F). DO NOT FORCE THAW
BY IMMERSION IN WATER BATHS OR BY MICROWAVE IRRADIATION. Thawed
solution is stable for 30 days under refrigeration or 48 hours at room
temperature. Do not refreeze.Baxter Logo
Baxter and Galaxy are registered trademarks of Baxter International Inc.
Manufactured by Baxter Healthcare Corporation Deerfield, IL 60015 USA
Made in USAPL2040 Plastic
07-34-00-1643
* BAR CODE POSITION
ONLY303383508416
Thaw at room temperature 20°C to 25°C (68°F to 77°F) or under refrigeration 2°C to 8°C (36 °F to 41°F).
DO NOT FORCE THAW BY IMMERSION IN WATER BATHS OR BY MICROWAVE IRRADIATION. Thawed solution is stable for 30 days under refrigeration or 48 hours at room temperature.
Do not refreeze.
Handle frozen product containers with care. Product containers may be fragile in the
frozen state.
PL 2040 Plastic
07-04-00-0802Cefazolin in Dextrose Injection, USP
2 g / 100 mL (20 mg/ mL)
Contains 6 units of Single Dose bags. Each bag contains 100 mL. Iso-osmotic
Store at or below -20°C (4°F). Do not refreeze.
Lot XXXXXX Exp. DD MMM YYRx Only
NDC 0338-3508-41
Code 2G3508*FOR BAR CODE POSITION ONLY
(01) 20303383508410GALAXY Container
Sterile Nonpyrogenic
Each 100 mL contains: 2 g Cefazolin, USP, equivalent to 2.1 g Cefazolin Sodium, USP with approx. 4 g
Dextrose Hydrous, USP added to adjust osmolality, and Water for Injection, USP. pH may have been
adjusted with sodium bicarbonate or sodium hydroxide/hydrochloric acid.Dosage: For Intravenous Infusion Only. See prescribing information.
Cautions: Do not add supplementary medications or additives.Baxter Logo
Baxter and Galaxy are registered trademarks of Baxter International, Inc.
Baxter Healthcare Corporation Deerfield, IL 60015 USA -
INGREDIENTS AND APPEARANCE
CEFAZOLIN
cefazolin sodium injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0338-3508 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CEFAZOLIN SODIUM (UNII: P380M0454Z) (CEFAZOLIN - UNII:IHS69L0Y4T) CEFAZOLIN 2 g in 100 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS DEXTROSE (UNII: 5SL0G7R0OK) 4 g in 100 mL SODIUM BICARBONATE (UNII: 8MDF5V39QO) WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0338-3508-41 12 in 1 CASE 08/07/2015 1 100 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA207131 08/07/2015 CEFAZOLIN
cefazolin sodium injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0338-3503 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CEFAZOLIN SODIUM (UNII: P380M0454Z) (CEFAZOLIN - UNII:IHS69L0Y4T) CEFAZOLIN 1 g in 50 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS DEXTROSE (UNII: 5SL0G7R0OK) 2 g in 50 mL SODIUM BICARBONATE (UNII: 8MDF5V39QO) WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0338-3503-41 24 in 1 CASE 08/07/2015 1 50 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA207131 08/07/2015 Labeler - Baxter Healthcare Corporation (005083209) Establishment Name Address ID/FEI Business Operations Baxter Healthcare Corporation 194684502 ANALYSIS(0338-3508, 0338-3503) , LABEL(0338-3508, 0338-3503) , MANUFACTURE(0338-3508, 0338-3503) , PACK(0338-3508, 0338-3503) , STERILIZE(0338-3508, 0338-3503)