Label: ENDARI- glutamine powder, for solution
- NDC Code(s): 42457-420-01, 42457-420-60
- Packager: Emmaus Medical, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated August 19, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ENDARI™ safely and effectively. See full prescribing information for ENDARI.
ENDARI (L-glutamine oral powder )
Initial U.S. Approval: 2017INDICATIONS AND USAGE
ENDARI is an amino acid indicated to reduce the acute complications of sickle cell disease in adult and pediatric patients 5 years of age and older. (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Oral Powder: 5 grams of L-glutamine powder per paper-foil-plastic laminate packet. (3)
CONTRAINDICATIONS
None (4)
ADVERSE REACTIONS
Most common adverse reactions (incidence > 10%) are constipation, nausea, headache, abdominal pain, cough, pain in extremity, back pain, and chest pain. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Emmaus Medical, Inc. at 1-877-420-6493 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 10/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
2.2 Preparation of Product
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 Clinical Studies
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
Administer Endari orally, twice per day at the dose based on body weight according to Table 1.
Table 1. Recommended Dosing Weight in kilograms Weight in pounds Per dose in grams Per day in grams Packets per dose Packets per day less than 30 less than 66 5 10 1 2 30 to 65 66 to 143 10 20 2 4 greater than 65 greater than 143 15 30 3 6 - 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to Endari in 187 patients, including 136 exposed for 6 months and 109 exposed for ≥1 year. Endari was studied in 2 placebo-controlled clinical trials (a phase 3 study, n=230 and a phase 2 study, n=70). In these trials, patients with sickle cell anemia or sickle β0-thalassemia were randomized to receive Endari (n=187) or placebo (n=111) orally twice daily for 48 weeks followed by 3 weeks of tapering. Both studies included pediatric and adult patients (5-58 years of age) and 54% were female. The majority of patients were black (97.3%), had a diagnosis of sickle cell anemia (89.9%) and were receiving hydroxyurea at baseline (63.4%).
Treatment discontinuation due to adverse reactions was reported in 2.7% (n=5) of patients receiving Endari. These adverse reactions included one case each of hypersplenism, abdominal pain, dyspepsia, burning sensation, and hot flash.
Serious adverse reactions were reported in both treatment groups, more frequently in the placebo group, and were consistent with the underlying disease.
Three deaths (3/187=1.6%) occurred during the study in the Endari treatment group as compared to none in the placebo treatment group. None of the deaths were considered to be related to Endari treatment. Adverse reactions occurring in greater than 10% of patients treated with Endari are shown in Table 2 below.
Table 2. Adverse Reactions Occurring at an Incidence > 10% in Clinical Studies of Endari Adverse reaction Endari
N = 187
(%)Placebo
N = 111
(%)- *
- Abdominal pain = abdominal pain and abdominal pain, upper
Constipation 21 18 Nausea 19 14 Headache 18 15 Abdominal Pain* 17 16 Cough 16 14 Pain in extremity 13 7 Back pain 12 5 Chest pain 12 8 -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data on Endari use in pregnant women to inform a drug-associated risk of major birth defects and miscarriage. Animal reproduction studies were not conducted with Endari.
Adverse outcomes in pregnancy occur regardless of the health of the mother or the use of medications. The background risk of major birth defects and miscarriage for the indicated population are unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data on the presence of Endari in human milk, the effect on the breastfed infant or the effect on milk production. The developmental and health benefits from breastfeeding should be considered along with the mother's clinical need for Endari and any potential adverse effects on the breastfed child from Endari or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of Endari have been established in pediatric patients 5 years and older. Use of Endari is supported by evidence from 2 placebo-controlled studies in adult and pediatric patients with sickle cell disease. The clinical studies enrolled 110 pediatric patients in the following age groups: 46 children (5 years up to less than 12 years) and 64 adolescents (12 years to less than 17 years).
The safety and effectiveness of Endari in pediatric patients with sickle cell disease younger than 5 years old has not been established.
8.5 Geriatric Use
Clinical studies of Endari did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
- 10 OVERDOSAGE
-
11 DESCRIPTION
Endari (L-glutamine) is an amino acid. L-glutamine is designated chemically as (S)-2-aminoglutaramic acid, L-glutamic acid 5-amide, or (S)-2,5-diamino-5-oxopentanoic acid. The molecular formula is C5H10N2O3 with the molecular weight of 146.15 g/mol and the following structural formula:
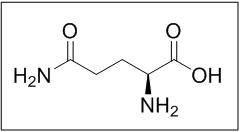
Endari is formulated as a white crystalline powder and is packaged as 5 grams in a paper-foil-plastic laminate packet for oral administration.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism of action of the amino acid L-glutamine in treating sickle cell disease (SCD) is not fully understood. Oxidative stress phenomena are involved in the pathophysiology of SCD. Sickle red blood cells (RBCs) are more susceptible to oxidative damage than normal RBCs, which may contribute to the chronic hemolysis and vaso-occlusive events associated with SCD. The pyridine nucleotides, NAD+ and its reduced form NADH, play roles in regulating and preventing oxidative damage in RBCs. L-glutamine may improve the NAD redox potential in sickle RBCs through increasing the availability of reduced glutathione.
12.2 Pharmacodynamics
In vivo analyses demonstrated that L-glutamine supplementation improved NAD redox potential.
12.3 Pharmacokinetics
The pharmacokinetics of L-glutamine has been studied in healthy subjects and a variety of disease states. Relevant results from published literature are summarized below.
Absorption
Following single-dose oral administration of L-glutamine at 0.1 g/kg, mean peak L-glutamine concentration was 1028 µM (or 150 mcg/mL) occurring approximately 30 minutes after administration. The pharmacokinetics following multiple oral doses has not been characterized.
Distribution
After an intravenous (IV) bolus dose, the volume of distribution was estimated to be approximately 200 mL/kg.
Elimination
After an intravenous bolus dose, the terminal half-life of L-glutamine was approximately one hour.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of L-glutamine.
L-glutamine was not mutagenic in a bacterial mutagenicity (Ames) assay, nor clastogenic in a chromosome aberration assay in mammalian (Chinese Hamster Lung CHL/IU) cells.
Animal reproduction studies and its potential for impairment of fertility have not been conducted with L-glutamine . It is also not known whether L-glutamine can cause fetal harm when administered to a pregnant woman or whether it can affect reproductive capacity.
-
14 Clinical Studies
The efficacy of Endari in sickle cell disease was evaluated in a randomized, double-blind, placebo-controlled, multi-center clinical trial entitled "A Phase III Safety and Efficacy Study of L-Glutamine to Treat Sickle Cell Disease or Sickle βo-thalassemia" [NCT01179217] (see Table 3).
The clinical trial evaluated the efficacy and safety of Endari in 230 patients (5 to 58 years of age) with sickle cell anemia or sickle β0-thalassemia who had 2 or more painful crises within 12 months prior to enrollment. Eligible patients stabilized on hydroxyurea for at least 3 months continued their therapy throughout the study. The trial excluded patients who had received blood products within 3 weeks, had renal insufficiency or uncontrolled liver disease, or were pregnant (or planning pregnancy) or lactating. Study patients received Endari or placebo for a treatment duration of 48 weeks followed by 3 weeks of tapering.
Efficacy was demonstrated by a reduction in the number of sickle cell crises through Week 48 and prior to the start of tapering among patients that received Endari compared to patients who received placebo. This clinical benefit was observed irrespective of hydroxyurea use. A sickle cell crisis was defined as a visit to an emergency room/medical facility for sickle cell disease-related pain which was treated with a parenterally administered narcotic or parenterally administered ketorolac. In addition, the occurrence of chest syndrome, priapism, and splenic sequestration were considered sickle cell crises. Treatment with Endari also resulted in fewer hospitalizations due to sickle cell pain at Week 48, fewer cumulative days in hospital and a lower incidence of acute chest syndrome.
Table 3. Results from the Endari Clinical Trial in Sickle Cell Disease Event Endari
(n = 152)Placebo
(n = 78)Median number of sickle cell crises (min,max)* 3 (0, 15) 4 (0, 15) Median number of hospitalizations for sickle cell pain (min, max)* 2 (0, 14) 3 (0, 13) Median cumulative days hospitalized (min, max)* 6.5 (0, 94) 11 (0, 187) Median time (days) to first sickle cell crisis (95% CI)*,† 84 (62, 109) 54 (31, 73) Patients with occurrences of acute chest syndrome (%)* 13 (8.6%) 18 (23.1%) The recurrent crisis event time analysis (Figure 1) yielded an intensity rate ratio (IRR) value of 0.75 with 95% CI= (0.62, 0.90) and (0.55, 1.01) based on unstratified models using the Andersen-Gill and Lin, Wei, Yang and Ying methods, respectively in favor of Endari, suggesting that over the entire 48-week period, the average cumulative crisis count was reduced by 25% from the Endari group over the placebo group.
Figure 1. Recurrent Event Time for Sickle Cell Crises by Treatment Group 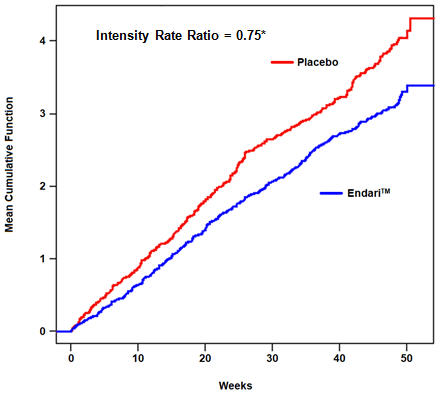
*Andersen-Gill: 95% CI (0.62, 0.90); Lin-Wei-Yang-Ying: 95% CI (0.55, 1.01) - 16 HOW SUPPLIED/STORAGE AND HANDLING
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Instructions for Use).
Dosage and Administration
Advise patient to take a missed dose as soon as they remember. Patient should not double the dose that they take.
Instruct patient to mix each dose in 8 oz. (240 mL) of cold or room temperature beverage or 4 to 6 oz. of food.
Advise patient that complete dissolution is not required prior to administration.
- SPL UNCLASSIFIED SECTION
-
INSTRUCTIONS FOR USE
ENDARI® (en-dar-ee)
(L-glutamine oral powder)Read this Instructions for Use before you start taking Endari and each time you get a refill. There may be new information. This Instructions for Use does not take the place of talking to your healthcare provider about your medical condition or treatment. You and your healthcare provider should talk about Endari before you start taking it and at regular checkups.
Endari is usually taken 2 times a day. Take Endari as prescribed by your healthcare provider.
You will need the following supplies to mix and take Endari: - Your prescribed dose of Endari (1, 2, or 3 packets as directed by your healthcare provider) .
- a clean cup or small bowl
- a spoon
- with a liquid, such as water, milk, or apple juice Or
- with food, such as applesauce or yogurt
How to mix and take a dose of Endari. Mixing with Liquid Mixing with Food Step 1:
Fill a cup with 8 ounces (240 mL) of liquid or a small bowl with 4 to 6 ounces of food.
The food or liquid should be cold or room temperature.
Do not use a hot food or liquid.

Step 2:
Find the perforations at the top of each side of the Endari packet.
Use the perforations to fully tear open each Endari packet.

Step 3:
Pour the contents of the Endari packet into the cup or bowl.
If more than 1 packet is needed, repeat steps 2 and 3 above for all of the packets needed to prepare your prescribed does of Endari.
Step 4:
Use the spoon to mix the prescribed dose of Endari with the liquid or food.
Endari may not fully dissolve. You can take your dose of Endari even if it does not fully dissolve.
Step 5:
Drink or eat the prescribed dose of Endari right away after mixing it.
Do not store the Endari mixture for later use.

If you miss a dose of Endari, take the missed dose as soon as you remember. Do not double the dose to make up for a missed dose.
How should I store Endari?
- Store Endari at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep Endari away from direct sunlight.
Keep Endari and all medicines out of the reach of children.
Manufactured for:
Emmaus Medical, Inc.
Torrance, CA 90503For more information got to www.EnadriRx.com or call 1-877-420-6493.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Issued: 04/2020
-
PRINCIPAL DISPLAY PANEL - 5 gram Packet Carton
NDC: 42457-420-60
ENDARI™
(L-glutamine oral powder)Contents: 60 Packets (5 grams/packet)
Directions: Mix the contents of each packet with cold or room temperature beverage or food
immediately before dosing.Administer the prescribed amount orally, twice daily, taking no more than 6 packets per day.
Package is not child resistant. Keep this and all drugs out of reach of children.
Store at 20°C to 25°C (68°F to 77°F) away from direct sunlight.
Rx Only
LOT XX XXX XX XX
EXP. MM/YYYYManufactured for Emmaus Medical, Inc.

-
INGREDIENTS AND APPEARANCE
ENDARI
glutamine powder, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:42457-420 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Glutamine (UNII: 0RH81L854J) (Glutamine - UNII:0RH81L854J) Glutamine 5 g Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42457-420-60 60 in 1 CARTON 07/07/2017 1 NDC:42457-420-01 1 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208587 07/07/2017 Labeler - Emmaus Medical, Inc. (784073434) Establishment Name Address ID/FEI Business Operations Ajinomoto do Brazil Industria e Comercio de Almentos Ltda. 914653634 API MANUFACTURE(42457-420) Establishment Name Address ID/FEI Business Operations AndersonBrecon, Inc. 053217022 PACK(42457-420)

