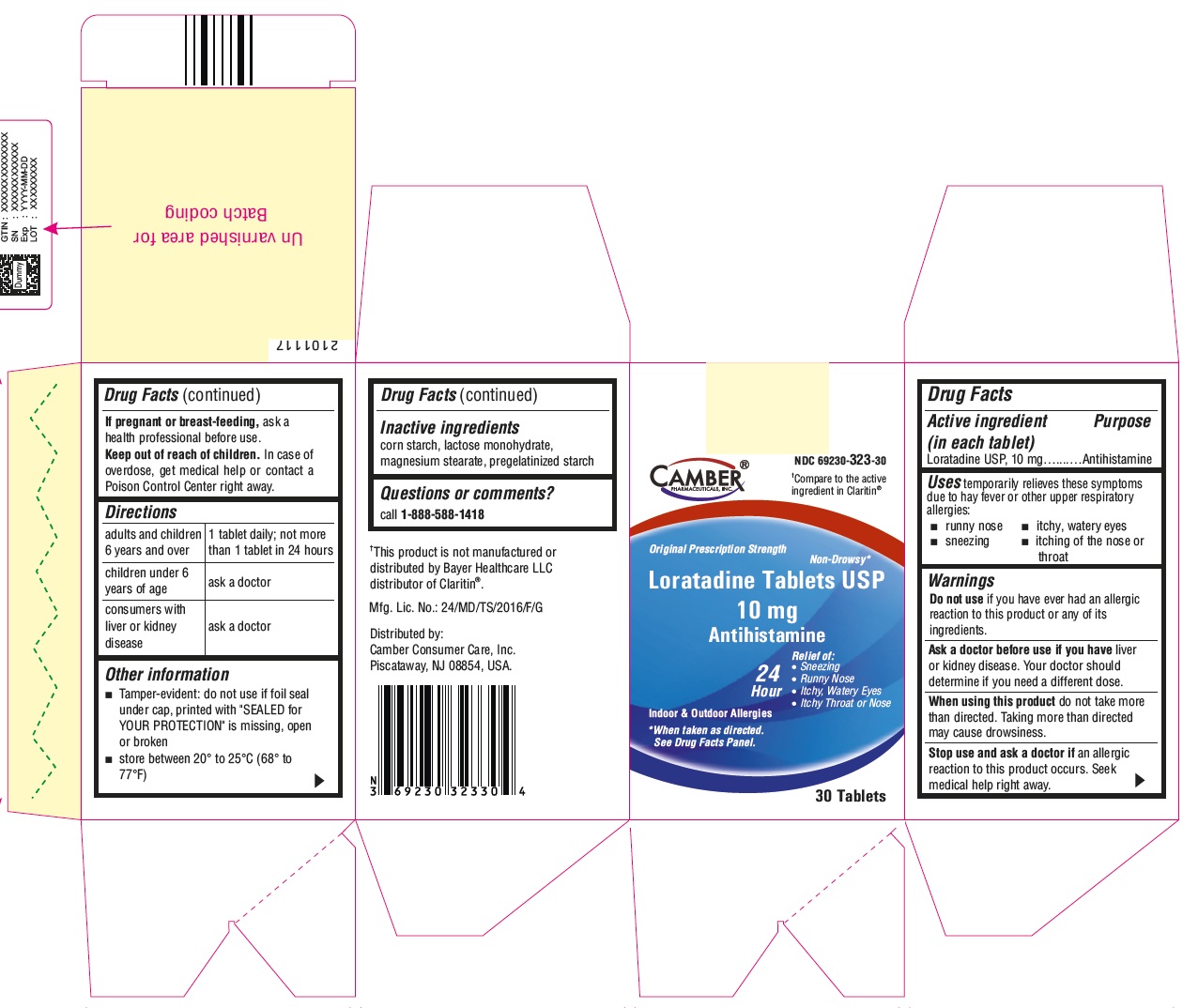
Label: LORATADINE- loratadine tablet
-
NDC Code(s):
69230-323-01,
69230-323-10,
69230-323-30,
69230-323-31, view more69230-323-32, 69230-323-33, 69230-323-34
- Packager: Camber Consumer Care Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated September 8, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT(S)
- PURPOSE
- USE(S)
- WARNINGS
- DO NOT USE
- ASK A DOCTOR BEFORE USE IF YOU HAVE
- ASK A DOCTOR OR PHARMACIST BEFORE USE IF
- WHEN USING THIS PRODUCT
- STOP USE AND ASK DOCTOR IF
- PREGNANCY/BREASTFEEDING
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
-
OTHER INFORMATION
Bottles:
- Tamper-evident: do not use if foil seal under cap, printed with "SEALED for YOUR PROTECTION" is missing, open or broken
- store between 20° to 25°C (68° to 77°F)
Blisters packs:
- safety sealed: do not use if the individual blister unit imprinted with loratadine is open or torn
- store between 20° to 25°C (68° to 77°F)
- protect from excessive moisture
- INACTIVE INGREDIENTS
- QUESTIONS OR COMMENTS?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LORATADINE
loratadine tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69230-323 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 10 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color white (White to Off white) Score no score Shape ROUND Size 6mm Flavor Imprint Code H;L20 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69230-323-30 1 in 1 CARTON 07/28/2023 1 30 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:69230-323-31 1 in 1 CARTON 07/28/2023 2 108 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:69230-323-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 07/28/2023 4 NDC:69230-323-01 10 in 1 CARTON 07/28/2023 4 NDC:69230-323-32 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 5 NDC:69230-323-33 1 in 1 CARTON 07/28/2023 5 100 in 1 BOTTLE; Type 0: Not a Combination Product 6 NDC:69230-323-34 1 in 1 CARTON 07/28/2023 6 300 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211718 07/28/2023 Labeler - Camber Consumer Care Inc (079539968) Establishment Name Address ID/FEI Business Operations Annora Pharma Private Limited 650980746 manufacture(69230-323)