Label: SALICYLIC ACID gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 70350-2610-1, 70350-2610-2 - Packager: SOLUTECH PHARMACEUTICALS LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated March 20, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- DESCRIPTION
-
CLINICAL PHARMACOLOGY
Salicylic acid has been shown to produce desquamation of the horny layer of skin while not effecting qualitative or quantitative changes in the structure of the viable epidermis. The mechanism of action has been attributed to a dissolution of intercellular cement substance. In a study of the percutaneous absorption of salicylic acid in a 6% salicylic acid gel in four patients with extensive active psoriasis, Taylor and Halprin showed that the peak serum salicylate levels never exceeded 5 mg/100 ml even though more than 60% of the applied salicylic acid was absorbed. Systemic toxic reactions are usually associated with much higher serum levels (30 to 40 mg/100 ml). Peak serum levels occurred within five hours of the topical application under occlusion. The sites were occluded for 10 hours over the entire body surface below the neck. Since salicylates are distributed in the extracellular space, patients with a contracted extracellular space due to dehydration or diuretics have higher salicylate levels than those with a normal extracellular space. (See PRECAUTIONS.) The major metabolites identified in the urine after topical administration are salicyluric acid (52%), salicylate glucuronides (42%) and free salicylic acid (6%). The urinary metabolites after percutaneous absorption differ from those after oral salicylate administration; those derived from percutaneous absorption contain more salicylate glucuronides and less salicyluric and salicylic acid. Almost 95% of a single dose of salicylate is excreted within 24 hours of its entrance into the extracellular space. Fifty to eighty percent of salicylate is protein bound to albumin. Salicylates compete with the binding of several drugs and can modify the action of these drugs. By similar competitive mechanisms other drugs can influence the serum levels of salicylate. (See Drug Interactions.)
-
INDICATIONS AND USAGE
For Dermatologic Use
Salicylic Acid 6% is a topical aid in the removal of excessive keratin in hyperkeratotic skin disorders including verrucae, and the various ichthyoses (vulgaris, sex-linked and lamellar), keratosis palmaris and plantaris keratosis pilaris, pityriasis rubra pilaris, and psoriasis (including body, scalp, palms and soles).
- CONTRAINDICATIONS
-
WARNINGS
Prolonged use over large areas, especially in children and those patients with significant renal or hepatic impairment could result in salicylism. Concomitant use of other drugs which may contribute to elevated serum salicylate levels should be avoided where the potential for toxicity is present. In children under 12 years of age and those patients with renal or hepatic impairment, the area to be treated should be limited and the patient monitored closely for signs of salicylate toxicity: nausea, vomiting, dizziness, loss of hearing, tinnitus, lethargy, hyperpnea, diarrhea, and psychic disturbances. In the event of salicylic acid toxicity, the use of Salicylic Acid 6% should be discontinued. Fluids should be administered to promote urinary excretion. Treatment with sodium bicarbonate (oral or intravenous) should be instituted as appropriate. Patients should be cautioned against the use of oral aspirin and other salicylate containing medications, such as sports injury creams, to avoid additional excessive exposure to salicylic acid. Where needed, aspirin should be replaced by an alternative non-steroidal anti-inflammatory agent that is not salicylate based. Patients should be advised not to apply occlusive dressings, clothing or other occlusive topical products such as petrolatum-based ointments to prevent excessive systemic exposure to salicylic acid. Excessive application of the product other than what is needed to cover the affected area will not result in a more rapid therapeutic benefit. Due to potential risk of developing Reye's syndrome, salicylate products should not be used in children and teenagers with varicella or influenza, unless directed by physician.
-
PRECAUTIONS
For external use only. Avoid contact with eyes, lips, broken or inflamed skin and all mucous membranes. Mild burning or stinging may occur. Peeling of the skin may increase as the salicylic acid works to loosen excess keratin. If excessive burning, stinging or peeling occurs, discontinue use and consult your physician. If swallowed, get medical help or contact Poison Control Center right away. KEEP THIS AND ALL MEDICATION OUT OF THE REACH OF CHILDREN.
DRUG INTERACTIONS
The following interactions are from a published review and include reports concerning both oral and topical salicylate administration. The relationship of these interactions to the use of Salicylic Acid 6% is not known.
I.Due to the competition of salicylate with other drugs for binding to serum albumin the following drug interactions may occur: DRUG DESCRIPTION OF INTERACTION Sulfonylureas Hypoglycemia potentiated. Methotrexate Decreases tubular reabsorption; clinical toxicity from methotrexate can result. Oral Anticoagulants Increased bleeding. II.Drugs changing salicylate levels by altering renal tubular reabsorption: DRUG DESCRIPTION OF INTERACTION Corticosteroids Decreases plasma salicylate level; tapering doses of steroids may promote salicylism. Acidifying Agents Increases plasma salicylate levels. Alkanizing Agents Decreased plasma salicylate levels. III.Drugs with complicated interactions with salicylates: DRUG DESCRIPTION OF INTERACTION Heparin Salicylate decreases platelet adhesiveness and interferes with hemostasis in heparin-treated patients. Pyrazinamide Inhibits pyrazinamide-induced hyperuricemia. Uricosuric Agents Effect of probenemide, sulfinpyrazone and phenylbutazone inhibited. The following alterations of laboratory tests have been reported during salicylate therapy: LABORATORY TESTS EFFECT OF SALICYLATES Thyroid Function Decreased PBI; increased t3 uptake. Urinary Sugar False negative with glucose oxidase; false positive with Clinitest with high-dose salicylate therapy (2-5g q.d.). 5-Hydroxyindole acetic acid False negative with fluorometric test. Acetone ketone bodies False positive FeCl3 in Gerhardt reaction; red color persists with boiling. 17-OH corticosteroids False reduced values with >4.8g q.d. salicylate. Vanilmandelic acid False reduced values. Uric Acid May increase or decrease depending on dose. Prothrombin Decreased levels; slightly increased prothrombin time. PREGNANCY
Category C
Salicylic acid has been shown to be teratogenic in rats and monkeys. It is difficult to extrapolate from oral doses of acetylsalicylic acid used in these studies to topical administration as the oral dose to monkeys may represent six times the maximal daily human dose of salicylic acid when applied topically over a large body surface. There are no adequate and well controlled studies in pregnant women. Salicylic Acid 6% should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
NURSING MOTHERS
Because of the potential for serious adverse reactions in nursing infants from the mother's use of Salicylic Acid 6%, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. If used by nursing mothers, it should not be used on the chest area to avoid accidental contamination of the child.
-
ADVERSE REACTIONS
Some adverse effects are: singing, burning and desquamation.
Peeling of the skin may increase as the salicylic acid works to loosen excess keratin. If excessive burning, stinging or peeling occurs, discontinue use and consult your physician.
Excessive erythema and scaling conceivably could result from use on open skin lesions. Call your physician for medical advice about side effects.
-
DOSAGE AND ADMINISTRATION
Apply Salicylic Acid 6% Gel to the affected area at night or as directed by your doctor. Preferably, skin should be hydrated for at least five minutes prior to application. The medication is washed off in the morning and if excessive drying and/or irritation is observed a blend cream or lotion may be applied. Unless hands are being treated, hands should be rinsed thoroughly after application. Once desired result is achieved, the occasional use of Salicylic Acid 6% will usually maintain the remission. Hydration by wet packs or baths prior to application can enhance the effect. Excessive repeated application of Salicylic Acid 6% will not necessarily increase its therapeutic benefit, but could result in increased local intolerance and systemic adverse effects such as salicylism (See WARNINGS.).
- How is Salicylic Acid 6% Gel Supplied
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 40 gm Jar Label
-
INGREDIENTS AND APPEARANCE
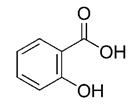
SALICYLIC ACID
salicylic acid gelProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70350-2610 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 60 mg in 1 g Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) HYDROXYETHYL CELLULOSE (3000 MPA.S AT 1%) (UNII: 7Q6P4JN1QT) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70350-2610-2 240 g in 1 JAR; Type 0: Not a Combination Product 06/15/2017 2 NDC:70350-2610-1 40 g in 1 JAR; Type 0: Not a Combination Product 06/15/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug other 06/15/2017 Labeler - SOLUTECH PHARMACEUTICALS LLC (080040396)