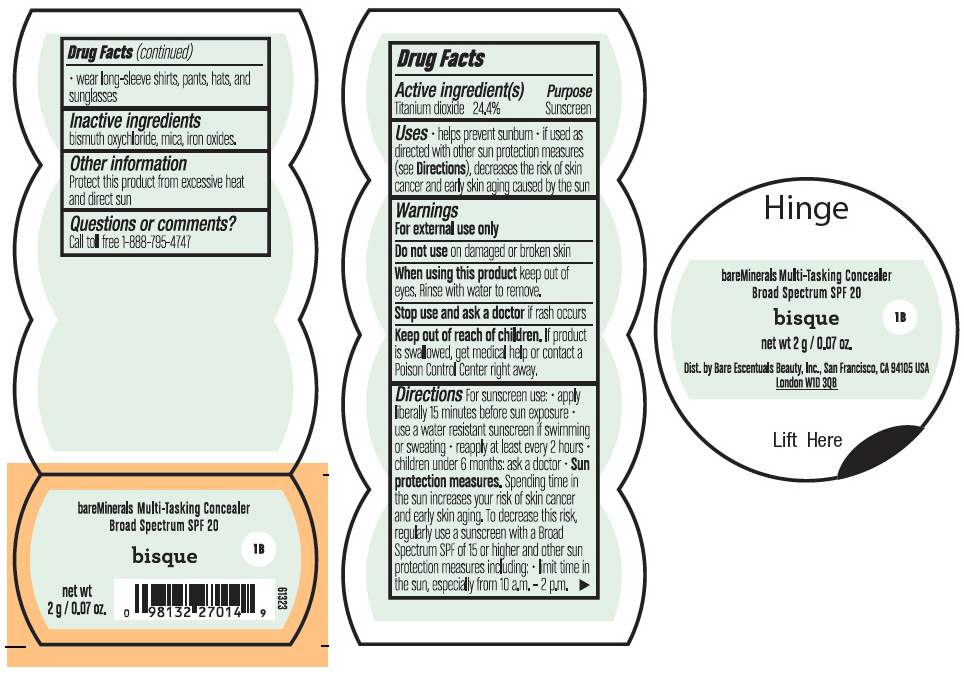
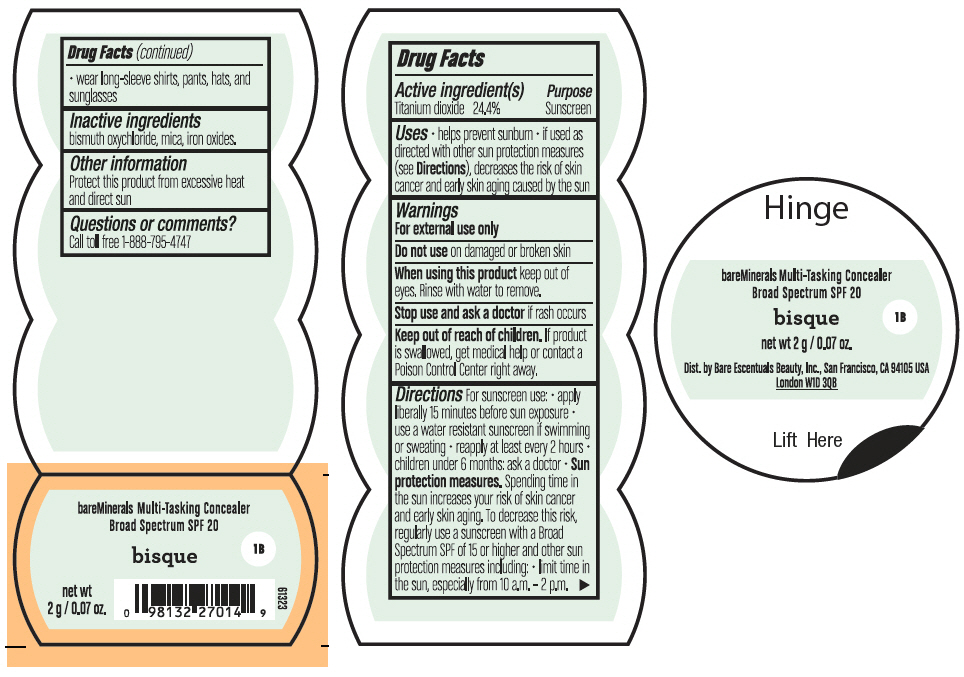
Label: BAREMINERALS MULTI-TASKING SPF 20 CONCEALER BISQUE- titanium dioxide powder
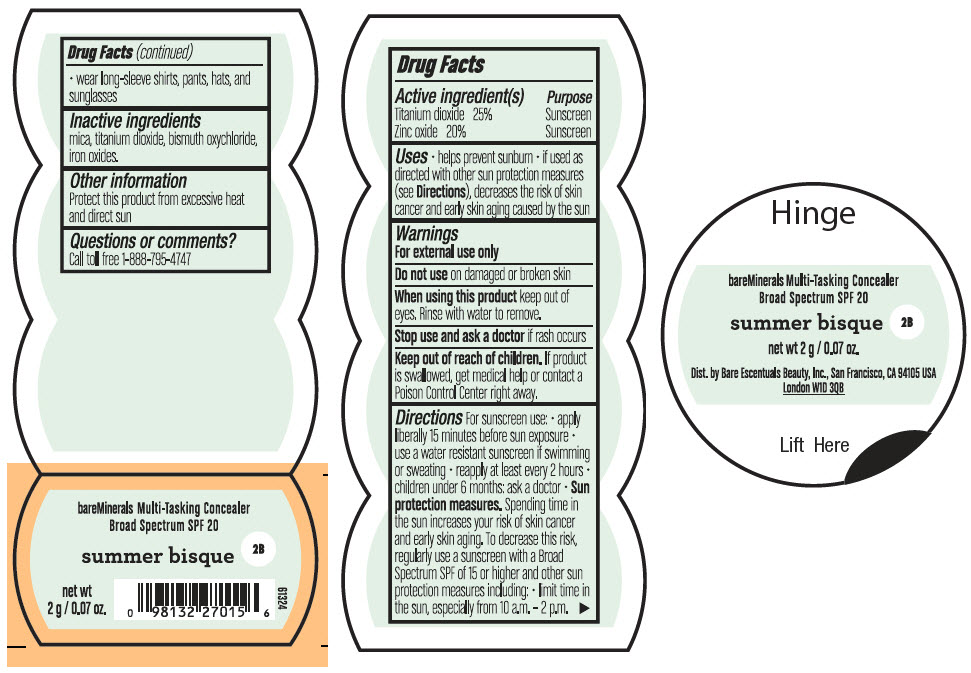
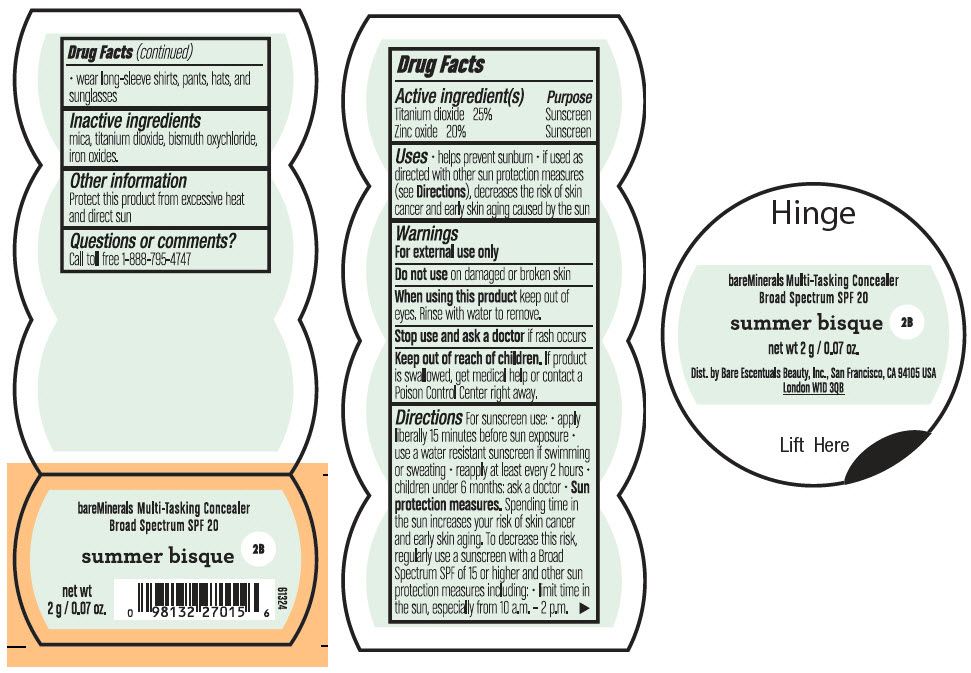
BAREMINERALS MULTI-TASKING SPF 20 CONCEALER SUMMER BISQUE- titanium dioxide and zinc oxide powder
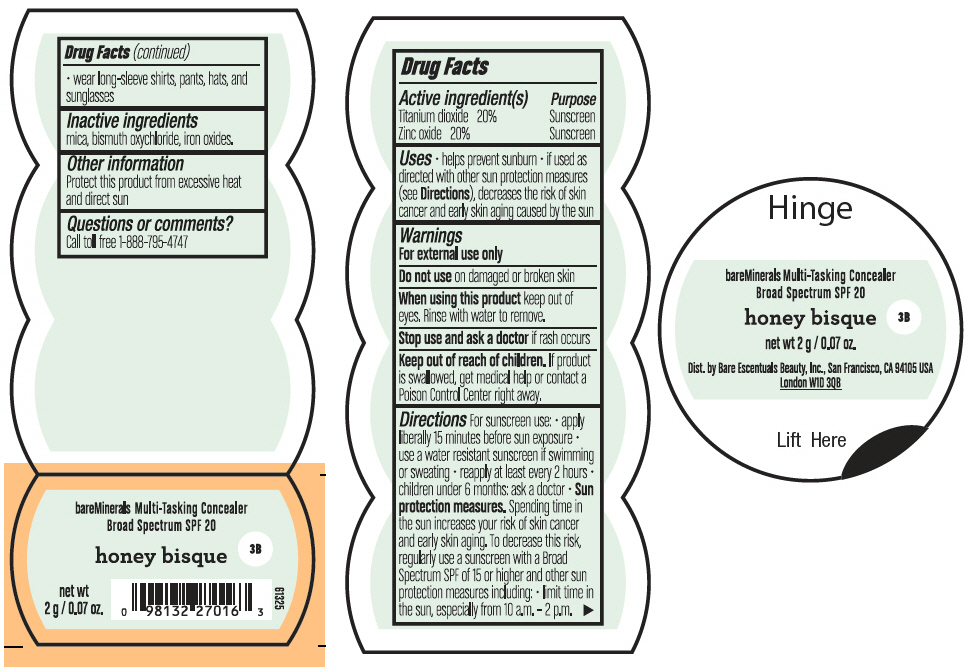
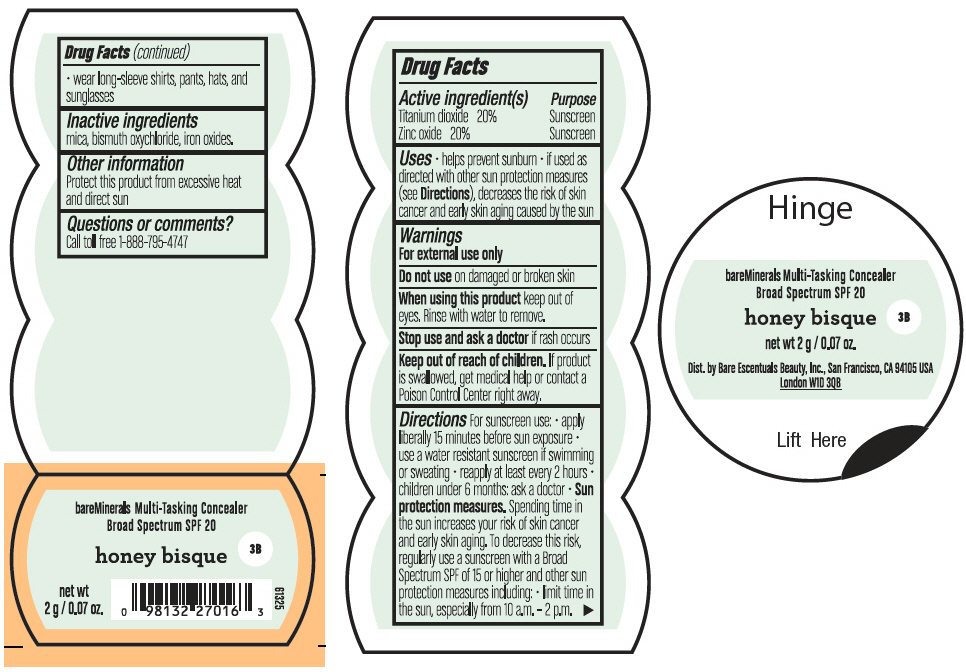
BAREMINERALS MULTI-TASKING SPF 20 CONCEALER HONEY BISQUE- titanium dioxide and zinc oxide powder
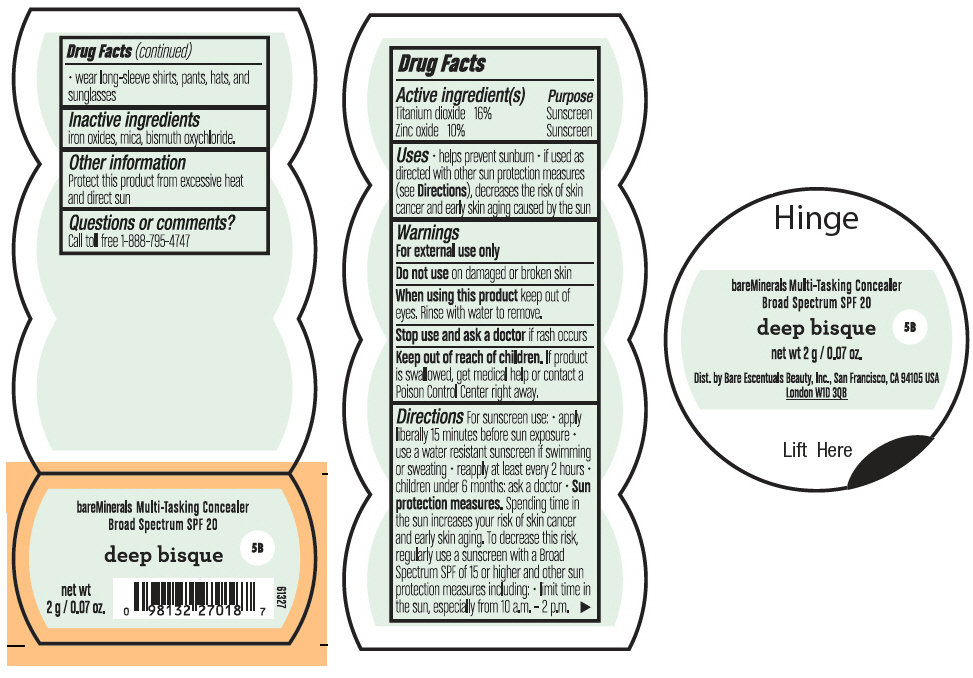
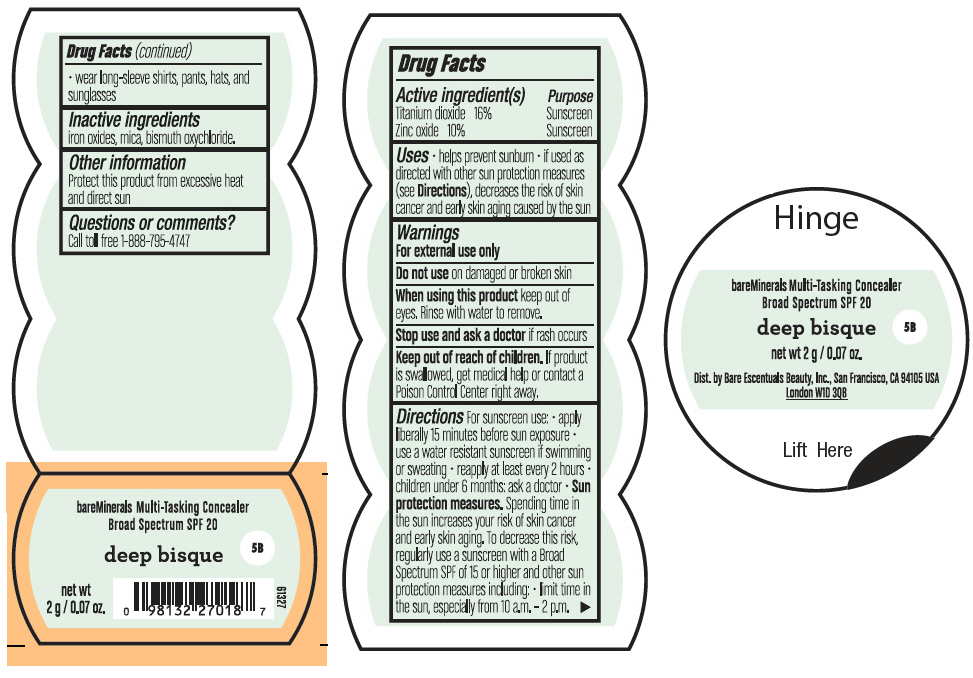
BAREMINERALS MULTI-TASKING SPF 20 CONCEALER DEEP BISQUE- titanium dioxide and zinc oxide powder
-
NDC Code(s):
98132-005-02,
98132-006-02,
98132-007-02,
98132-008-01, view more98132-008-02, 98132-008-03
- Packager: Orveon Global US LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 4, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Purpose
-
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
- Warnings
-
Directions
For sunscreen use:
- Apply liberally 15 minutes before sun exposure
- Use a water-resistant sunscreen if swimming or sweating
- Reapply at least every 2 hours
- Children under 6 months: ask a doctor
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses
- Inactive Ingredients
- Other information
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 2 g Jar Label - Bisque
- PRINCIPAL DISPLAY PANEL - 2 g Jar Label - Summer Bisque
- PRINCIPAL DISPLAY PANEL - 2 g Jar Label - Honey Bisque
- PRINCIPAL DISPLAY PANEL - 2 g Jar Label - Deep Bisque
-
INGREDIENTS AND APPEARANCE
BAREMINERALS MULTI-TASKING SPF 20 CONCEALER BISQUE
titanium dioxide powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-005 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 488 mg in 2 g Inactive Ingredients Ingredient Name Strength BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-005-02 2 g in 1 JAR; Type 0: Not a Combination Product 08/01/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 08/01/2021 BAREMINERALS MULTI-TASKING SPF 20 CONCEALER SUMMER BISQUE
titanium dioxide and zinc oxide powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-006 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 500 mg in 2 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 400 mg in 2 g Inactive Ingredients Ingredient Name Strength BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-006-02 2 g in 1 JAR; Type 0: Not a Combination Product 08/01/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 08/01/2021 BAREMINERALS MULTI-TASKING SPF 20 CONCEALER HONEY BISQUE
titanium dioxide and zinc oxide powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 400 mg in 2 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 400 mg in 2 g Inactive Ingredients Ingredient Name Strength BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-007-02 2 g in 1 JAR; Type 0: Not a Combination Product 08/01/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 08/01/2021 BAREMINERALS MULTI-TASKING SPF 20 CONCEALER DEEP BISQUE
titanium dioxide and zinc oxide powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:98132-008 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 320 mg in 2 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 200 mg in 2 g Inactive Ingredients Ingredient Name Strength BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:98132-008-02 2 g in 1 JAR; Type 0: Not a Combination Product 08/01/2021 2 NDC:98132-008-01 0.75 g in 1 JAR; Type 0: Not a Combination Product 08/01/2021 3 NDC:98132-008-03 8 g in 1 JAR; Type 0: Not a Combination Product 08/01/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 08/01/2021 Labeler - Orveon Global US LLC (118344494)