Label: BRYHALI- halobetasol propionate lotion
- NDC Code(s): 0187-0002-01, 0187-0002-03, 0187-0002-60
- Packager: Bausch Health US, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated June 1, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use BRYHALI safely and effectively. See full prescribing information for BRYHALI.
BRYHALI® (halobetasol propionate) lotion, for topical use
Initial U.S. Approval: 1990INDICATIONS AND USAGE
BRYHALI lotion is a corticosteroid indicated for the topical treatment of plaque psoriasis in adults. (1)
DOSAGE AND ADMINISTRATION
- •
- Apply a thin layer of BRYHALI Lotion to the affected areas once daily. (2)
- •
- Treatment beyond 8 weeks is not recommended. Discontinue treatment if control is achieved before 8 weeks. (2)
- •
- Do not use with occlusive dressings unless directed by a physician. (2)
- •
- Avoid use on the face, groin, or axillae. (2)
- •
- Not for oral, ophthalmic, or intravaginal use. (2)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
- None (4).
WARNINGS AND PRECAUTIONS
- •
- Reversible hypothalamic-pituitary-adrenal (HPA) axis suppression was observed and may occur with the potential for glucocorticosteroid insufficiency during or after treatment. (5.1)
- •
- Systemic effects of topical corticosteroids may also include Cushing’s syndrome, hyperglycemia, and glucosuria. (5.1)
- •
- Systemic absorption may require evaluation for HPA axis suppression. (5.1)
- •
- Use of potent corticosteroids on large areas, for prolonged durations, under occlusive dressings, or on an altered skin barrier may increase systemic exposure. (5.1)
- •
- Children may be more susceptible to systemic toxicity when treated with topical corticosteroids. (5.1, 8.4)
- •
- Local adverse reactions may include atrophy, striae, telangiectasias, hypopigmentation, and allergic contact dermatitis. Some local adverse reactions may be irreversible. (5.2)
- •
- Use of topical corticosteroids may increase the risk of posterior subcapsular cataracts and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist for evaluation. (5.3)
ADVERSE REACTIONS
- •
- The most common adverse reactions (≥1%) were upper respiratory tract infection, application site dermatitis and hyperglycemia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Bausch Health US, LLC at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 6/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression
5.2 Local Adverse Reactions
5.3 Ophthalmic Adverse Reactions
5.4 Concomitant Skin Infections
5.5 Allergic Contact Dermatitis
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
Apply a thin layer of BRYHALI Lotion to affected areas once daily. Rub in gently. Wash hands after each application, unless BRYHALI Lotion is for treatment of the hands.
BRYHALI Lotion treatment beyond 8 weeks is not recommended, and the total dosage should not exceed approximately 50 g per week. Discontinue treatment if control is achieved before 8 weeks. Do not use with occlusive dressings unless directed by a physician.
BRYHALI Lotion should not be used on the face, groin, or in the axillae.
BRYHALI Lotion is not for oral, ophthalmic, or intravaginal use.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression
BRYHALI Lotion has been shown to suppress the hypothalamic-pituitary-adrenal (HPA) axis.
Systemic effects of topical corticosteroids may include reversible HPA axis suppression with the potential for glucocorticosteroid insufficiency. This may occur during treatment or upon withdrawal of treatment with the topical corticosteroid.
The potential for hypothalamic-pituitary-adrenal (HPA) axis suppression with BRYHALI Lotion was evaluated in a study of 19 adult subjects with moderate to severe plaque psoriasis involving ≥20% of their body surface area (BSA). HPA axis suppression was reported for 1 (5.6%) subject at Week 4 and for 3 (15.8%) subjects at Week 8. All 3 subjects had normal HPA axis suppression test with discontinuation of treatment [see Clinical Pharmacology (12.2)].
Because of the potential for systemic absorption, use of topical corticosteroids, including BRYHALI Lotion, may require that patients be evaluated periodically for evidence of HPA axis suppression. Factors that predispose a patient using a topical corticosteroid to HPA axis suppression include the use of more potent corticosteroids, use over large surface areas, occlusive use, use on an altered skin barrier, concomitant use of multiple corticosteroid-containing products, liver failure, and young age. An adrenocorticotropic hormone (ACTH) stimulation test may be helpful in evaluating patients for HPA axis suppression.
If HPA axis suppression is documented, attempt to gradually withdraw the drug, reduce the frequency of application, or substitute a less potent steroid. Manifestations of adrenal insufficiency may require supplemental systemic corticosteroids. Recovery of HPA axis function is generally prompt and complete upon discontinuation of topical corticosteroids.
Systemic effects of topical corticosteroids may also include Cushing’s syndrome, hyperglycemia, and glucosuria. Use of more than one corticosteroid-containing product at the same time may increase the total systemic exposure to corticosteroids. Pediatric patients may be more susceptible than adults to systemic toxicity from the use of topical corticosteroids due to their larger surface-to-body-mass ratios [see Use in Specific Populations (8.4)].
5.2 Local Adverse Reactions
Local adverse reactions from topical corticosteroids may include atrophy, striae, telangiectasias, burning, itching, irritation, dryness, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, and miliaria. These may be more likely with occlusive use, prolonged use, or use of higher potency corticosteroids, including BRYHALI Lotion. Some local adverse reactions may be irreversible.
5.3 Ophthalmic Adverse Reactions
Use of topical corticosteroids may increase the risk of posterior subcapsular cataracts and glaucoma. Cataracts and glaucoma have been reported in postmarketing experience with the use of topical corticosteroid products. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
5.4 Concomitant Skin Infections
Use an appropriate antimicrobial agent if a skin infection is present or develops. If a favorable response does not occur promptly, discontinue use of BRYHALI Lotion until the infection has been adequately treated.
5.5 Allergic Contact Dermatitis
Allergic contact dermatitis with corticosteroids is usually diagnosed by observing failure to heal rather than noting a clinical exacerbation. Consider confirmation of a clinical diagnosis of allergic contact dermatitis by appropriate patch testing. Discontinue BRYHALI Lotion if allergic contact dermatitis occurs.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In randomized, double-blind, multicenter, vehicle-controlled clinical trials, 426 adults with plaque psoriasis were treated with BRYHALI Lotion and had post-baseline safety data. Subjects applied BRYHALI Lotion once daily for up to 8 weeks. Table 1 presents adverse reactions that occurred in at least 1% of subjects treated with BRYHALI Lotion and more frequently than in vehicle-treated subjects.
Table 1: Adverse Reactions Occurring in ≥1% of the Subjects Treated with BRYHALI Lotion through Week 8 BRYHALI Lotion
(N=284)
Vehicle Lotion
(N=142)
Adverse Reaction
%
%
Upper Respiratory Tract Infection
2%
1%
Application Site Dermatitis
1%
0
Hyperglycemia
1%
0
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data on BRYHALI Lotion use in pregnant women to inform a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
In animal reproduction studies, increased malformations, including cleft palate and omphalocele, were observed after oral administration of halobetasol propionate during organogenesis to pregnant rats and rabbits. The available data do not support relevant comparisons of systemic halobetasol propionate exposures achieved in the animal studies to exposures observed in humans after topical use of BRYHALI Lotion.
The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
Halobetasol propionate has been shown to cause malformations in rats and rabbits when given orally during organogenesis at doses of 0.04 to 0.1 mg/kg/day in rats and 0.01 mg/kg/day in rabbits. Halobetasol propionate was embryotoxic in rabbits but not in rats. Cleft palate was observed in both rats and rabbits. Omphalocele was seen in rats but not in rabbits.
8.2 Lactation
Risk Summary
There are no data on the presence of halobetasol propionate or its metabolites in human milk, the effects on the breastfed infant, or the effects on milk production after treatment with BRYHALI Lotion.
Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for BRYHALI Lotion and any potential adverse effects on the breastfed child from BRYHALI Lotion.
Clinical Considerations
Advise breastfeeding women not to apply BRYHALI Lotion directly to the nipple and areola to avoid direct infant exposure.
8.4 Pediatric Use
Safety and effectiveness of BRYHALI Lotion in pediatric patients under the age of 18 years have not been evaluated.
Because of higher skin-surface-area-to-body-mass ratios, pediatric patients are at a greater risk than adults of HPA axis suppression and Cushing’s syndrome when they are treated with topical corticosteroids. They are therefore also at greater risk of adrenal insufficiency during or after withdrawal of treatment. Adverse reactions including striae have been reported with use of topical corticosteroids in infants and children [see Warnings and Precautions (5.1)].
HPA axis suppression, Cushing’s syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include low plasma cortisol levels and an absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema [see Warnings and Precautions (5.1)].
-
11 DESCRIPTION
BRYHALI (halobetasol propionate) lotion contains a corticosteroid, halobetasol propionate, as the active ingredient in a white to off-white lotion formulation intended for topical use.
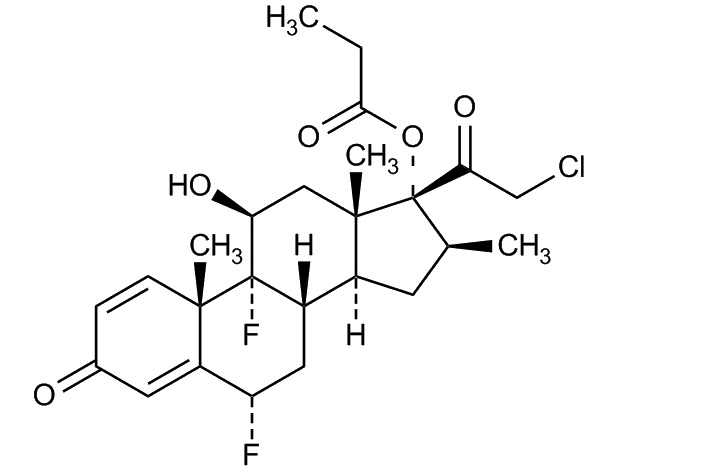
Halobetasol propionate is a synthetic corticosteroid. The chemical name for halobetasol propionate is 21-chloro-6α, 9-difluoro-11β, 17-dihydroxy-16β-methylpregna-1, 4-diene-3, 20 –dione, 17-propionate. Halobetasol propionate is a white to off-white crystalline powder with a molecular weight of 484.96 and a molecular formula of C25H31ClF2O5. It is practically insoluble in water and freely soluble in dichloromethane and in acetone. The structural formula for halobetasol propionate is represented below:
Each gram of BRYHALI Lotion contains 0.1 mg (0.01%) halobetasol propionate in a white to off-white lotion base consisting of carbomer copolymer type B, carbomer homopolymer type A, diethyl sebacate, edetate disodium dihydrate, light mineral oil, methylparaben, propylparaben, purified water, sodium hydroxide, sorbitan monooleate and sorbitol solution, 70%.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Corticosteroids play a role in cellular signaling, immune function, inflammation, and protein regulation; however, the precise mechanism of action in plaque psoriasis is unknown.
12.2 Pharmacodynamics
Vasoconstrictor Assay
A vasoconstrictor assay in healthy subjects with BRYHALI Lotion indicated that the formulation is in the potent to superpotent range of potency as compared to other topical corticosteroids. However, similar blanching scores do not necessarily imply therapeutic equivalence.
Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression
The potential for HPA axis suppression was evaluated in 19 adult subjects with moderate to severe plaque psoriasis with at least 20% BSA involved. An approximate dose of 7 g BRYHALI Lotion was applied once daily for 8 weeks. An abnormal HPA axis suppression test, as indicated by a 30-minute post-stimulation cortisol level ≤18 mcg/dL, was reported for 1 (5.6%) subject at Week 4 and for 3 (15.8%) subjects at Week 8. The subject suppressed at Week 4 was also suppressed at Week 8. These effects were reversible as recovery of HPA axis function was generally prompt with the discontinuation of treatment. [See Warnings and Precautions (5.1).]
12.3 Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors, including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings. Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin may increase percutaneous absorption.
In an open-label, randomized, pharmacokinetic study, 23 subjects aged 18 years and older with moderate to severe plaque psoriasis applied approximately 7 grams of BRYHALI Lotion to a mean BSA of 27.7±11.3% once daily for 28 days. Systemic concentrations were at steady state by Day 14. Only 5 out of 20 subjects had one or more quantifiable systemic concentrations of halobetasol propionate on Day 14. The mean ± SD for maximum systemic concentration (Cmax) on Day 14 was 31.2±62.2 pg/mL. The mean area under the concentration versus time curve (AUC) could not be reliably estimated due to insufficient number of quantifiable timepoints.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed to evaluate the carcinogenic potential of halobetasol propionate.
Halobetasol propionate was not genotoxic in the Ames assay, in the sister chromatid exchange test in Chinese hamster somatic cells, in chromosome aberration studies of germinal and somatic cells of rodents, or in a mammalian spot test. Positive mutagenicity effects were observed in a mouse lymphoma gene mutation assay in vitro and in a Chinese hamster micronucleus test.
Studies in rats following oral administration of halobetasol propionate at dose levels up to 0.05 mg/kg/day indicated no impairment of fertility or general reproductive performance.
-
14 CLINICAL STUDIES
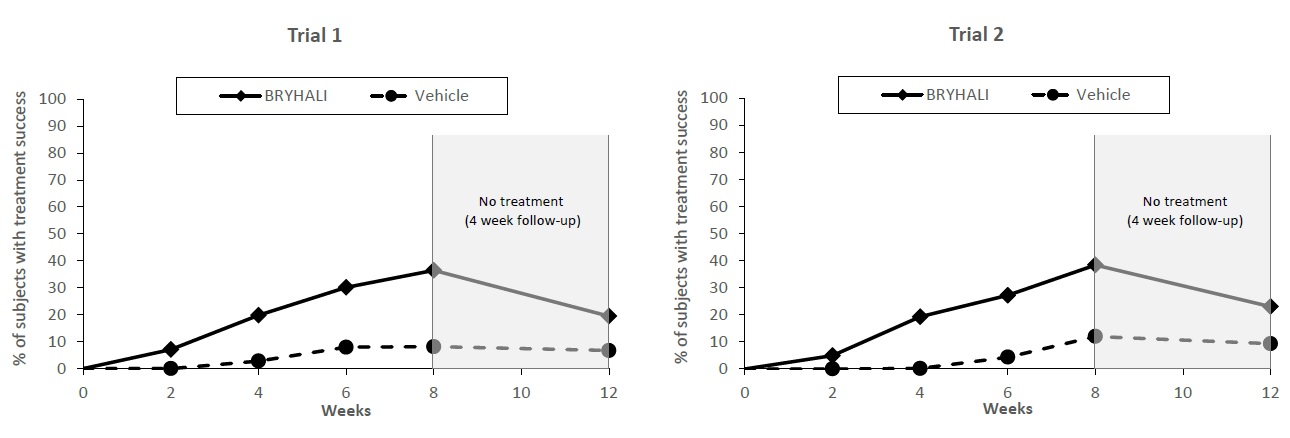
BRYHALI Lotion was evaluated for the treatment of moderate to severe plaque psoriasis in two prospective, multicenter, randomized, double-blind clinical trials (Trial 1 [NCT02514577] and Trial 2 [NCT02515097]). These trials were conducted in 430 subjects 18 years of age and older with moderate to severe plaque psoriasis that covered a body surface area (BSA) between 3% and 12% excluding the face, scalp, palms, soles, axillae, and intertriginous areas. Disease severity was determined by a 5-grade Investigator’s Global Assessment (IGA). Subjects applied BRYHALI Lotion or vehicle to all affected areas once daily for up to 8 weeks. Subjects had a follow-up visit 4 weeks after the end of treatment (Week 12) where safety and efficacy were evaluated.
The primary efficacy endpoint was the proportion of subjects with treatment success at Week 8, where treatment success was defined as at least a 2-grade improvement from baseline in IGA score and an IGA score equating to “clear” or “almost clear”. Table 2 lists the primary efficacy results for Trials 1 and 2. The secondary efficacy endpoints evaluated treatment success sequentially at Weeks 12, 6, 4, and 2. Figure 1 shows the primary and secondary efficacy results over time.
Table 2: Primary Efficacy Results at Week 8 in Subjects with Moderate to Severe Plaque Psoriasis Trial 1 Trial 2 BRYHALI Vehicle BRYHALI Vehicle N=143 N=74 N=142 N=71 - *
- Treatment success was defined as at least a 2-grade improvement from baseline in IGA score and an IGA score equating to “clear” or “almost clear”. Clear = no evidence of scaling, no evidence of erythema, no evidence of plaque elevation above normal skin level. Almost clear = some plaques with fine scales, faint pink/light red erythema on most plaques, slight or barely perceptible elevation of plaques above normal skin level.
IGA Treatment Success at Week 8*
37%
8%
38%
12%
*The treatment difference at Week 2 in Trial 2 was not statistically significant.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
BRYHALI (halobetasol propionate) lotion, 0.01% is a white to off-white lotion supplied in a white aluminum tube as follows:
- •
- 60 g (NDC 0187-0002-60)
- •
- 100 g (NDC 0187-0002-01)
Storage and Handling Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Protect from freezing.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
This information is intended to aid in the safe and effective use of this medication. It is not a disclosure of all administration instructions or all possible adverse or unintended effects.
Advise patients using BRYHALI Lotion of the following information and instructions:
Important Administration Instructions
Instruct patients to discontinue BRYHALI Lotion when psoriasis is controlled. Inform patients that BRYHALI Lotion is to be used as directed by the physician and should not be used for longer than the prescribed time period. Total dosage should not exceed 50 grams per week [see Dosage and Administration (2)].
Instruct patients to avoid bandaging, wrapping or otherwise occluding the treatment area(s), unless directed by physician. Advise patients to avoid use on the face, groin, or axillae [see Dosage and Administration (2)].
Inform patients that BRYHALI Lotion is for external use only. Advise patients that BRYHALI Lotion is not for oral, ophthalmic, or intravaginal use [see Dosage and Administration (2)].
Breastfeeding women should not apply BRYHALI Lotion directly to the nipple and areola to avoid directly exposing the infant [see Use in Specific Populations (8.2)].
Effects on Endocrine System
BRYHALI Lotion may cause HPA axis suppression. Advise patients that use of topical corticosteroids, including BRYHALI Lotion, may require periodic evaluation for HPA axis suppression. Topical corticosteroids may have other endocrine effects. Concomitant use of multiple corticosteroid-containing products may increase the total systemic exposure to topical corticosteroids [see Warnings and Precautions (5.1)].
Local Adverse Reactions
Inform patients that BRYHALI Lotion may cause local adverse reactions. These reactions may be more likely to occur with occlusive use or prolonged use of BRYHALI Lotion [see Warnings and Precautions (5.2)].
Distributed by:
Bausch Health US, LLC
Bridgewater, NJ 08807 USAManufactured by:
Bausch Health Companies Inc.
Laval, Quebec H7L 4A8, CanadaU.S. Patent Numbers: 8,809,307 and 10,478,502
BRYHALI is a trademark of Bausch Health Companies Inc. or its affiliates.
© 2020 Bausch Health Companies Inc. or its affiliates
9652104TEAR HERE (Patient Information)
-
Patient Package Insert
PATIENT INFORMATION
BRYHALI® (bry-HAL-ee)
(halobetasol propionate) Lotion
Important: BRYHALI is for use on the skin only. Do not apply BRYHALI in your mouth, eyes, or vagina.
What is BRYHALI?
BRYHALI is a prescription corticosteroid medicine used on the skin (topical) to treat adults with plaque psoriasis.
It is not known if BRYHALI is safe and effective in children under 18 years of age.
Before using BRYHALI, tell your doctor about all of your medical conditions, including if you:
- •
- have had irritation or other skin reaction to a steroid medicine in the past.
- •
- have a skin infection. You may need medicine to treat the skin infection before using BRYHALI.
- •
- have diabetes.
- •
- have adrenal gland problems.
- •
- have liver problems.
- •
- are pregnant or plan to become pregnant. It is not known if BRYHALI will harm your unborn baby.
- •
- are breastfeeding or plan to breastfeed. It is not known if BRYHALI passes into your breast milk. If you use BRYHALI and breastfeed, do not apply BRYHALI to your nipple or areola to avoid getting BRYHALI into your baby’s mouth.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your doctor if you take other corticosteroids medicines by mouth, or injection, or use other products on your skin that contain corticosteroids.
How should I use BRYHALI?
- •
- Use BRYHALI exactly as your doctor tells you to use it.
- •
- Apply a thin layer of BRYHALI to the affected areas 1 time each day and rub in gently.
- •
- You should not use more than 50 g of BRYHALI in 1 week.
- •
- Do not bandage, wrap, or cover the treated skin area(s) unless your doctor tells you to.
- •
- Avoid using BRYHALI on your face, groin, or underarms (armpits).
- •
- Talk to your doctor if your skin does not improve after 8 weeks of treatment with BRYHALI.
- •
- You should not use BRYHALI longer than 8 weeks unless your doctor tells you to.
- •
- Wash your hands after using BRYHALI unless you are using the medicine to treat your hands.
What are the possible side effects of BRYHALI?
BRYHALI may cause serious side effects, including:
- •
- BRYHALI can pass through your skin. Too much BRYHALI passing through your skin can cause adrenal glands to stop working.
- •
- Cushing’s syndrome, a condition that happens when your body is exposed to too much of the hormone cortisol.
- •
- High blood sugar (hyperglycemia).
- •
- Skin reactions at the treated skin site. Tell your doctor if you get any skin reactions or skin infections.
- •
- Vision problems. BRYHALI may increase your chance of developing cataract(s) and glaucoma. Tell your healthcare provider if you develop blurred vision or other vision problems during treatment with BRYHALI.
- •
- Effects on growth and weight in children.
The most common side effects of BRYHALI include burning, stinging, itching, dryness (application site dermatitis), upper respiratory tract infection and high blood sugar (hyperglycemia).
These are not all the possible side effects of BRYHALI.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store BRYHALI?
- •
- Store BRYHALI at room temperature between 68° to 77°F (20° to 25°C).
- •
- Protect from freezing.
- Keep BRYHALI and all medicines out of the reach of children.
General information about the safe and effective use of BRYHALI.
- Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use BRYHALI for a condition for which it was not prescribed. Do not give BRYHALI to other people, even if they have the same condition you have. It may harm them. You can ask your pharmacist or doctor for information about BRYHALI that is written for health professionals.
What are the ingredients in BRYHALI?
Active ingredients: halobetasol propionate
Inactive ingredients: carbomer copolymer type B, carbomer homopolymer type A, diethyl sebacate, edetate disodium dihydrate, light mineral oil, methylparaben, propylparaben, purified water, sodium hydroxide, sorbitan monooleate and sorbitol solution, 70%.
Distributed by: Bausch Health US, LLC, Bridgewater, NJ 08807 USA
Manufactured by: Bausch Health Companies Inc., Laval, Quebec H7L 4A8, CanadaU.S. Patent Numbers: 8,809,307 and 10,478,502
BRYHALI is a trademark of Bausch Health Companies Inc. or its affiliates.
© 2020 Bausch Health Companies Inc. or its affiliates
- For more information, go to www.bauschhealth.com or call 1-800-321-4576.
- This Patient Information has been approved by the U.S. Food and Drug Administration.
- 9652104
- Revised: 06/2020
- PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BRYHALI
halobetasol propionate lotionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0187-0002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HALOBETASOL PROPIONATE (UNII: 91A0K1TY3Z) (HALOBETASOL - UNII:9P6159HM7T) HALOBETASOL PROPIONATE 0.1 mg in 1 g Inactive Ingredients Ingredient Name Strength CARBOMER COPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 809Y72KV36) CARBOMER HOMOPOLYMER TYPE A (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: F68VH75CJC) DIETHYL SEBACATE (UNII: I41B9FJK6V) EDETATE DISODIUM (UNII: 7FLD91C86K) LIGHT MINERAL OIL (UNII: N6K5787QVP) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) SORBITAN MONOOLEATE (UNII: 06XEA2VD56) SORBITOL (UNII: 506T60A25R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0187-0002-03 1 in 1 CARTON 11/06/2018 1 3 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:0187-0002-60 1 in 1 CARTON 11/06/2018 2 60 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:0187-0002-01 1 in 1 CARTON 11/06/2018 3 100 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209355 11/06/2018 Labeler - Bausch Health US, LLC (831922468) Establishment Name Address ID/FEI Business Operations Bausch Health Companies Inc. 245141858 MANUFACTURE(0187-0002) , PACK(0187-0002) , LABEL(0187-0002)