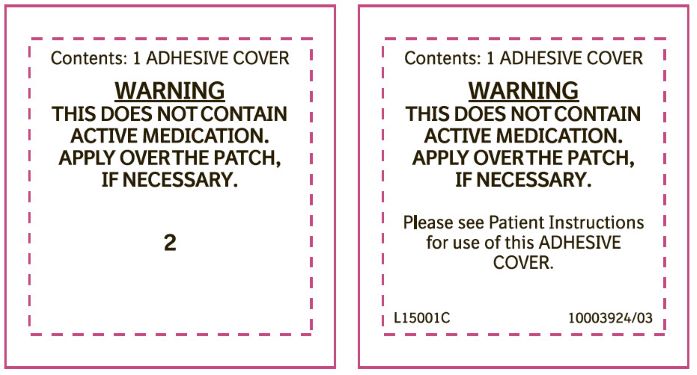
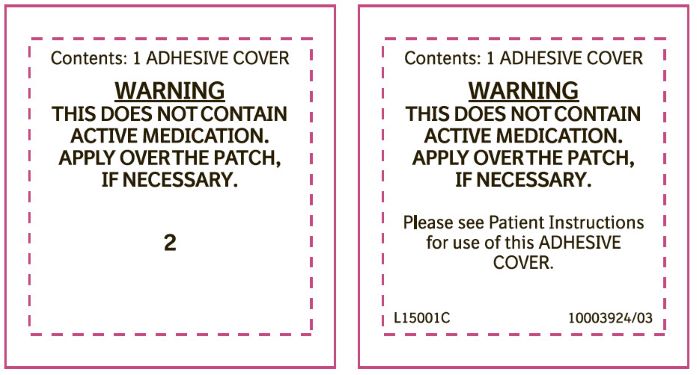
Label: CATAPRES-TTS-1- clonidine transdermal system patch
CATAPRES-TTS-2- clonidine transdermal system patch
CATAPRES-TTS-3- clonidine transdermal system patch
- NDC Code(s): 82089-101-34, 82089-102-34, 82089-103-34
- Packager: Technomed Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated October 11, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
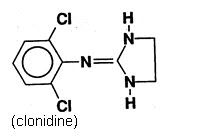
Catapres‑TTS® (clonidine transdermal system) is a transdermal system providing continuous systemic delivery of clonidine for 7 days at an approximately constant rate. Clonidine is a centrally acting alpha‑agonist hypotensive agent. It is an imidazoline derivative with the chemical name 2, 6‑dichloro‑N‑2‑imidazolidinylidenebenzenamine and has the following chemical structure:
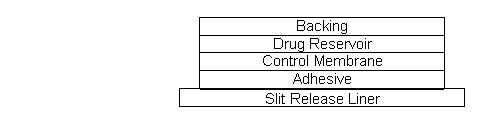
System Structure and Components
Catapres‑TTS® is a multi-layered film, 0.2 mm thick, containing clonidine as the active agent. The system areas are 3.5 cm2 (Catapres‑TTS®‑1), 7.0 cm2 (Catapres‑TTS®‑2) and 10.5 cm2 (Catapres‑TTS®‑3) and the amount of drug released is directly proportional to the area (see Release Rate Concept). The composition per unit area is the same for all three doses.
Proceeding from the visible surface towards the surface attached to the skin, there are four consecutive layers: 1) a backing layer of pigmented polyester and aluminum film; 2) a drug reservoir of clonidine, mineral oil, polyisobutylene, and colloidal silicon dioxide; 3) a microporous polypropylene membrane that controls the rate of delivery of clonidine from the system to the skin surface; 4) an adhesive formulation of clonidine, mineral oil, polyisobutylene, and colloidal silicon dioxide. Prior to use, a protective slit release liner of polyester that covers the adhesive layer is removed.
Cross Section of the System:
Release Rate Concept
Catapres‑TTS® (clonidine transdermal system) is programmed to release clonidine at an approximately constant rate for 7 days. The energy for drug release is derived from the concentration gradient existing between a saturated solution of drug in the system and the much lower concentration prevailing in the skin. Clonidine flows in the direction of the lower concentration at a constant rate, limited by the rate‑controlling membrane, so long as a saturated solution is maintained in the drug reservoir.
Following system application to intact skin, clonidine in the adhesive layer saturates the skin site below the system. Clonidine from the drug reservoir then begins to flow through the rate‑controlling membrane and the adhesive layer of the system into the systemic circulation via the capillaries beneath the skin. Therapeutic plasma clonidine levels are achieved 2 to 3 days after initial application of Catapres‑TTS®.
The 3.5, 7.0, and 10.5 cm2 systems deliver 0.1, 0.2, and 0.3 mg of clonidine per day, respectively. To ensure constant release of drug for 7 days, the total drug content of the system is higher than the total amount of drug delivered. Application of a new system to a fresh skin site at weekly intervals continuously maintains therapeutic plasma concentrations of clonidine. If the Catapres‑TTS® is removed and not replaced with a new system, therapeutic plasma clonidine levels will persist for about 8 hours and then decline slowly over several days. Over this time period, blood pressure returns gradually to pretreatment levels.
-
CLINICAL PHARMACOLOGY
Clonidine stimulates alpha‑adrenoreceptors in the brain stem. This action results in reduced sympathetic outflow from the central nervous system and in decreases in peripheral resistance, renal vascular resistance, heart rate, and blood pressure. Renal blood flow and glomerular filtration rate remain essentially unchanged. Normal postural reflexes are intact; therefore, orthostatic symptoms are mild and infrequent.
Acute studies with clonidine hydrochloride in humans have demonstrated a moderate reduction (15% to 20%) of cardiac output in the supine position with no change in the peripheral resistance; at a 45° tilt there is a smaller reduction in cardiac output and a decrease of peripheral resistance.
During long‑term therapy, cardiac output tends to return to control values, while peripheral resistance remains decreased. Slowing of the pulse rate has been observed in most patients given clonidine, but the drug does not alter normal hemodynamic responses to exercise.
Tolerance to the antihypertensive effect may develop in some patients, necessitating a reevaluation of therapy.
Other studies in patients have provided evidence of a reduction in plasma renin activity and in the excretion of aldosterone and catecholamines. The exact relationship of these pharmacologic actions to the antihypertensive effect of clonidine has not been fully elucidated.
Clonidine acutely stimulates the release of growth hormone in children as well as adults but does not produce a chronic elevation of growth hormone with long‑term use.
Pharmacokinetics
Catapres‑TTS® delivers clonidine at an approximately constant rate for 7 days. The absolute bioavailability of clonidine from the Catapres‑TTS® dosage form is approximately 60%. Steady-state clonidine plasma levels are obtained within 3 days after transdermal application to the upper outer arm and increase linearly with increasing size of the transdermal patch. Mean steady-state plasma concentrations with the 3.5 cm2, 7.0 cm2 and 10.5 cm2 systems are approximately 0.4 ng/mL, 0.8 ng/mL, and 1.1 ng/mL, respectively. Similar clonidine steady-state concentrations are reached after application to the chest. Steady-state clonidine plasma levels remain constant after removal of one system and application of a new system of the same size.
Following intravenous administration clonidine displays biphasic disposition with a distribution half-life of about 20 minutes and an elimination half-life ranging from 12 to 16 hours. The half-life increases up to 41 hours in patients with severe impairment of renal function. Clonidine has a total clearance of 177 mL/min and a renal clearance of 102 mL/min. The apparent volume of distribution (Vz) of clonidine is 197 L (2.9 L/kg). Clonidine crosses the placental barrier. It has been shown to cross the blood brain barrier in rats.
Following oral administration, about 40% to 60% of the absorbed dose is recovered in the urine as unchanged drug within 24 hours. About 50% of the absorbed dose is metabolized in the liver.
After removal of the Catapres‑TTS®, clonidine plasma concentrations decline slowly with a half-life of approximately 20 hours.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
Withdrawal
Patients should be instructed not to discontinue therapy without consulting their physician. Sudden cessation of clonidine treatment has, in some cases, resulted in symptoms such as nervousness, agitation, headache, tremor, and confusion accompanied or followed by a rapid rise in blood pressure and elevated catecholamine concentrations in the plasma. The likelihood of such reactions to discontinuation of clonidine therapy appears to be greater after administration of higher doses or continuation of concomitant beta‑blocker treatment and special caution is therefore advised in these situations. Rare instances of hypertensive encephalopathy, cerebrovascular accidents and death have been reported after clonidine withdrawal. When discontinuing therapy with Catapres, the physician should reduce the dose gradually over 2 to 4 days to avoid withdrawal symptomatology.
An excessive rise in blood pressure following discontinuation of Catapres‑TTS® therapy can be reversed by administration of oral clonidine hydrochloride or by intravenous phentolamine. If therapy is to be discontinued in patients receiving a beta‑blocker and clonidine concurrently, the beta‑blocker should be withdrawn several days before the gradual discontinuation of Catapres‑TTS®.
-
PRECAUTIONS
General
In patients who have developed localized contact sensitization to Catapres‑TTS®, continuation of Catapres‑TTS® or substitution of oral clonidine hydrochloride therapy may be associated with development of a generalized skin rash.
In patients who develop an allergic reaction to Catapres‑TTS®, substitution of oral clonidine hydrochloride may also elicit an allergic reaction (including generalized rash, urticaria, or angioedema).
The sympatholytic action of clonidine may worsen sinus node dysfunction and atrioventricular (AV) block, especially in patients taking other sympatholytic drugs. There are post-marketing reports of patients with conduction abnormalities and/or taking other sympatholytic drugs who developed severe bradycardia requiring IV atropine, IV isoproterenol and temporary cardiac pacing while taking clonidine.
In hypertension caused by pheochromocytoma, no therapeutic effect of Catapres‑TTS® can be expected.
In rare instances, loss of blood pressure control has been reported in patients using Catapres‑TTS® according to the instructions for use.
Perioperative Use
Catapres‑TTS® should not be interrupted during the surgical period. Blood pressure should be carefully monitored during surgery and additional measures to control blood pressure should be available if required. Physicians considering starting Catapres‑TTS® therapy during the perioperative period must be aware that therapeutic plasma clonidine levels are not achieved until 2 to 3 days after initial application of Catapres-TTS® (see DOSAGE AND ADMINISTRATION).
Defibrillation or Cardioversion
The clonidine transdermal systems should be removed before attempting defibrillation or cardioversion because of the potential for altered electrical conductivity which may increase the risk of arcing, a phenomenon associated with the use of defibrillators.
MRI
Skin burns have been reported at the patch site in several patients wearing an aluminized transdermal system during a magnetic resonance imaging scan (MRI). Because the Catapres-TTS® contains aluminum, it is recommended to remove the system before undergoing an MRI.
Information for Patients
Patients should be cautioned against interruption of Catapres‑TTS® (clonidine transdermal system) therapy without their physician’s advice.
Since patients may experience a possible sedative effect, dizziness, or accommodation disorder with use of clonidine, caution patients about engaging in activities such as driving a vehicle or operating appliances or machinery. Also, inform patients that this sedative effect may be increased by concomitant use of alcohol, barbiturates, or other sedating drugs.
Patients who wear contact lenses should be cautioned that treatment with Catapres-TTS® may cause dryness of eyes.
Patients should be instructed to consult their physicians promptly about the possible need to remove the patch if they observe moderate to severe localized erythema and/or vesicle formation at the site of application or generalized skin rash.
If a patient experiences isolated, mild localized skin irritation before completing 7 days of use, the system may be removed and replaced with a new system applied to a fresh skin site.
If the system should begin to loosen from the skin after application, the patient should be instructed to place the adhesive cover directly over the system to ensure adhesion during its 7‑day use.
Used Catapres‑TTS® patches contain a substantial amount of their initial drug content which may be harmful to infants and children if accidentally applied or ingested. THEREFORE, PATIENTS SHOULD BE CAUTIONED TO KEEP BOTH USED AND UNUSED CATAPRES‑TTS® PATCHES OUT OF THE REACH OF CHILDREN. After use, CATAPRES‑TTS should be folded in half with the adhesive sides together and discarded away from children’s reach.
Instructions for use, storage and disposal of the system are provided at the end of this monograph. These instructions are also included in each box of Catapres‑TTS®.
Drug Interactions
Clonidine may potentiate the CNS-depressive effects of alcohol, barbiturates or other sedating drugs. If a patient receiving clonidine is also taking tricyclic antidepressants, the hypotensive effect of clonidine may be reduced, necessitating an increase in the clonidine dose. If a patient receiving clonidine is also taking neuroleptics, orthostatic regulation disturbances (e.g., orthostatic hypotension, dizziness, fatigue) may be induced or exacerbated.
Monitor heart rate in patients receiving clonidine concomitantly with agents known to affect sinus node function or AV nodal conduction e.g., digitalis, calcium channel blockers, and beta‑blockers. Sinus bradycardia resulting in hospitalization and pacemaker insertion has been reported in association with the use of clonidine concomitantly with diltiazem or verapamil.
Amitriptyline in combination with clonidine enhances the manifestation of corneal lesions in rats (see Toxicology).
Toxicology
In several studies with oral clonidine hydrochloride, a dose-dependent increase in the incidence and severity of spontaneous retinal degeneration was seen in albino rats treated for six months or longer. Tissue distribution studies in dogs and monkeys showed a concentration of clonidine in the choroid.
In view of the retinal degeneration seen in rats, eye examinations were performed during clinical trials in 908 patients before, and periodically after, the start of clonidine therapy. In 353 of these 908 patients, the eye examinations were carried out over periods of 24 months or longer. Except for some dryness of the eyes, no drug‑related abnormal ophthalmological findings were recorded and, according to specialized tests such as electroretinography and macular dazzle, retinal function was unchanged.
In combination with amitriptyline, clonidine hydrochloride administration led to the development of corneal lesions in rats within 5 days.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Chronic dietary administration of clonidine was not carcinogenic to rats (132 weeks) or mice (78 weeks) dosed, respectively, at up to 46 to 70 times the maximum recommended daily human dose as mg/kg (9 or 6 times the MRDHD on a mg/m2 basis). There was no evidence of genotoxicity in the Ames test for mutagenicity or mouse micronucleus test for clastogenicity.
Fertility of male and female rats was unaffected by clonidine doses as high as 150 µg/kg (approximately 3 times the MRDHD). In a separate experiment, fertility of female rats appeared to be affected at dose levels of 500 to 2000 µg/kg (10 to 40 times the oral MRDHD on a mg/kg basis; 2 to 8 times the MRDHD on a mg/m2 basis).
Pregnancy
Reproduction studies performed in rabbits at doses up to approximately 3 times the oral maximum recommended daily human dose (MRDHD) of Catapres® (clonidine hydrochloride) produced no evidence of a teratogenic or embryotoxic potential in rabbits. In rats, however, doses as low as 1/3 the oral MRDHD (1/15 the MRDHD on a mg/m2 basis) of clonidine were associated with increased resorptions in a study in which dams were treated continuously from 2 months prior to mating. Increased resorptions were not associated with treatment at the same or at higher dose levels (up to 3 times the oral MRDHD) when the dams were treated on gestation days 6 to 15. Increases in resorption were observed at much higher dose levels (40 times the oral MRDHD on a mg/kg basis; 4 to 8 times the MRDHD on a mg/m2 basis) in mice and rats treated on gestation days 1 to 14 (lowest dose employed in the study was 500 µg/kg).
No adequate well‑controlled studies have been conducted in pregnant women. Clonidine crosses the placental barrier (see CLINICAL PHARMACOLOGY, Pharmacokinetics). Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
-
ADVERSE REACTIONS
Clinical trial experience with Catapres-TTS®
Most systemic adverse effects during Catapres-TTS® therapy have been mild and have tended to diminish with continued therapy. In a 3‑month multi-clinic trial of Catapres-TTS® in 101 hypertensive patients, the systemic adverse reactions were, dry mouth (25 patients) and drowsiness (12), fatigue (6), headache (5), lethargy and sedation (3 each), insomnia, dizziness, impotence/sexual dysfunction, dry throat (2 each) and constipation, nausea, change in taste and nervousness (1 each).
In the above mentioned 3‑month controlled clinical trial, as well as other uncontrolled clinical trials, the most frequent adverse reactions were dermatological and are described below.
In the 3‑month trial, 51 of the 101 patients had localized skin reactions such as erythema (26 patients) and/or pruritus, particularly after using an adhesive cover throughout the 7‑day dosage interval. Allergic contact sensitization to Catapres‑TTS® was observed in 5 patients. Other skin reactions were localized vesiculation (7 patients), hyperpigmentation (5), edema (3), excoriation (3), burning (3), papules (1), throbbing (1), blanching (1), and a generalized macular rash (1).
In additional clinical experience, contact dermatitis resulting in treatment discontinuation was observed in 128 of 673 patients (about 19 in 100) after a mean duration of treatment of 37 weeks. The incidence of contact dermatitis was about 34 in 100 among white women, about 18 in 100 in white men, about 14 in 100 in black women, and approximately 8 in 100 in black men. Analysis of skin reaction data showed that the risk of having to discontinue Catapres‑TTS® treatment because of contact dermatitis was greatest between treatment weeks 6 and 26, although sensitivity may develop either earlier or later in treatment.
In a large‑scale clinical acceptability and safety study by 451 physicians in a total of 3539 patients, other allergic reactions were recorded for which a causal relationship to Catapres‑TTS® was not established: maculopapular rash (10 cases); urticaria (2 cases); and angioedema of the face (2 cases), which also affected the tongue in one of the patients.
Marketing Experience with Catapres-TTS®
The following adverse reactions have been identified during post-approval use of Catapres-TTS®. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate reliably their frequency or establish a causal relationship to drug exposure. Decisions to include these reactions in labeling are typically based on one or more of the following factors: (1) seriousness of the reaction, (2) frequency of reporting, or (3) strength of causal connection to Catapres-TTS®.
Body as a Whole: Fever; malaise; weakness; pallor; and withdrawal syndrome.
Cardiovascular: Congestive heart failure; cerebrovascular accident; electrocardiographic abnormalities (i.e., bradycardia, sick sinus syndrome disturbances and arrhythmias); chest pain; orthostatic symptoms; syncope; increases in blood pressure; sinus bradycardia and AV block with and without the use of concomitant digitalis; Raynaud’s phenomenon; tachycardia; bradycardia; and palpitations.
Central and Peripheral Nervous System/Psychiatric: Delirium; mental depression; hallucinations (including visual and auditory); localized numbness; vivid dreams or nightmares; restlessness; anxiety; agitation; irritability; other behavioral changes; and drowsiness.
Dermatological: Angioneurotic edema; localized or generalized rash; hives; urticaria; contact dermatitis; pruritus; alopecia; and localized hypo or hyper pigmentation.
Gastrointestinal: Anorexia and vomiting.
Genitourinary: Difficult micturition; loss of libido; and decreased sexual activity.
Metabolic: Gynecomastia or breast enlargement and weight gain.
Musculoskeletal: Muscle or joint pain; and leg cramps.
Ophthalmological: Blurred vision; burning of the eyes and dryness of the eyes.
Adverse Events Associated with Oral CATAPRES® Therapy: Most adverse effects are mild and tend to diminish with continued therapy. The most frequent (which appear to be dose-related) are dry mouth, occurring in about 40 of 100 patients; drowsiness, about 33 in 100; dizziness, about 16 in 100; constipation and sedation, each about 10 in 100. The following less frequent adverse experiences have also been reported in patients receiving CATAPRES® (clonidine hydrochloride, USP) tablets, but in many cases patients were receiving concomitant medication and a causal relationship has not been established.
Body as a Whole: Fatigue, fever, headache, pallor, weakness, and withdrawal syndrome. Also reported were a weakly positive Coombs’ test and increased sensitivity to alcohol.
Cardiovascular: Bradycardia, congestive heart failure, electrocardiographic abnormalities (i.e., sinus node arrest, junctional bradycardia, high degree AV block and arrhythmias), orthostatic symptoms, palpitations, Raynaud’s phenomenon, syncope, and tachycardia. Cases of sinus bradycardia and AV block have been reported, both with and without the use of concomitant digitalis.
Central Nervous System: Agitation, anxiety, delirium, delusional perception, hallucinations (including visual and auditory), insomnia, mental depression, nervousness, other behavioral changes, paresthesia, restlessness, sleep disorder, and vivid dreams or nightmares.
Dermatological: Alopecia, angioneurotic edema, hives, pruritus, rash, and urticaria.
Gastrointestinal: Abdominal pain, anorexia, constipation, hepatitis, malaise, mild transient abnormalities in liver function tests, nausea, parotitis, pseudo-obstruction (including colonic pseudo-obstruction), salivary gland pain, and vomiting.
Genitourinary: Decreased sexual activity, difficulty in micturition, erectile dysfunction, loss of libido, nocturia, and urinary retention.
Hematologic: Thrombocytopenia.
Metabolic: Gynecomastia, transient elevation of blood glucose or serum creatine phosphokinase, and weight gain.
Musculoskeletal: Leg cramps and muscle or joint pain.
Oro-otolaryngeal: Dryness of the nasal mucosa.
Ophthalmological: Accommodation disorder, blurred vision, burning of the eyes, decreased lacrimation, and dryness of the eyes.
-
OVERDOSAGE
Hypertension may develop early and may be followed by hypotension, bradycardia, respiratory depression, hypothermia, drowsiness, decreased or absent reflexes, weakness, irritability and miosis. The frequency of CNS depression may be higher in children than adults. Large overdoses may result in reversible cardiac conduction defects or dysrhythmias, apnea, coma and seizures. Signs and symptoms of overdose generally occur within 30 minutes to two hours after exposure. As little as 0.1 mg of clonidine has produced signs of toxicity in children.
If symptoms of poisoning occur following dermal exposure, remove all Catapres‑TTS® transdermal systems. After their removal, the plasma clonidine levels will persist for about 8 hours, then decline slowly over a period of several days. Rare cases of Catapres‑TTS® poisoning due to accidental or deliberate mouthing or ingestion of the patch have been reported, many of them involving children.
There is no specific antidote for clonidine overdosage. Ipecac syrup‑induced vomiting and gastric lavage would not be expected to remove significant amounts of clonidine following dermal exposure. If the patch is ingested, whole bowel irrigation may be considered and the administration of activated charcoal and/or cathartic may be beneficial. Supportive care may include atropine sulfate for bradycardia, intravenous fluids and/or vasopressor agents for hypotension and vasodilators for hypertension. Naloxone may be a useful adjunct for the management of clonidine‑induced respiratory depression, hypotension and/or coma; blood pressure should be monitored since the administration of naloxone has occasionally resulted in paradoxical hypertension. Dialysis is not likely to significantly enhance the elimination of clonidine.
The largest overdose reported to date, involved a 28‑year old male who ingested 100 mg of clonidine hydrochloride powder. This patient developed hypertension followed by hypotension, bradycardia, apnea, hallucinations, semicoma, and premature ventricular contractions. The patient fully recovered after intensive treatment. Plasma clonidine levels were 60 ng/mL after 1 hour, 190 ng/mL after 1.5 hours, 370 ng/mL after 2 hours, and 120 ng/mL after 5.5 and 6.5 hours. In mice and rats, the oral LD50 of clonidine is 206 and 465 mg/kg, respectively.
-
DOSAGE AND ADMINISTRATION
Apply Catapres‑TTS® (clonidine transdermal system) once every 7 days to a hairless area of intact skin on the upper outer arm or chest. Each new application of Catapres‑TTS® should be on a different skin site from the previous location. If the system loosens during 7‑day wearing, the adhesive cover should be applied directly over the system to ensure good adhesion. There have been rare reports of the need for patch changes prior to 7 days to maintain blood pressure control.
To initiate therapy, Catapres‑TTS® dosage should be titrated according to individual therapeutic requirements, starting with Catapres‑TTS®-1. If after one or two weeks the desired reduction in blood pressure is not achieved, increase the dosage by adding another Catapres‑TTS®-1 or changing to a larger system. An increase in dosage above two Catapres‑TTS®-3 is usually not associated with additional efficacy.
When substituting Catapres‑TTS® for oral clonidine or for other antihypertensive drugs, physicians should be aware that the antihypertensive effect of Catapres‑TTS® may not commence until 2‑3 days after initial application. Therefore, gradual reduction of prior drug dosage is advised. Some or all previous antihypertensive treatment may have to be continued, particularly in patients with more severe forms of hypertension.
-
HOW SUPPLIED
Catapres‑TTS®-1, Catapres‑TTS®-2, and Catapres-TTS®-3 are supplied as 4 pouched systems and 4 adhesive covers per carton. See chart below.
Programmed Delivery
Clonidine in vivoPer Day Over 1 Week Clonidine
Content
Size
CodeCatapres-TTS®-1
(clonidine transdermal system)
NDC 82089-101-340.1 mg 2.5 mg 3.5 cm2 BI-31 Catapres-TTS®-2
(clonidine transdermal system)
NDC 82089-102-340.2 mg 5.0 mg 7.0 cm2 BI-32 Catapres-TTS®-3
(clonidine transdermal system)
NDC 82089-103-340.3 mg 7.5 mg 10.5 cm2 BI-33 -
STORAGE AND HANDLING
Store at 25°C (77°F); excursions permitted to 15°- 30°C (59°- 86°F) [see USP Controlled Room Temperature].
Manufactured by:
Alza Corporation,
Vacaville, CA 95688, USACatapres-TTS® is a registered trademark of Boehringer Ingelheim International GmbH used under license
Address medical inquiries to: (866) 380-1080
Copyright © 2021 Technomed Inc.
ALL RIGHTS RESERVED222068/C
-
PATIENT INSTRUCTIONS
Catapres-TTS®
(clonidine transdermal system)(Read the following instructions carefully before using this medication. If you have any questions, please consult with your doctor.)
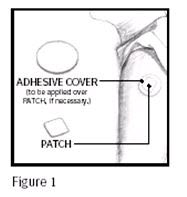
Catapres‑TTS® (clonidine transdermal system) is a square, tan adhesive PATCH containing an active blood‑pressure‑lowering medication. It is designed to deliver the drug into the body through the skin smoothly and consistently for one full week. Normal exposure to water, as in showering, bathing, and swimming, should not affect the PATCH.
The optional white, round ADHESIVE COVER should be applied directly over the PATCH, should the PATCH begin to separate from the skin. The ADHESIVE COVER ensures that the PATCH sticks to the skin. The Catapres‑TTS® PATCH must be replaced with a new one on a fresh skin site if the one in use significantly loosens or falls off.
Skin burns have been reported at the patch site in several patients wearing an aluminized transdermal system during a magnetic resonance imaging scan (MRI). Because the Catapres-TTS® PATCH contains aluminum, it is recommended to remove the system before undergoing an MRI.
Each box of Catapres-TTS® contains two types of pouches:

Figure 2
How to Apply the Catapres-TTS® PATCH
1. Apply the square, tan Catapres‑TTS® PATCH once a week, preferably at a convenient time on the same day of the week (i.e., prior to bedtime on Tuesday of week one; prior to bedtime on Tuesday of week two, etc.).
2. Select a hairless area such as on the upper, outer arm or upper chest. The area chosen should be free of cuts, abrasions, irritation, scars or calluses and should not be shaved before applying the Catapres‑TTS® PATCH. Do not place the Catapres‑TTS® PATCH on skin folds or under tight undergarments, since premature loosening may occur.
3. Wash hands with soap and water and thoroughly dry them.
4. Clean the area chosen with soap and water. Rinse and wipe dry with a clean, dry tissue.
5. Select the pouch with the red and orange colors labeled Catapres-TTS® and open it as illustrated in Figure 3. Remove the square, tan PATCH from the pouch.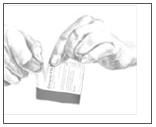
Figure 3
6. Remove the clear plastic protective backing from the PATCH by gently peeling off one half of the backing at a time as shown in Figure 4. Avoid touching the sticky side of the Catapres‑TTS®PATCH.
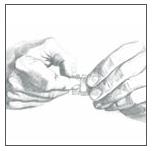
Figure 4
7. Place the Catapres‑TTS® PATCH on the prepared skin site (sticky side down) by applying firm pressure over the PATCH to ensure good contact with the skin, especially around the edges (Figure 5). Discard the clear plastic protective backing and wash your hands with soap and water to remove any drug from your hands.
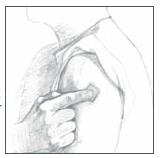
Figure 5
8. After one week, remove the old PATCH and discard it (refer to Instructions for Disposal). After choosing a different skin site, repeat instructions 2 through 7 for the application of your next Catapres‑TTS® PATCH.
What to do if your Catapres-TTS® PATCH becomes loose while wearing:
How to Apply the ADHESIVE COVER
Note: The white, round, ADHESIVE COVER does not contain any drug and should not be used alone. The COVER should be applied directly over the Catapres‑TTS® PATCH only if the PATCH begins to separate from the skin, thereby ensuring that it sticks to the skin for seven full days.
- Wash hands with soap and water and thoroughly dry them.
- Using a clean, dry tissue, make sure that the area around the square, tan Catapres-TTS® PATCH is clean and dry. Press gently on the Catapres‑TTS® PATCH to ensure that the edges are in good contact with the skin.
- Take the white, round, ADHESIVE COVER (Figure 6) from the plain white pouch and remove the paper liner backing from the COVER.
- Carefully center the round, white ADHESIVE COVER over the square, tan Catapres‑TTS® PATCH and apply firm pressure, especially around the edges in contact with the skin.
How should I store the Catapres-TTS® PATCH?
Store the Catapres-TTS® PATCH at room temperature 68°F to 77°F (20°C to 25°C)Instructions for Disposal
KEEP OUT OF REACH OF CHILDRENDuring or even after use, a PATCH contains active medication which may be harmful to infants and children if accidentally applied or ingested. After use, fold in half with the sticky sides together. Dispose of carefully out of reach of children.
Manufactured by:
Alza Corporation,
Vacaville, CA 95688 USACatapres-TTS® is a registered trademark of Boehringer Ingelheim International GmbH used under license
Copyright © 2021 Technomed Inc.
ALL RIGHTS RESERVED222068/C
- PRINCIPAL DISPLAY PANEL - 0.1 mg Carton
- PRINCIPAL DISPLAY PANEL - 0.1 mg Adhesive Cover
- PRINCIPAL DISPLAY PANEL - 0.1 mg Pouch
- PRINCIPAL DISPLAY PANEL - 0.2 mg Carton
- PRINCIPAL DISPLAY PANEL - 0.2 mg Adhesive Cover
- PRINCIPAL DISPLAY PANEL - 0.2 mg Pouch
- PRINCIPAL DISPLAY PANEL carton - 0.3 mg
- PRINCIPAL DISPLAY PANEL - 0.3 mg Adhesive Cover
- PRINCIPAL DISPLAY PANEL - 0.3 mg Pouch
-
INGREDIENTS AND APPEARANCE
CATAPRES-TTS-1
clonidine transdermal system patchProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:82089-101 Route of Administration TRANSDERMAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLONIDINE (UNII: MN3L5RMN02) (CLONIDINE - UNII:MN3L5RMN02) CLONIDINE 0.1 mg in 1 d Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) POLYISOBUTYLENE (1100000 MW) (UNII: FLT10CH37X) POLYISOBUTYLENE (35000 MW) (UNII: 98553S1MHQ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color Score Shape Size Flavor Imprint Code BI;31 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82089-101-34 4 in 1 CARTON 03/01/2022 10/30/2026 1 1 in 1 POUCH 1 1 d in 1 PATCH; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018891 03/01/2022 10/30/2026 CATAPRES-TTS-2
clonidine transdermal system patchProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:82089-102 Route of Administration TRANSDERMAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLONIDINE (UNII: MN3L5RMN02) (CLONIDINE - UNII:MN3L5RMN02) CLONIDINE 0.2 mg in 1 d Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) POLYISOBUTYLENE (1100000 MW) (UNII: FLT10CH37X) POLYISOBUTYLENE (35000 MW) (UNII: 98553S1MHQ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color Score Shape Size Flavor Imprint Code BI;32 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82089-102-34 4 in 1 CARTON 03/01/2022 10/30/2026 1 1 in 1 POUCH 1 1 d in 1 PATCH; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018891 03/01/2022 10/30/2026 CATAPRES-TTS-3
clonidine transdermal system patchProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:82089-103 Route of Administration TRANSDERMAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLONIDINE (UNII: MN3L5RMN02) (CLONIDINE - UNII:MN3L5RMN02) CLONIDINE 0.3 mg in 1 d Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) POLYISOBUTYLENE (1100000 MW) (UNII: FLT10CH37X) POLYISOBUTYLENE (35000 MW) (UNII: 98553S1MHQ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color Score Shape Size Flavor Imprint Code BI;33 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82089-103-34 4 in 1 CARTON 03/01/2022 10/30/2026 1 1 in 1 POUCH 1 1 d in 1 PATCH; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018891 03/01/2022 10/30/2026 Labeler - Technomed Inc. (116982116)